Balance The Following Redox Reaction In Acidic Solution Cr2o7
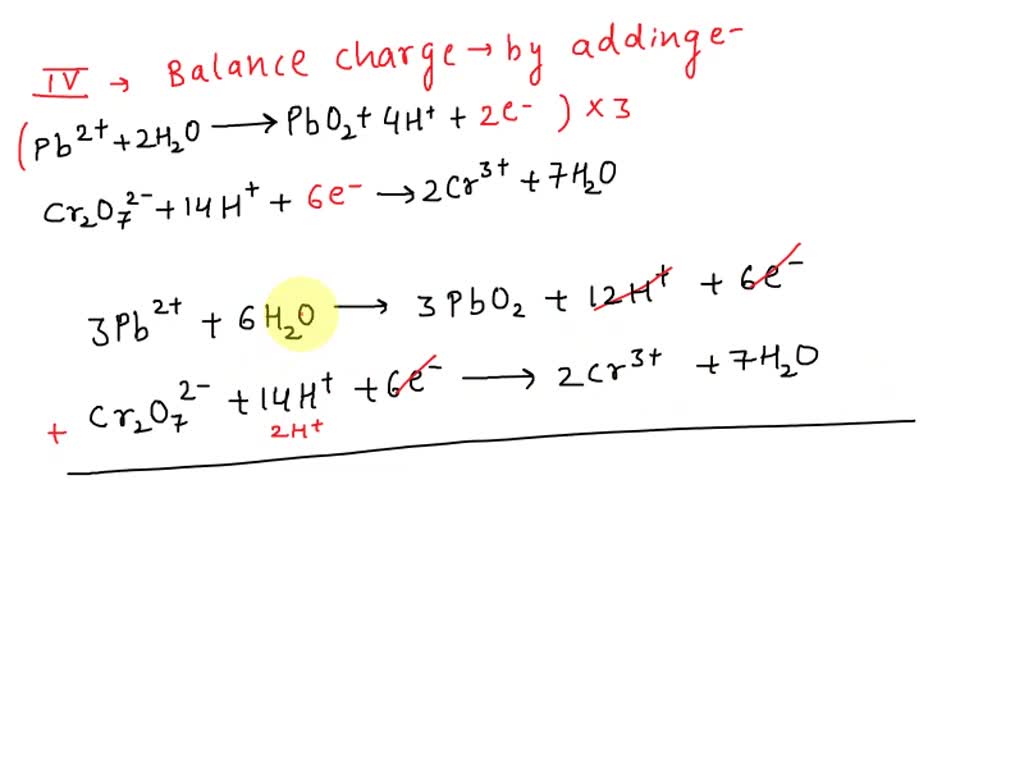
Imagine your favorite recipe. You've got the ingredients, you've got the instructions, but sometimes, things just don't quite add up. Maybe you added too much flour, or not enough sugar, and your cookies turned out… well, let's just say they became "artistic experiments." That's a little like what happens in the wild and wacky world of chemistry when things get out of whack. Today, we're going to peek behind the curtain at one such culinary (or rather, chemical) mishap, and how clever scientists, like tiny kitchen wizards, put it all back together. Our star of the show? A rather dramatic character named Dichromate, or Cr2O7, as it's known to its friends. Think of Dichromate as a super-charged ingredient, always eager to join the party and lend a hand – or in this case, a lot of electrons!
Now, Dichromate isn't usually a solo act. It likes to hang out with other chemical characters. But when it gets into a situation that's a bit like a sour lemonade stand – an acidic solution, where things are a bit zesty and tart – oh boy, does it get excited! It's like a playful puppy who suddenly discovers a squeaky toy. It starts bouncing around, ready to engage in a bit of chemical give-and-take. And what's this give-and-take all about? It's about something called redox reactions. Don't let the fancy name scare you! Think of it like a game of "hot potato" with tiny particles called electrons. Some characters in the chemical kitchen are really good at giving away electrons, and others are absolutely thrilled to take them. It’s a constant exchange, a chemical ballet of sorts.
Our star, Dichromate (Cr2O7), is a bit of a diva. It loves to get involved when the mood is just right – especially in a zesty, acidic solution!
The tricky part is, sometimes, when Dichromate gets involved in this electron exchange, things can get a little… chaotic. It's like trying to bake a cake with too many cooks in the kitchen. Everyone's adding their own little bits, and before you know it, the batter is all over the place. In our chemical kitchen, Dichromate, in its energetic state within the acidic solution, can get a bit… unorganized. It's not that it's trying to be naughty; it's just a very enthusiastic participant in the redox dance. It wants to share its electrons, but it also wants to make sure everyone is playing fair.
So, what happens? Well, when scientists look at the mess, they see a lot of chemical ingredients floating around, and they know that the electrons haven't been shared out perfectly. It's like looking at your baking ingredients after a whirlwind of mixing and realizing you've got a bit of a surplus of eggs on one side and a shortage of flour on the other. The goal is to make everything just right, so the reaction can proceed smoothly, like a perfectly baked cookie, golden brown and delicious.

This is where the real magic, and a touch of delightful problem-solving, comes in. Scientists, armed with their knowledge and a good dose of patience, step in to "balance" the reaction. Think of it as tidying up the chemical kitchen after a particularly enthusiastic baking session. They need to make sure that every atom, every electron, is accounted for. It's like counting your ingredients before you start, and then again after you've finished to make sure nothing's gone missing.
For our friend Dichromate (Cr2O7) in its acidic playground, balancing its redox reaction is like orchestrating a symphony. There are instruments (atoms) playing different notes (electrons), and they all need to come together in perfect harmony. The scientists essentially give the reaction a set of instructions, a gentle nudge, to ensure that the electrons are passed around fairly. They don't change the fundamental nature of the ingredients; they just help them play nicely together. It's about ensuring that for every electron that's given away, another one is perfectly received.
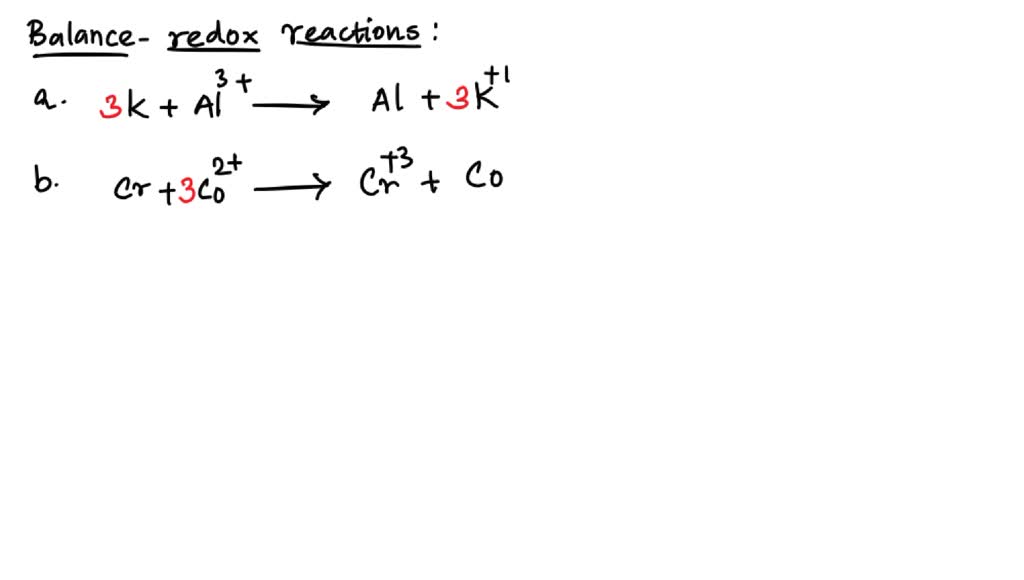
The beauty of it all is that even though it sounds complex, the underlying principle is wonderfully simple: fairness. In the chemical world, just like in life, balance is key. And when we talk about balancing a redox reaction like the one involving Dichromate (Cr2O7) in an acidic solution, we're really talking about bringing order to potential chaos, ensuring that everyone gets their fair share of the electron-y goodness.
It’s a bit like watching a skilled chef, who, after a flurry of activity, calmly surveys their workspace, makes a few adjustments, and voilà! A masterpiece. The scientists, in their own way, do the same. They look at the chaotic dance of atoms and electrons and, with a few calculated moves, bring everything into perfect equilibrium. The result? A reaction that proceeds as intended, a testament to the underlying order that governs even the most energetic chemical interactions. It’s a quiet triumph, a reminder that even in the tiniest of worlds, a little bit of balance can lead to something truly remarkable. And who knows, maybe that perfectly balanced chemical reaction is the first step to creating something wonderful, just like your favorite perfectly baked cookie.
