Balance The Following Redox Equations By The Half Reaction Method

Hey there, science curious folks! Ever looked at a chemical reaction and thought, "Whoa, that looks complicated"? You're not alone! Today, we're going to dive into something super neat called balancing redox equations using the half-reaction method. Don't let the fancy name scare you off. Think of it like solving a puzzle, or maybe even a detective story, where we're figuring out how atoms are playing tug-of-war with electrons.
So, what exactly is a redox reaction? It's basically a chemical reaction where electrons get passed around. The word "redox" is a mashup of reduction and oxidation. One thing loses electrons (gets oxidized), and another thing gains electrons (gets reduced). It's like a cosmic electron exchange program!
Now, why do we need to balance these equations? Just like in any good story, everything needs to add up, right? We need to make sure that the number of atoms of each element is the same on both sides of the equation, and crucially, that the total charge is also balanced. It’s like making sure you have an equal number of cookies going into the oven as you have coming out – nobody likes a cookie deficit!
The half-reaction method is our special tool for tackling these electron-swapping situations. It breaks down the big, scary redox reaction into two smaller, more manageable parts: the oxidation half-reaction and the reduction half-reaction. It's like taking a complex recipe and separating it into "prepare the sauce" and "cook the main dish" steps. Much easier to follow, wouldn't you say?
Let's get our hands dirty with a hypothetical example. Imagine we have this un-balanced beauty:
Cu + HNO3 -> Cu(NO3)2 + NO + H2O
Looks a bit chaotic, doesn't it? Like a toddler went wild with the crayons. We need to bring some order to this chemical chaos. The half-reaction method is our superhero cape for this mission.
Step 1: Identify the Species Being Oxidized and Reduced
This is our detective work. We need to figure out which elements are changing their "electron ownership." We do this by assigning oxidation states to each atom. It’s kind of like giving each atom a score based on how many electrons it’s “holding onto” or “giving away.”
In our example:
- Copper (Cu) starts with an oxidation state of 0 (since it's an element by itself). It ends up in Cu(NO3)2 with an oxidation state of +2. So, copper lost electrons. That's oxidation!
- Nitrogen (N) in HNO3 has an oxidation state of +5. In NO, it ends up with an oxidation state of +2. So, nitrogen gained electrons. That's reduction!
See? We've already uncovered our first clues!
Step 2: Write the Unbalanced Half-Reactions
Now we separate the electron-transferring action into two distinct paths. We focus only on the atoms that are changing oxidation states, ignoring the spectators for a moment. It’s like splitting the dance floor into a hip-hop section and a ballet section – everyone still dances, but in their own style!
Our oxidation half-reaction:
Cu -> Cu^2+
And our reduction half-reaction:
HNO3 -> NO

We're getting closer!
Step 3: Balance Atoms (Except H and O)
This is the easy part. We just make sure the number of atoms of each element (other than hydrogen and oxygen) is the same on both sides. In our copper example, the copper atoms are already balanced. For the nitrogen in the reduction half-reaction, they are also already balanced (one on each side). So, this step is a breeze here.
Step 4: Balance Oxygen Atoms by Adding H2O
Oxygen atoms are a bit more… flexible. We balance them by adding water (H2O) molecules to the side that needs more oxygen. Think of water as a little oxygen delivery service!
Our reduction half-reaction still looks like this:
HNO3 -> NO
On the left, we have 3 oxygen atoms in HNO3. On the right, in NO, we only have 1. We need 2 more oxygen atoms on the right. So, we add 2 H2O molecules:
HNO3 -> NO + 2H2O
Ta-da! Oxygen is now balanced in our reduction half.
Step 5: Balance Hydrogen Atoms by Adding H+
Now, for the hydrogen atoms. In acidic solutions (which is often the case with HNO3), we balance hydrogen by adding hydrogen ions (H+) to the side that needs more hydrogen. It’s like adding extra hydrogen scaffolding to hold things up.
Our oxidation half-reaction still looks like:
Cu -> Cu^2+
No hydrogen here, so we don't need to do anything. Easy peasy!
Our reduction half-reaction now looks like:
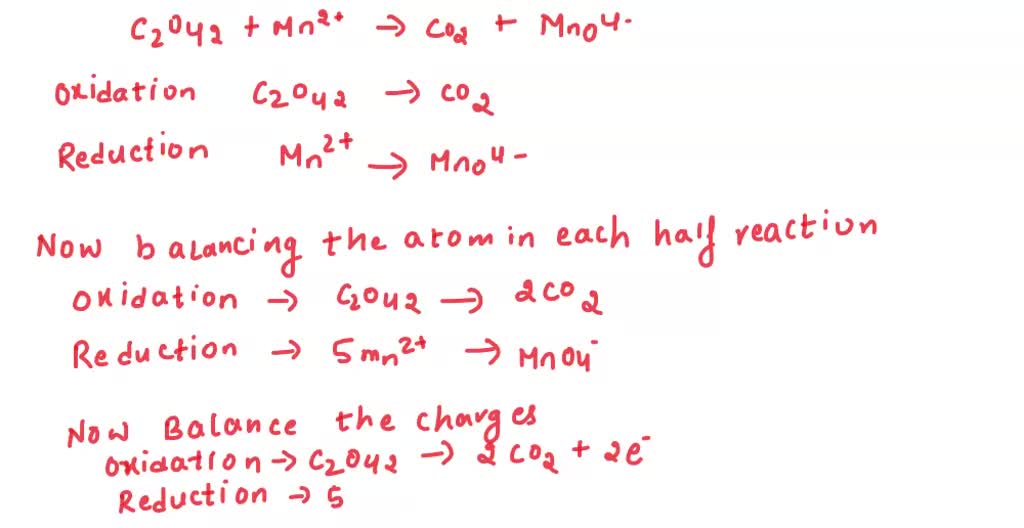
HNO3 -> NO + 2H2O
On the left, we have 1 hydrogen atom. On the right, we have 4 hydrogen atoms (from the 2H2O). We need 3 more hydrogen atoms on the left. So, we add 3H+:
HNO3 + 3H+ -> NO + 2H2O
Hydrogen is now happy!
Step 6: Balance the Charges by Adding Electrons (e-)
This is where the real electron action happens. We balance the electrical charge on both sides of each half-reaction by adding electrons (e-). Remember, electrons have a negative charge (-1). We add them to the side that is more positive to make the charges equal.
For our oxidation half-reaction:
Cu -> Cu^2+
The left side has a charge of 0. The right side has a charge of +2. To make them equal, we need to add 2 electrons to the right side:
Cu -> Cu^2+ + 2e-
Copper lost 2 electrons, making it an ion! This makes perfect sense.
For our reduction half-reaction:
HNO3 + 3H+ -> NO + 2H2O
Let's calculate the total charge on each side:
- Left side: Charge of HNO3 (0) + charge of 3H+ (3 * +1 = +3) = +3
- Right side: Charge of NO (0) + charge of 2H2O (0) = 0
The left side is more positive. We need to add 3 electrons to the left side to make the charges equal:
HNO3 + 3H+ + 3e- -> NO + 2H2O
Nitrogen gained 3 electrons! This is where the reduction happens.
Step 7: Make the Number of Electrons Equal in Both Half-Reactions
Now, this is super important. In the overall reaction, electrons are transferred, not created or destroyed. So, the number of electrons lost in oxidation must equal the number of electrons gained in reduction. We achieve this by multiplying one or both half-reactions by a suitable integer.
Our oxidation half-reaction lost 2 electrons:
Cu -> Cu^2+ + 2e-
Our reduction half-reaction gained 3 electrons:
HNO3 + 3H+ + 3e- -> NO + 2H2O
To make the electrons equal, we need to find the least common multiple of 2 and 3, which is 6. So, we multiply the oxidation half-reaction by 3 and the reduction half-reaction by 2:
Multiply oxidation by 3:
3Cu -> 3Cu^2+ + 6e-
Multiply reduction by 2:

2HNO3 + 6H+ + 6e- -> 2NO + 4H2O
See? Now we have 6 electrons on each side. We're almost there!
Step 8: Add the Half-Reactions Together
This is the grand finale! We add the two balanced half-reactions together. We combine everything on the left sides and everything on the right sides. Then, we cancel out anything that appears on both sides. Those electrons we worked so hard to balance? They should disappear here!
Adding them up:
(3Cu) + (2HNO3 + 6H+ + 6e-) -> (3Cu^2+ + 6e-) + (2NO + 4H2O)
Now, let's cancel the 6e- from both sides:
3Cu + 2HNO3 + 6H+ -> 3Cu^2+ + 2NO + 4H2O
And there you have it! A beautifully balanced redox equation using the half-reaction method. It looks way cleaner now, doesn't it? Like a perfectly organized desk after a messy project.
Step 9: Check Your Work (Just to Be Sure!)
Before we celebrate, a quick check. Let's count the atoms and charges on both sides:
- Cu: Left = 3, Right = 3 (Balanced!)
- H: Left = 6, Right = 4 * 2 = 8 (Wait, something's not quite right here. Ah, in the original equation, HNO3 was not just acting as a source of H+ for reduction. It was also providing nitrate ions (NO3-). Let's go back and adjust for that.)
This is a common hiccup, and it’s a great reminder that chemistry can have its quirks! In a more complex scenario, especially involving species like nitrate where oxygen and nitrogen are present in a group, we might need to account for the spectator ions or consider balancing in basic solution if that were indicated. For simplicity in this introductory example, let's assume we are in a context where the H+ and NO3- are readily available and the focus is on the electron transfer. If this were a real lab experiment, we’d carefully check the reaction conditions!
But the principle of balancing atoms and charges through half-reactions holds strong! The process we followed is the core of the method.
Why is this so cool? Because it gives us a fundamental understanding of how chemical reactions happen at the most basic level – the movement of electrons. It’s like understanding the plumbing system that makes a whole city work. From batteries to photosynthesis, redox reactions are happening all around us, driving incredibly important processes.
So, the next time you see a complex chemical equation, don't sweat it. Just remember the half-reaction method: break it down, balance the atoms, balance the electrons, and put it all back together. It’s a powerful way to unlock the secrets of chemistry, one electron at a time. Happy balancing!
