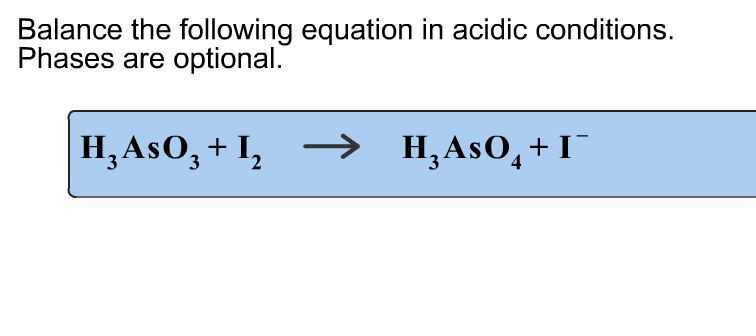
Balance The Following Equation In Acidic Conditions H3aso3 I2
Hey there, science curious folks! Ever feel like life is a bit of a balancing act? You know, juggling work, family, that ever-growing to-do list, and maybe even trying to remember to water the plants? Well, guess what? Even the tiniest things in the universe, like chemical reactions, need a good old balancing act too! Today, we're going to peek behind the curtain at one of these balancing acts, specifically involving something called H₃AsO₃ and I₂. Now, before you go thinking this is going to be a dry chemistry lecture, stick with me! We're going to make this as easy-going and relatable as a Sunday morning coffee. And believe it or not, understanding this little dance between molecules can actually make you feel a bit more connected to the world around you, and why things happen the way they do.
So, what are these mysterious H₃AsO₃ and I₂? Think of H₃AsO₃ as Arsenous Acid. Don't let the "arsenous" scare you! It's a compound, and in this context, it's like one of the players in our reaction. Imagine it’s like a slightly shy guest at a party, holding onto its hydrogen friends (those 'H's). On the other side, we have I₂, which is Iodine. Iodine is a bit more energetic, like that friend who’s always ready for an adventure. When these two get together, they’re going to have a little chemical "chat" or reaction. But just like a good conversation, it needs to be balanced. We can't have one person talking way too much and the other not saying anything, right? The universe likes things to be fair and even-steven.
Now, why should you, my lovely everyday reader, care about balancing chemical equations? Isn't that for folks in lab coats with goggles? Well, think about it like this: everything you see, touch, and even are is a result of countless chemical reactions. When you bake a cake, you're balancing ingredients to get a delicious outcome. When your body digests food, it’s a series of chemical reactions working in harmony. Even the rust on an old bike is a chemical reaction. So, understanding the principles of how these reactions work, like balancing, gives you a little peek into the fundamental language of the universe. It’s like learning a few words of a new language – it opens up new understandings and makes the world feel a little less mysterious and a lot more awesome.
In this particular reaction, we're doing our balancing in acidic conditions. What does that mean? Imagine you’re trying to have a chat with your friend, and you both agree to talk in a certain room, maybe a cozy café with lots of comfy chairs. Acidic conditions are like that cozy café for our chemical reactants. They provide a specific environment where the reaction can happen smoothly. In chemistry, this usually means there are extra hydrogen ions (H⁺) hanging around, ready to lend a hand or, in this case, a proton. Think of them as the helpful waiters in our café, making sure everything runs just right.
Let's Get Down to Business (Without Getting Messy!)
Okay, so we have our players: H₃AsO₃ and I₂. And we're in our nice, acidic café. What’s going to happen? Generally, in this setup, the Arsenous Acid (H₃AsO₃) is going to get oxidized, and the Iodine (I₂) is going to get reduced. Sounds fancy, right? Let's break it down with a relatable analogy.
Imagine H₃AsO₃ is like a person who’s holding onto some valuable items (electrons, in chemistry speak). They're feeling a bit shy and keeping them close. Iodine (I₂) is like a magnet, and it loves to snatch up those valuable items. So, the Iodine essentially takes some of those electrons from the Arsenous Acid. When H₃AsO₃ loses electrons, it's called oxidation. When I₂ gains electrons, it’s called reduction. It’s like a little give-and-take happening.
But, as we said, the universe likes balance. We can't just have one molecule giving away all its stuff and another hoarding it all. We need to make sure that for every "give," there's a corresponding "take," and that all the atoms end up in the right places. Think of it like a trade deal. If one company is selling a lot of widgets, the other company needs to be buying a corresponding amount, and the money needs to flow back and forth to keep things fair.
The Balancing Act: Step-by-Step (The Fun Version!)
The official way to balance equations involves using something called "oxidation states" and "half-reactions." Now, I know that sounds like it’s from a textbook, but we can think of it like this:
First, we need to figure out who’s giving what and who’s taking what. We saw that Arsenous Acid (H₃AsO₃) is losing something, and Iodine (I₂) is gaining. So, we'll represent this as:
Step 1: Write down the unbalanced reaction.

H₃AsO₃ + I₂ → products
We know the Iodine will become something like iodide ions (I⁻), and the Arsenous Acid will transform into Arsenic Acid (H₃AsO₄). So, our unbalanced equation looks a bit more like this:
H₃AsO₃ + I₂ → H₃AsO₄ + I⁻
Now, here’s where the balancing act really kicks in. We need to make sure the number of each type of atom is the same on both sides of the arrow. It’s like a recipe: you can’t just throw in a cup of flour and expect a cake if the recipe calls for two cups. You need the right proportions!
Step 2: Balance the atoms that are changing.
The Iodine is changing from I₂ to I⁻. We have two I atoms in I₂ and only one in I⁻. So, we need to put a ‘2’ in front of the I⁻ to balance the iodine atoms:
H₃AsO₃ + I₂ → H₃AsO₄ + 2I⁻

The Arsenic atom (As) is already balanced – there’s one on each side. Phew, one less thing to worry about!
Step 3: Balance the oxygen atoms using water (H₂O).
Let’s count the oxygens. On the left, we have 3 oxygens in H₃AsO₃. On the right, we have 4 oxygens in H₃AsO₄. We’re short one oxygen on the left. To add that oxygen, we’ll add a water molecule (H₂O) to the side that needs it. But wait! Adding water also adds hydrogen. This is like adding an ingredient that comes with a little bonus: more of another ingredient!
Wait a minute! In this specific reaction, the Arsenic is going from a +3 oxidation state to a +5 oxidation state. The Iodine is going from 0 to -1. So, let's think about what's actually happening in terms of electron transfer. Arsenous acid (H₃AsO₃) loses two electrons to become arsenate (H₃AsO₄). Iodine (I₂) gains two electrons to become two iodide ions (2I⁻).
So, the core reaction looks like:
H₃AsO₃ + I₂ → H₃AsO₄ + 2I⁻
Let’s re-evaluate the oxygen balancing. We have 3 oxygens on the left and 4 on the right. We're short one oxygen on the left. To add that oxygen, we add a water molecule (H₂O) to the left side.

H₃AsO₃ + H₂O + I₂ → H₃AsO₄ + 2I⁻
Step 4: Balance the hydrogen atoms using H⁺ ions (because we're in acidic conditions!).
Now, let’s count our hydrogens. On the left, we have 3 in H₃AsO₃, 2 in H₂O, for a total of 5. On the right, we have 3 in H₃AsO₄. We’re short 2 hydrogens on the right. Since we're in acidic conditions, we can add hydrogen ions (H⁺) to balance them. So we add 2H⁺ to the right side.
H₃AsO₃ + H₂O + I₂ → H₃AsO₄ + 2I⁻ + 2H⁺
Step 5: Check the charge balance!
This is super important! The total charge on the left side needs to equal the total charge on the right side. Let's see:
Left side: (0 from H₃AsO₃) + (0 from H₂O) + (0 from I₂) = 0

Right side: (0 from H₃AsO₄) + (2 * -1 from 2I⁻) + (2 * +1 from 2H⁺) = -2 + 2 = 0
Ta-da! The charges are balanced! We have 0 on both sides. This means our equation is now correctly balanced.
So, the balanced equation is:
H₃AsO₃ + H₂O + I₂ → H₃AsO₄ + 2I⁻ + 2H⁺
It might look a little complicated, but each step is like putting a puzzle piece in the right spot. We make sure the atoms are there, then we add water for oxygen, then hydrogen ions for hydrogen, and finally, we double-check that the overall "charge" of the reaction is fair and balanced. It's all about making sure everything is accounted for!
Why Does This Matter to You?
Beyond the satisfaction of a neatly balanced equation, understanding these processes helps us appreciate the intricate workings of the world. When you learn about how chemicals interact, you start to see connections everywhere. This specific reaction, for instance, is related to redox reactions, which are fundamental to everything from batteries powering your phone to the way your own cells generate energy. It's the unseen choreography of the universe, playing out constantly.
Think of it like understanding how a recipe works. You don't need to be a master chef to know that if you forget the baking soda, your cake won't rise. Similarly, you don't need to be a chemist to appreciate that reactions require precise conditions and balanced components to work. It's about understanding the underlying principles that govern our physical reality. It’s about seeing the beauty in the order, even in the smallest of chemical interactions.
So, the next time you see something happening in the world, whether it's a leaf changing color, a light turning on, or even just water boiling, you can have a little nod to the amazing, balanced dance of molecules that's making it all possible. And who knows, maybe this little dive into balancing equations will make you feel a bit more scientifically empowered and curious about the world around you. Happy balancing, folks!
