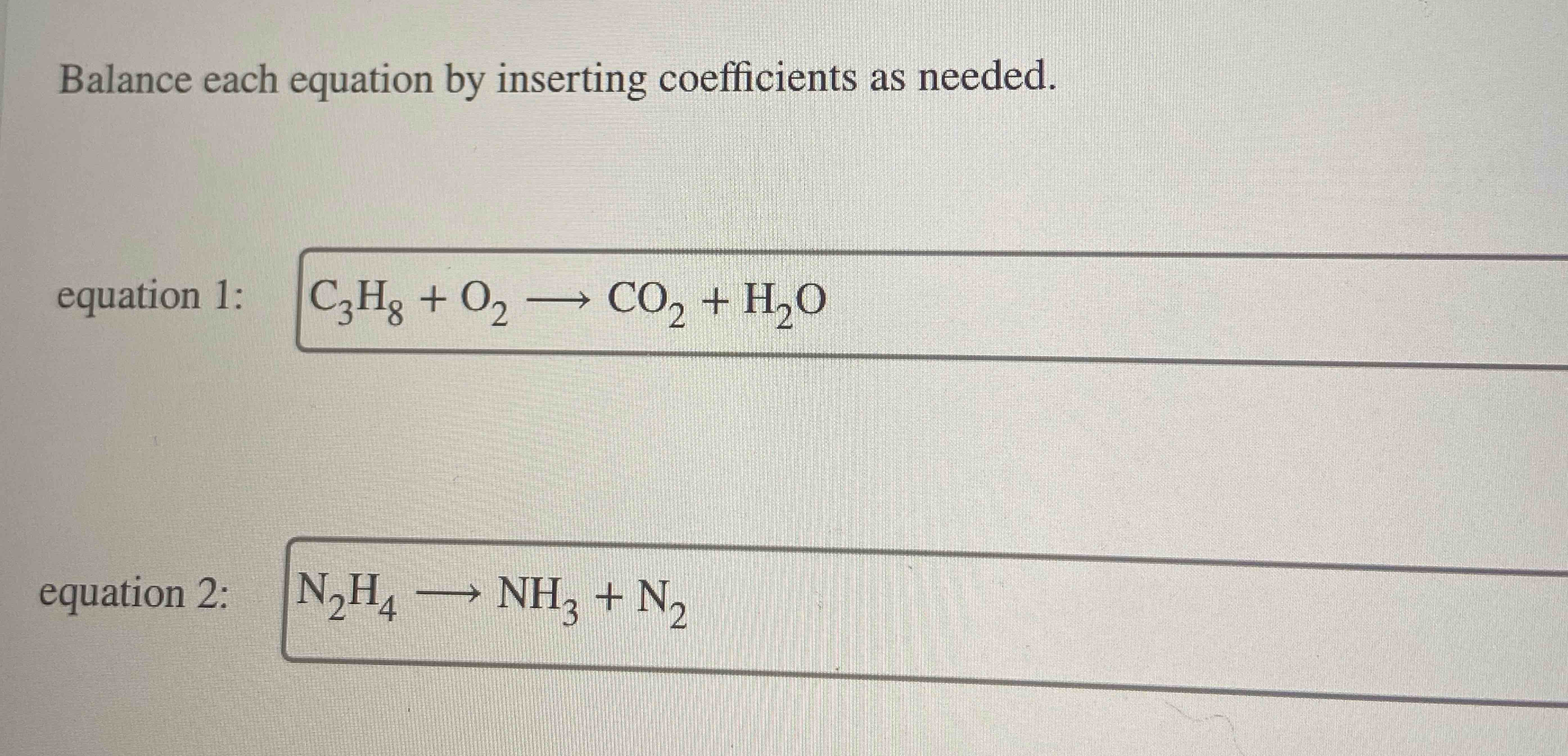
Balance The Equations By Inserting Coefficients As Needed

Ever looked at a science experiment on TV and thought, "Wow, that looks complicated!"? Well, sometimes the magic of chemistry, especially when it comes to making things react just right, is all about getting the numbers to balance. Think of it like a recipe: if you have too much of one ingredient, the final dish might not turn out as planned. In the world of chemistry, we call this balancing chemical equations, and it's actually a really neat puzzle that’s both fun and incredibly useful.
So, what exactly is balancing an equation? Imagine you're building with LEGOs. You start with a bunch of bricks, and you want to assemble them into a specific model. A chemical equation is like the blueprint, showing you what you start with (reactants) and what you end up with (products). Balancing the equation means making sure you have the exact same number of each type of atom on both sides. It’s like saying, "Okay, I used 4 red LEGO bricks to build this part, so I need to make sure there are also 4 red LEGO bricks in the final structure." This principle, known as the Law of Conservation of Mass, is fundamental to all of chemistry – nothing is created or destroyed, it just rearranges!
Why is this so cool? For beginners in science, it's a gentle introduction to the quantitative side of chemistry, showing them that chemical reactions follow strict mathematical rules. It’s a great way to develop problem-solving skills and a sense of logical thinking. For families, it can be a fantastic educational activity. You can turn it into a game! Imagine trying to balance the equation for making water from hydrogen and oxygen: H₂ + O₂ → H₂O. It looks a bit off, right? You need two hydrogens and two oxygens on the left, but only two hydrogens and one oxygen on the right. Balancing it means adding coefficients (those numbers we insert) to make it H₂ + O₂ → 2H₂O, and then realizing you actually need 2H₂ + O₂ → 2H₂O. See? It’s like a little detective game!
For hobbyists, whether it’s brewing, gardening (understanding fertilizers!), or even just a keen interest in how things work, balancing equations helps you understand the proportions involved. It gives you a deeper appreciation for the chemical processes happening around you. For example, understanding how batteries work or how fuels burn efficiently all relies on balanced equations.
Getting started is easier than you think! Don't be intimidated by the chemical formulas. Focus on the elements. For instance, in the water example (H₂ + O₂ → H₂O), you’ve got Hydrogen (H) and Oxygen (O). You just need to count them on each side. If you’re stuck, try tackling simpler equations first. Think of it like learning to count before you do calculus. Websites and textbooks often have plenty of practice problems, and many even offer step-by-step guides.
Ultimately, balancing chemical equations is more than just a classroom exercise. It’s a fundamental concept that unlocks a deeper understanding of the world. It’s a satisfying puzzle that rewards careful observation and logical thinking, making the often-mysterious world of chemistry feel accessible and engaging.
