Balance The Equation Using Lowest Whole-number Coefficients
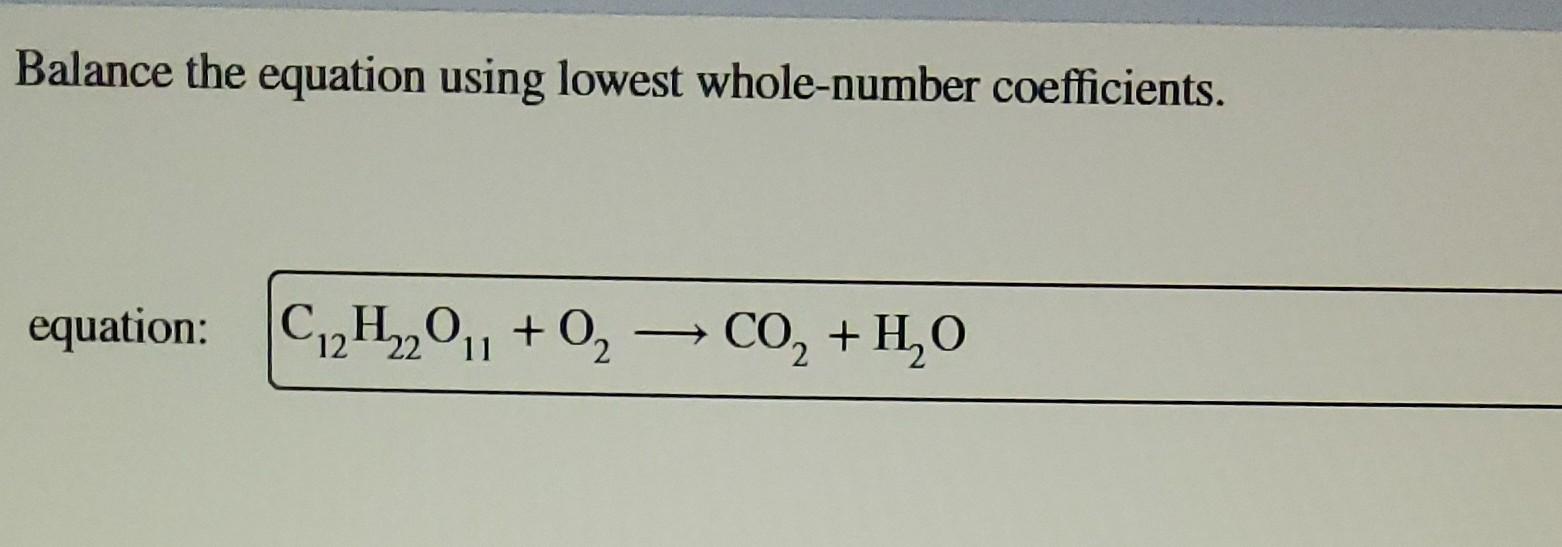
Ever feel like your life is a bit of a jumbled mess? Like you’ve got too much of one thing and not enough of another, and it’s all just… off? Maybe you've got a closet overflowing with mismatched socks, or a pantry stocked with fifty cans of tuna but no pasta to go with it. That, my friends, is the everyday version of an unbalanced equation. And just like you’d try to sort out that sock drawer or whip up a surprisingly decent tuna-noodle casserole, we’re going to do the same for chemical reactions.
Think of a chemical reaction like a recipe. You can’t just throw random ingredients into a pot and expect a gourmet meal. You need the right proportions, right? You wouldn't make a cake with one cup of flour and twenty eggs, would you? That’d be… a disaster. Or a really weird, eggy pancake. Either way, not ideal. Similarly, in the world of chemistry, the tiny particles called atoms need to be in the right number on both sides of the equation to make sure everything is accounted for. That’s where the magic of "balancing the equation using the lowest whole-number coefficients" comes in.
It sounds a bit fancy, doesn't it? "Lowest whole-number coefficients." It's like being told you need to use the exact right amount of sprinkles on your cupcake, not just a haphazard dusting. But really, it’s just about making sure that for every atom that goes into the reaction, the same number of atoms of that same type comes out. It's like a cosmic accounting system, ensuring that nothing is created or destroyed, just rearranged. Pretty neat, huh?
Let’s imagine you’re baking cookies. You’ve got your flour, your sugar, your eggs, and your chocolate chips. Let’s say your recipe calls for 2 cups of flour, 1 egg, and 10 chocolate chips to make, let's say, a dozen perfect cookies. Now, what if you decide you want to make two dozen cookies? You’d double everything, right? You'd need 4 cups of flour, 2 eggs, and 20 chocolate chips. You’re not suddenly going to need 3 cups of flour and 1.5 eggs (ew, raw egg). You need whole numbers, and you need them in the right ratio. That’s the essence of balancing chemical equations!
The Recipe for Disaster (or Success!)
So, what does an unbalanced equation look like? Imagine you're trying to make water (H₂O). You know you need hydrogen (H) and oxygen (O). The simplest way to combine them is for two hydrogen atoms to hook up with one oxygen atom. So, the basic "recipe" looks something like this:
H₂ + O₂ → H₂O
Now, let's play chef and see what we've got. On the left side (the ingredients), we have two hydrogen atoms (that’s the H₂) and two oxygen atoms (the O₂). On the right side (the finished product), we have two hydrogen atoms and one oxygen atom (the H₂O). Uh oh. Where did that other oxygen atom go? Did it stage a dramatic exit? Did it elope with a stray nitrogen atom from the air? This is our unbalanced equation, and it’s like trying to make water with a missing ingredient.
This is where we bring in our trusty "coefficients." Think of coefficients as the little numbers you put in front of the chemical formulas. They're like the serving sizes on your recipe. They tell you how many molecules of that particular substance you need. We can't change the little numbers within the formulas (those are called subscripts, and they tell you how many atoms are in one molecule of that substance – like the '2' in H₂O tells you there are two hydrogen atoms in one water molecule). Messing with those is like trying to make a car by adding another wheel to a bicycle; it just fundamentally changes what you’re making.
Let's Get Our Hands Dirty (with Numbers!)
We need to make sure our oxygen atoms are happy. We've got two on the left and only one on the right. The easiest way to fix this is to put a coefficient of '2' in front of the H₂O on the right side. This tells us we need two molecules of water for every molecule of hydrogen and oxygen we start with.

H₂ + O₂ → 2H₂O
Now, let's count again. On the left, we still have 2 hydrogen atoms and 2 oxygen atoms. On the right, we now have… two molecules of water. And each water molecule has 2 hydrogen atoms and 1 oxygen atom. So, in total on the right, we have 2 * 2 = 4 hydrogen atoms and 2 * 1 = 2 oxygen atoms.
Our oxygen is balanced! We have 2 on the left and 2 on the right. Victory! But wait, our hydrogen is now out of whack. We have 2 hydrogen atoms on the left and 4 on the right. It’s like we went to make two dozen cookies and accidentally ended up with four dozen!
No worries, we just need to adjust our hydrogen "ingredient" on the left. We need 4 hydrogen atoms to match the 4 we have on the right. Since hydrogen comes in pairs (H₂), we need two of those pairs. So, we put a coefficient of '2' in front of the H₂ on the left.
2H₂ + O₂ → 2H₂O
Let's do a final count. On the left: 2 molecules of H₂, which is 2 * 2 = 4 hydrogen atoms. And 1 molecule of O₂, which is 2 oxygen atoms. On the right: 2 molecules of H₂O, which is 2 * 2 = 4 hydrogen atoms and 2 * 1 = 2 oxygen atoms.

Voilà! We have 4 hydrogen atoms and 2 oxygen atoms on both sides. The equation is balanced using the lowest whole-number coefficients. We've successfully made water without any rogue atoms disappearing or mysteriously appearing. It’s like perfectly portioning out your ingredients so nothing goes to waste and you get exactly what you intended.
Why Bother? It's Just Tiny Stuff, Right?
You might be thinking, "This is all well and good for water, but why does it matter in the grand scheme of things?" Well, imagine you're a scientist working in a pharmaceutical lab. You're trying to synthesize a life-saving drug. If your chemical reactions aren't balanced, you might end up with a product that's impure, or worse, completely inactive. It's like baking a cake for a wedding and accidentally leaving out the baking soda – it might look like a cake, but it's going to be a sad, dense brick. Not exactly what you want for a celebratory occasion!
Or think about industrial processes. Factories that produce everything from plastics to fertilizers rely on precise chemical reactions. If those reactions are unbalanced, you could be wasting precious raw materials, producing toxic byproducts, or just not getting the yield you need. It’s like trying to run a bakery where you keep accidentally throwing away half your flour – you’re going to go broke pretty quickly.
Balancing equations ensures that we’re being efficient and responsible. It’s about understanding the fundamental principles of how matter interacts. It’s the scientific equivalent of not being wasteful, of making sure every atom plays its part and gets accounted for. It's about order in the universe, from the smallest particles to the grandest explosions (which, by the way, also follow these rules!).
When Things Get a Bit More "Chopped"
Sometimes, it’s not as simple as just adding a ‘2’. You might encounter reactions where you need bigger numbers, or you have multiple elements to juggle. Let’s take the combustion of propane (C₃H₈), which is like burning gas in your BBQ.
C₃H₈ + O₂ → CO₂ + H₂O
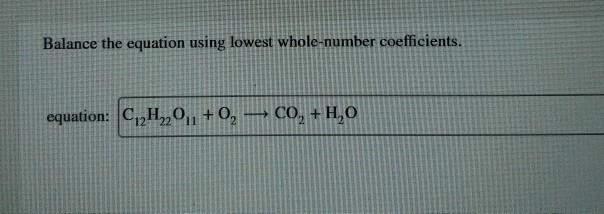
Let’s count: * Carbon (C): 3 on the left, 1 on the right. * Hydrogen (H): 8 on the left, 2 on the right. * Oxygen (O): 2 on the left, 2 in CO₂ + 1 in H₂O = 3 on the right.
This is a bit like trying to sort out a truly chaotic junk drawer. We’ve got a few different things to adjust!
Let’s start with carbon. We have 3 carbons on the left, so we need 3 molecules of CO₂ on the right to match them.
C₃H₈ + O₂ → 3CO₂ + H₂O
Now, carbon is balanced (3 on each side). Let's look at hydrogen. We have 8 hydrogens on the left. Since H₂O has 2 hydrogens, we need 4 molecules of H₂O to get 8 hydrogens (4 * 2 = 8).
C₃H₈ + O₂ → 3CO₂ + 4H₂O

Now, carbon is balanced (3 and 3). Hydrogen is balanced (8 and 8). But look at our oxygen! On the right side, we have (3 molecules of CO₂ * 2 oxygens/CO₂) + (4 molecules of H₂O * 1 oxygen/H₂O) = 6 + 4 = 10 oxygen atoms.
On the left, we only have 2 oxygen atoms in our O₂ molecule. We need 10 oxygen atoms to match. Since oxygen comes in pairs (O₂), we need 5 molecules of O₂ to get 10 oxygen atoms (5 * 2 = 10).
C₃H₈ + 5O₂ → 3CO₂ + 4H₂O
Let’s do our final check: * Carbon (C): Left = 3, Right = 3. (Balanced!) * Hydrogen (H): Left = 8, Right = 4 * 2 = 8. (Balanced!) * Oxygen (O): Left = 5 * 2 = 10, Right = (3 * 2) + (4 * 1) = 6 + 4 = 10. (Balanced!)
See? It’s like a puzzle. You adjust one piece, and it might throw another piece off. But with a little patience and systematic shuffling, you can get everything to fit perfectly. It’s a satisfying feeling, like finally finding that one last matching sock in the laundry abyss.
Tips for the Savvy Balancer
Here are a few tricks that might make your life easier:
- Balance the tricky stuff first: Elements that appear in only one reactant and one product are usually good starting points. Elements that appear in multiple places (like oxygen in our propane example, which is in O₂, CO₂, and H₂O) are often best left for later.
- Metals before non-metals: Sometimes, it’s helpful to tackle metals first, then non-metals, and finally elements like oxygen and hydrogen.
- Treat polyatomic ions as single units: If you have a group of atoms that sticks together throughout the reaction (like sulfate, SO₄²⁻), you can balance it as a single unit instead of balancing the individual sulfur and oxygen atoms. It’s like treating a pre-made sandwich as one item, rather than dissecting it into bread, filling, and garnish.
- Fractions are okay... temporarily: Sometimes, you might end up with a fractional coefficient. For example, you might get 1.5 O₂. If that happens, just multiply the entire equation by the denominator of the fraction (in this case, 2) to get whole numbers. It's like if your recipe calls for 1.5 cups of sugar, and you decide to make double the recipe to avoid the fraction – you'd use 3 cups.
- Practice makes perfect (or at least better): The more you do it, the more intuitive it becomes. It’s like learning to ride a bike; at first, you wobble, but soon you’re cruising.
So, the next time you find yourself with an unbalanced situation, whether it's in the kitchen, your finances, or even your social life, remember the power of balanced equations. It's all about making sure that for every input, there's a corresponding, equal output. It’s the universe’s way of saying, "Keep it fair, keep it even, and everything will be just right." And who doesn't want a little more "just right" in their life?
