Balance The Equation On The Wall Display Labster

Ever felt like your brain is juggling flaming torches while trying to do a handstand on a unicycle? Yeah, me too! Sometimes, the sheer number of things buzzing around in our heads can feel like a chaotic circus. But imagine this: a secret superpower that helps you whip that circus into a perfectly synchronized, dazzling performance. That, my friends, is the magic of balancing! And guess what? Even when it comes to the teeny-tiny, super-important world of chemistry, balancing equations is like giving your brain a superhero cape!
Now, I know what you're thinking. "Chemistry? Equations? Sounds like homework I'd rather forget." But stick with me, because we're not talking about dusty textbooks and intimidating formulas. We're talking about the Labster Wall Display! Think of it as your super-cool, digital science playground where you can actually see and play with the building blocks of the universe. And one of the coolest games in this playground? Balancing The Equation!
Imagine you're baking cookies. You’ve got your flour, your sugar, your eggs, and your chocolate chips. You can’t just throw them all in the oven willy-nilly and expect deliciousness, right? Nope! You need the right amounts of each ingredient. Too much sugar, and they’re sickeningly sweet. Not enough flour, and they’re a gooey mess. It’s all about getting the proportions just right!
Balancing equations is like making sure every atom invited to the party shows up and has a good time, no more, no less!
That’s exactly what’s happening with chemical reactions. We've got these little guys called atoms, and they love to hang out and form new buddies, creating new substances. But here’s the golden rule, the absolute, can't-break-it decree of the universe: you can't create or destroy atoms. They just rearrange themselves, like a super-organized team playing musical chairs. So, if you start with, say, two shiny red atoms and three bouncy blue atoms on one side of your reaction (the reactants, if you want to sound fancy), you better darn well end up with two shiny red atoms and three bouncy blue atoms on the other side (the products)!
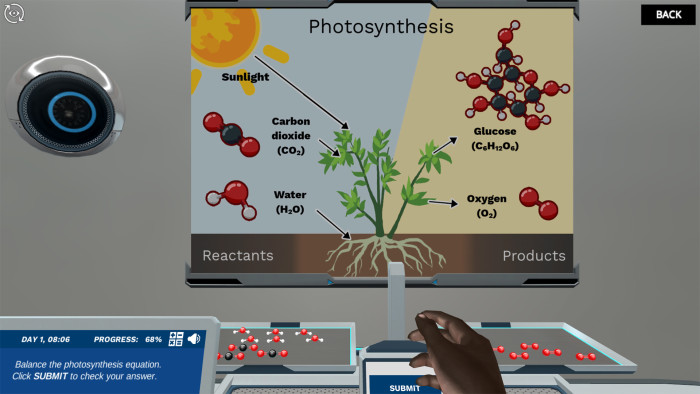
And that’s where the Labster Wall Display truly shines. Instead of squinting at numbers on a page, you get to see this whole process unfold visually. It’s like having a tiny, animated world in front of you where you can literally move atoms around. You see the starting ingredients, and then you adjust the amounts (these are the numbers you add in front of the chemical formulas, called coefficients – they’re like the portion sizes for our atom ingredients) until the number of each type of atom on both sides is perfectly equal. Poof! You’ve balanced the equation!
Let’s say you’re trying to make water. Sounds simple, right? You need hydrogen and oxygen. You might start with H₂ (two hydrogen atoms) and O₂ (two oxygen atoms). But if you just stick them together, you get… well, you don't get water! You need two molecules of water (H₂O) for every molecule of oxygen. So, you’d need to make sure you have enough hydrogen atoms to pair up with the oxygen atoms to form those two water molecules. On the Labster Wall Display, you'd see your hydrogen and oxygen atoms bumping into each other, and you'd adjust those coefficients until, presto! You’ve got perfect little water molecules, and every single atom is accounted for. It’s incredibly satisfying, like solving a puzzle where the pieces are atoms!

Think of it like building with LEGOs. You have a pile of red bricks and a pile of blue bricks. If your instructions say you need four red bricks and two blue bricks for your masterpiece, you better make sure you have exactly that many. If you’re short on red bricks, you can’t build the thing properly. If you have too many blue ones, they're just going to be cluttering up the place. Balancing equations is about making sure you have the perfect number of "atom bricks" to build whatever new molecule is supposed to be created. The Labster Wall Display makes this LEGO-building adventure interactive and fun!
And the best part? This skill isn't just for scientists in white coats. Understanding how things combine and in what proportions is fundamental to so many things. From cooking and baking to understanding how medicines work, and even how pollution affects our environment, the principles of balancing are everywhere. It's about understanding relationships, proportions, and how things interact in a balanced way. The Labster Wall Display gives you this awesome, hands-on experience that makes these abstract concepts feel totally real and, dare I say, exciting!
So, the next time you’re presented with a chemical equation, don’t groan. Smile! Think of it as a fun challenge, a puzzle waiting to be solved. And with the incredible, visual playground that is the Labster Wall Display, you've got the coolest tools at your fingertips to become a master of atomic organization. You'll be balancing equations like a pro, making sure every atom gets its moment in the spotlight, and feeling that fantastic rush of accomplishment. Go forth and balance, my friends!
