Balance Each Redox Reaction Occurring In Acidic Aqueous Solution

Hey there, chemistry curious cats! Ever feel like life’s a bit… unbalanced? Well, guess what? So are chemical reactions! And today, we’re diving into the wild world of redox reactions in acidic solutions. Sounds fancy, right? But honestly, it’s like a fun puzzle for your brain. And acidic solutions? Think of them as the energetic party starter of the chemical world.
So, what's a redox reaction? Think of it as a chemical exchange. One thing loses electrons, and another thing gains them. It's like a electron swap meet! The thing that loses electrons gets oxidized – it’s like it’s getting a glow-up. The thing that gains them gets reduced – like it’s getting a tiny energy boost. Pretty neat, huh?
Now, when we throw in an acidic solution, things get even more interesting. Acids are like the super-enthusiastic referees in this electron game. They love to get involved and help things along. They’re full of hydrogen ions (H+), and these little guys are key players.
Why is balancing these reactions important? Because nature likes things neat and tidy! We need to make sure that for every electron lost, one is gained. It’s all about conservation, people! And it’s also how we understand how things like batteries work, or how your body digests food. Pretty fundamental stuff, disguised as a fun challenge.
The Acidic Party Starter: Hydrogen Ions!
Imagine a chemistry lab as a dance floor. An acidic solution is like the DJ dropping a killer beat. Those H+ ions are everywhere, ready to mingle. In redox reactions, they love to jump in and grab onto oxygen atoms. Yes, oxygen! Those sneaky O’s often get involved in these electron exchanges, and H+ is like, "Ooh, let me help you with that!"
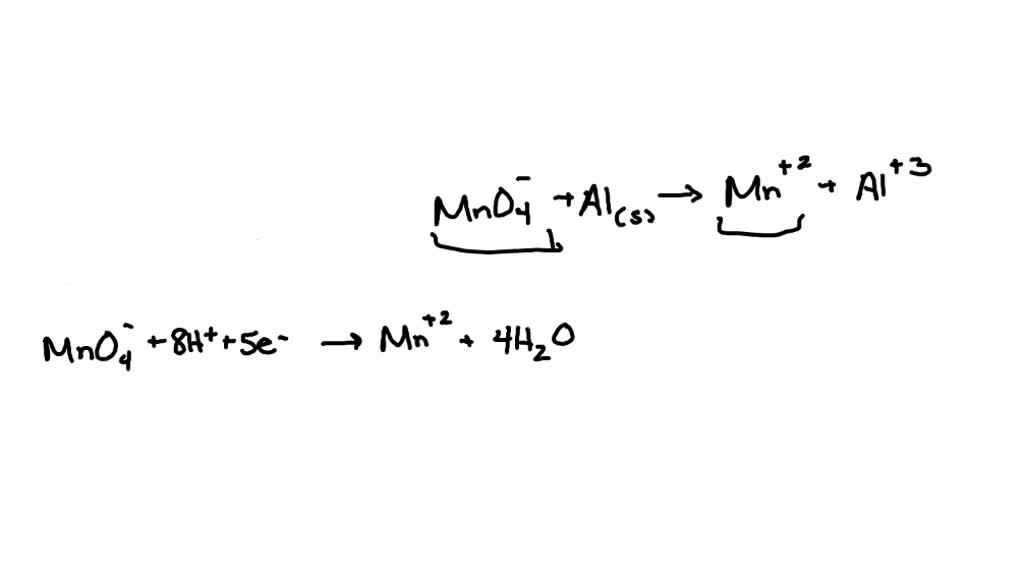
This is where the magic, and the balancing act, truly begins. You’ll often see water (H₂O) popping up as a product. It’s like the result of a happy little chemical marriage between oxygen and those eager hydrogen ions. And sometimes, you’ll see more H+ appearing on the reactant side, showing how crucial they are in kickstarting the whole process.
The Balancing Act: A Step-by-Step Shimmy
Alright, so how do we actually balance these things? It’s not as scary as it sounds. We break it down into two parts: the oxidation half-reaction and the reduction half-reaction. Think of it like splitting a dance routine into two distinct moves.
First, you identify which species is getting oxidized and which is getting reduced. This is where you might need to look at oxidation numbers. Don’t let that term intimidate you! It's just a way of tracking electrons. Like little electron scorecards.

Then, for each half-reaction, we balance it. This involves a few cool tricks:
- Balance atoms (other than O and H): This is the easiest part. Just make sure you have the same number of, say, carbon atoms on both sides. Simple enough, right?
- Balance oxygen atoms using H₂O: If you’re short on oxygen, add water molecules to that side. It’s like saying, "Need more oxygen? Here’s some water, it’s got plenty!"
- Balance hydrogen atoms using H+: This is where our acidic party starter comes in! If you’re short on hydrogen, add those precious H+ ions to balance it out. They’re the heroes of acidic redox balancing.
- Balance charge using electrons (e-): This is the crucial step. Make sure the total charge on both sides of the half-reaction is the same. You do this by adding electrons. If a side is too positive, add electrons (which are negative!) to balance it.
Once you’ve balanced both half-reactions separately, you have one last big dance move: combining them! You need to make sure the number of electrons lost in the oxidation half-reaction equals the number of electrons gained in the reduction half-reaction. If they don’t match, you multiply one or both half-reactions by a number to make them equal. It’s like choreographing a duet where both dancers have the same number of steps.
Finally, you add the two balanced half-reactions together. And poof! You cancel out anything that appears on both sides (like those electrons!). What you’re left with is your perfectly balanced redox equation. It’s a thing of beauty, really. Like a perfectly executed pirouette.

Quirky Facts and Funny Details
Did you know that some of the most common redox reactions happen when you see things rust or when your toast gets burnt? Yep! It’s all about electrons on the move. Rusting is essentially iron losing electrons (oxidizing) and oxygen gaining them (reducing).
And those bubbly reactions you sometimes see when you add an acid to something? Often, that’s a redox reaction at play, with gases like hydrogen being produced. It’s like a little chemical eruption!
The term "redox" itself is a portmanteau, a fancy word for a word made by blending two other words. "Red" from reduction and "ox" from oxidation. Clever, right? It’s like giving a chemical process a superhero nickname.
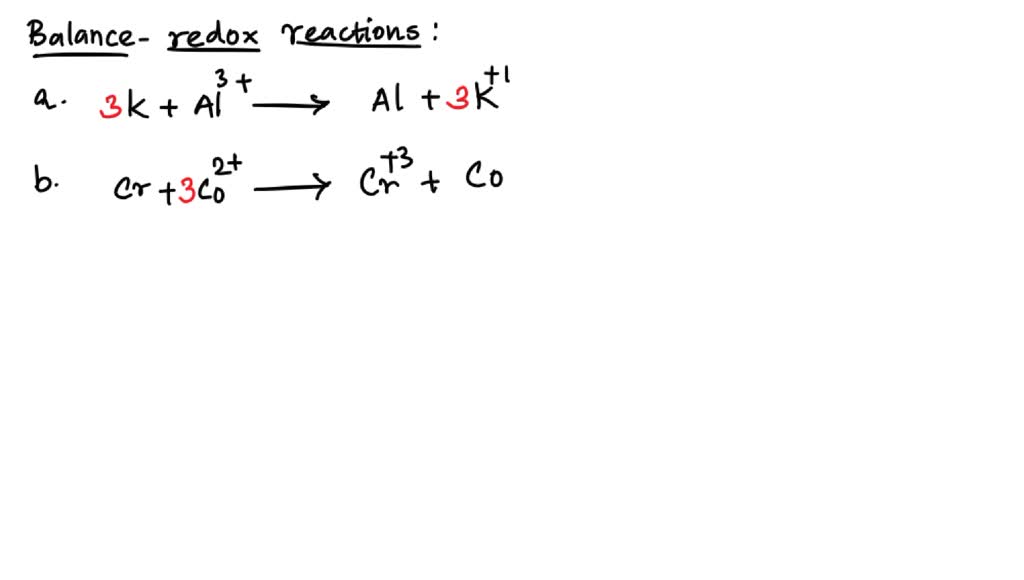
Why is balancing in acidic solution so common in textbooks? Because it’s a great starting point! Once you master this, moving to neutral or basic solutions is just a slightly more complex dance. It’s like learning the basic waltz before tackling the tango.
Honestly, the beauty of redox reactions in acidic solution is how they highlight the fundamental principles of chemistry. Electrons don't just disappear; they're transferred. And acids are active participants, not just passive bystanders. It’s a dynamic, energetic process that’s happening all around us, all the time.
So, next time you’re feeling a bit unbalanced, remember those electrons in their epic journey. And the ever-helpful hydrogen ions, always ready to join the party and make things happen. It’s a little bit of organized chaos, and a whole lot of fascinating chemistry. Go forth and balance!
