Autoionization Of Water Endothermic Or Exothermic

Hey there, you wonderful curious minds! Ever found yourself staring into a glass of water, completely mesmerized by its sheer… well, water-ness? It’s just so there, isn't it? We drink it, we swim in it, we use it to boil pasta (the ultimate test of its character, I think). But what if I told you that this seemingly placid liquid is actually a tiny, bustling metropolis, a microscopic dance party happening right before your very eyes?
Today, we’re diving into a topic that sounds a little… well, science-y. We're talking about the autoionization of water. Now, before you click away thinking, "Ugh, chemistry, no thank you!", stick with me! This isn't about memorizing complicated formulas (unless you want to, you brilliant nerd!). This is about uncovering a little secret that water holds, a secret that's actually pretty darn inspiring and, dare I say, a little bit fun!
So, what is this "autoionization" thing, anyway? Imagine water molecules, those H₂O buddies, having a bit of a chat. Sometimes, in their energetic little lives, one water molecule will kindly, or perhaps a little dramatically, lend a hydrogen atom (that’s the 'H' part) to another water molecule. Poof! Suddenly, you've got some ions zipping around. Specifically, you get a positively charged hydronium ion (H₃O⁺) and a negatively charged hydroxide ion (OH⁻).
Think of it like a game of molecular hot potato. One water molecule is feeling a bit too much hydrogen and passes it on. The recipient, now a bit overloaded with hydrogen, becomes a hydronium ion. The donor, having lost a hydrogen, is left with just oxygen and hydrogen – a hydroxide ion. It's a constant, fleeting exchange, like a silent, microscopic ballet. Pretty cool, right?
Now, the big question that might be tickling your brain cells: is this whole molecular hand-off an endothermic or exothermic process? Don't worry if those words sound like ancient runes. In super-duper simple terms:
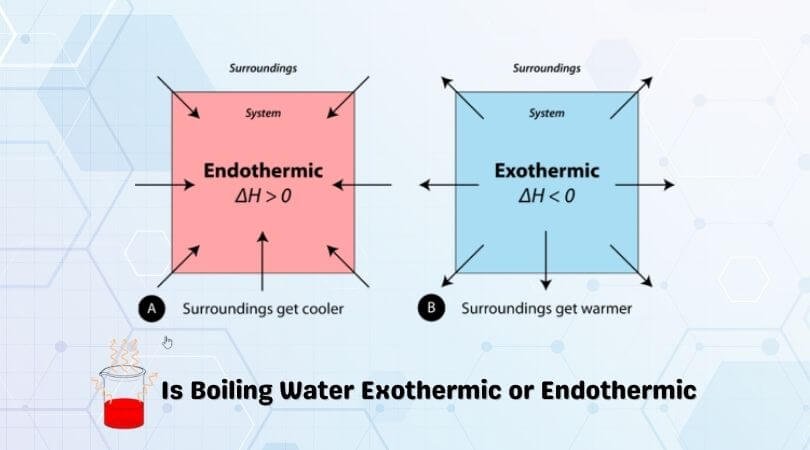
- Endothermic means it absorbs heat. Think of it like needing a little boost of energy to make something happen.
- Exothermic means it releases heat. Like a little burst of energy coming out.
So, which one is it for water’s self-ionization party? Drumroll, please… it's endothermic! Yep, that's right. This little molecular shuffle requires a bit of energy. Water molecules need a nudge, a little warmth, to get their hydrogen-swapping groove on.
Why is this important? Well, it tells us something fundamental about how water behaves. Because it's endothermic, the more heat you add to water, the more autoionization happens. More hydronium ions and hydroxide ions are born into the world! Isn't that a delightful thought? Adding a little warmth to your day literally sparks more chemical reactions in your water.

Imagine a cozy cup of tea on a chilly morning. As the heat seeps into the water, you're not just warming your hands; you're also encouraging a flurry of H₃O⁺ and OH⁻ ions to do their thing! It’s like your tea is throwing a tiny, invisible party, fueled by the heat you’re providing. Who knew a simple beverage could be so… chemically lively?
This also explains why the concentration of these ions is usually so incredibly low. At room temperature, for every 10 million water molecules, only about two have gone through this ionization process. That’s like finding two people out of a stadium full of 50,000 who are wearing a specific, brightly colored hat! It’s rare, but it happens!

The fact that water is so good at maintaining this delicate balance, even with these minuscule numbers of ions, is truly remarkable. It’s why pure water is considered to have a neutral pH. It’s this inherent ability to create both positive and negative charges, in equal measure, that makes water so universally adaptable and essential for life as we know it.
Think about it: life itself is a complex dance of chemical reactions, and water is the ultimate, ever-present dance floor. The autoionization of water, even in its tiny, shy way, sets the stage for so much of this. It’s the subtle hum beneath the surface, the quiet engine driving countless biological processes.

And here’s a little thought to ponder: the world is full of these hidden processes, these silent symphonies of chemistry and physics happening all around us, all the time. The autoionization of water is just one tiny, shining example. It reminds us that even the most familiar things can hold astonishing secrets.
So, the next time you’re sipping on some H₂O, take a moment. Appreciate the minuscule, energetic world within. Marvel at how this simple substance, requiring a little bit of heat to fuel its internal transformations, is so fundamental to everything. It's a beautiful reminder that learning about the world around us, even its seemingly complex corners, can be incredibly rewarding and, yes, even a little bit magical.
Don't let the jargon intimidate you. Dive in! Ask questions! The universe is an open book, and the chapters on water's autoionization are just waiting for you to explore. Who knows what other wonderful secrets you'll uncover? Keep that curiosity burning bright, and you'll find inspiration and fun in the most unexpected places. Go forth and be wonderfully curious!
