Atoms Containing Radioactive Nuclei Are Called ________.

Ever wondered about those tiny building blocks of everything around us? We’re talking about atoms, the universe's LEGO bricks. But what happens when one of these super small guys has a nucleus that’s a bit… well, energetic?
It turns out, there's a super cool name for atoms like that! They're not just any old atoms; they have a special kind of nucleus that’s a little bit unstable. This means they're doing something pretty fascinating all the time.
So, what do we call these atoms with a zippy nucleus? Drumroll, please… they are called radioactive! Yes, that’s right, atoms containing radioactive nuclei are called radioactive. It's like they have a built-in disco ball in their center, constantly shedding bits of energy.
Think of it this way: most atoms are like perfectly still dancers on a stage. They’re content and stable. But a radioactive atom? That one’s got the best moves, constantly jumping, spinning, and releasing little bursts of light and energy. It's a party happening at the atomic level!
And this "radioactive" quality isn't just a nerdy science term. It's the reason behind some seriously amazing things in the world. It's like having a secret superpower that these atoms possess.
One of the most mind-blowing aspects of radioactive atoms is their ability to spontaneously change. It's not like they decide to swap their hats; it's a fundamental transformation happening within their core. This process is called radioactive decay.
Imagine a tiny, ticking clock inside the atom's nucleus. This clock is counting down, and when it reaches zero, poof! The nucleus releases some energy and sometimes even a tiny particle. This is what makes them radioactive.
This isn't something scary or dangerous in the way you might think. For most of these atoms, it's just their natural way of being. They’re just… naturally energetic! It's like a firefly glowing – it's just what it does.
The energy they release is called radiation. This radiation can take different forms, like little packets of light (photons) or even tiny subatomic particles zipping out. It's like a miniature fireworks show happening inside every radioactive atom.
So, why is this so entertaining? Because it's a peek into the hidden world of the incredibly small, where things are constantly in motion and full of surprise. It’s like having a secret decoder ring for the universe!
The fact that atoms can be radioactive opens up a whole universe of possibilities. It’s the key to unlocking some of nature’s most powerful secrets. These aren't just simple spheres; they're dynamic little entities with a lot going on under the hood.
One of the coolest things about radioactive atoms is their age. We can use them to figure out how old really, really old things are. Think dinosaurs, ancient rocks, even the Earth itself!

Scientists use a process called radiometric dating. They look at how much of a certain radioactive atom is left in a sample and compare it to how much of its "decayed" form there is. It’s like forensic science for fossils!
Imagine a sample of a rock. It started with a certain amount of radioactive stuff, like a full tank of gas. As time goes by, that gas slowly gets used up, turning into something else. By measuring how much "gas" is left, scientists can tell exactly how long ago that rock was formed. Pretty neat, huh?
This technology has revolutionized our understanding of history. It’s allowed us to put timelines on ancient civilizations and understand the evolution of life on our planet. All thanks to these energetic little atoms!
But it's not just about digging up the past. Radioactive atoms are also incredibly useful in the present, especially in medicine. You might have heard of radioactive isotopes being used in treatments.
In some cases, these radioactive atoms are used to create detailed images of the inside of our bodies. Doctors can track how certain substances move through your system, helping them spot problems early on. It’s like having a tiny, internal GPS for health!
They can also be used to target and destroy harmful cells, like cancer cells. The radiation from the radioactive atoms can be directed with incredible precision to areas that need treatment. It’s a powerful tool in the fight for health.
And don’t forget about energy! The power of radioactive decay is harnessed in nuclear power plants. This is a way to generate a massive amount of electricity from a relatively small amount of fuel.
It’s a complex process, of course, but at its heart, it’s about carefully controlling the energy released by radioactive atoms. This energy can then be used to heat water, create steam, and spin turbines to generate electricity for our homes and cities.
So, when we say atoms containing radioactive nuclei are called radioactive, we’re not just giving them a label. We’re talking about the very stuff that makes up our world and the incredible forces that shape it. It’s a constant reminder that even the smallest things can have the biggest impact.
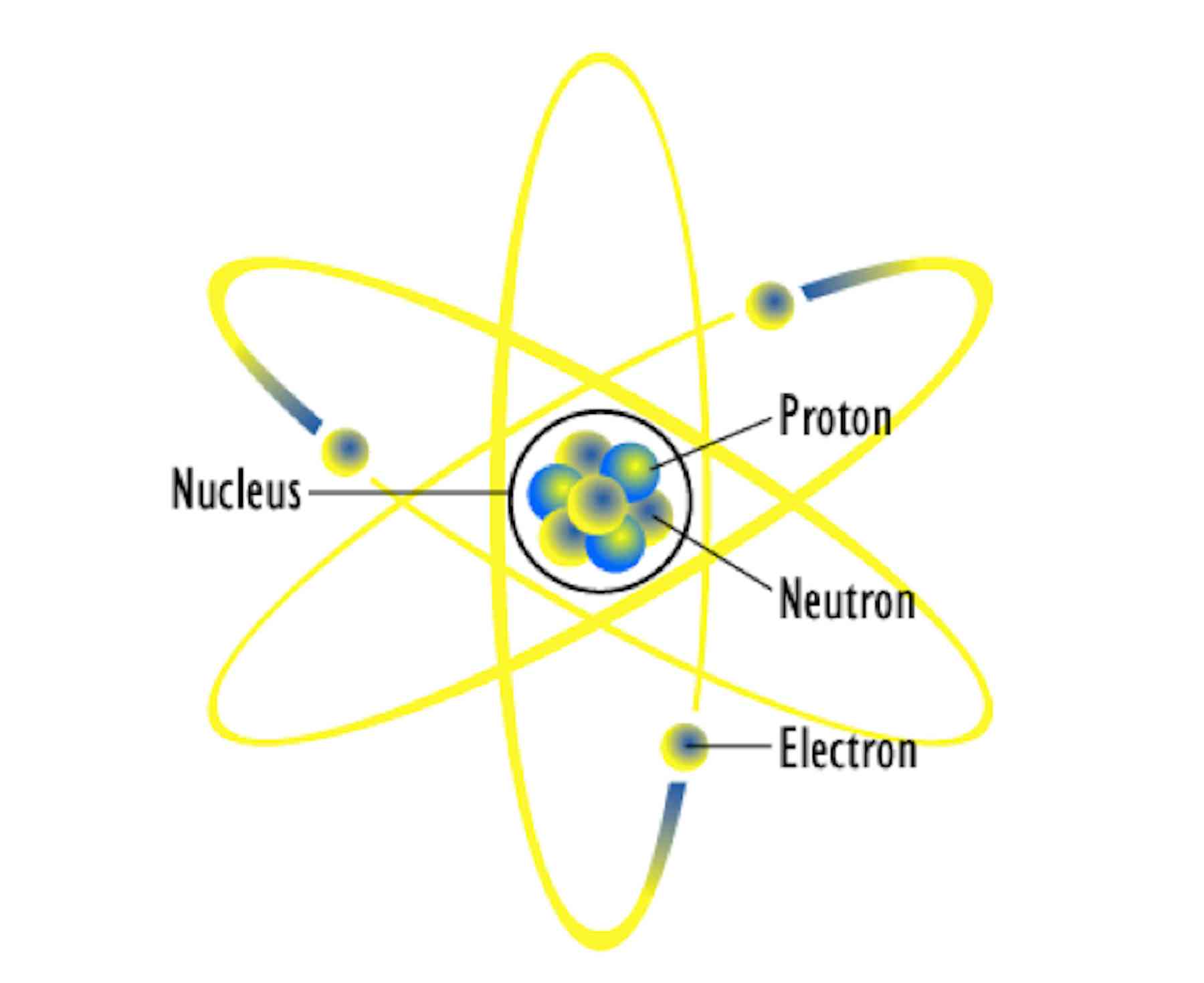
Think about it: the glow of a watch dial in the dark, the energy that powers some of our cities, the ability to see inside a patient’s body – all of these are thanks to the fascinating properties of radioactive atoms. They’re the unsung heroes of science and technology.
It’s like discovering a hidden level in a video game, where the ordinary transforms into the extraordinary. These atoms, with their energetic nuclei, are a testament to the wonders of the universe. They’re a constant source of discovery and innovation.
The word radioactive might sound a bit technical, but at its core, it’s about something incredibly dynamic. It’s about change, energy, and the unseen forces that are constantly at play. It’s a concept that’s both fundamental and awe-inspiring.
So, the next time you hear the word radioactive, don’t just think of science diagrams. Think of ancient mysteries unlocked, of life-saving medical treatments, and of the powerful forces that shape our planet. It’s a concept that’s truly out of this world.
It’s this inherent energy, this constant state of flux, that makes radioactive atoms so special. They are the universe’s tiny powerhouses, constantly giving off energy and transforming. It’s a never-ending story of change and discovery.
And the best part? This whole amazing world of radioactive atoms is out there, waiting to be explored. From the stars in the sky to the rocks beneath our feet, they’re everywhere, quietly doing their energetic dance.
So, remember the answer to that intriguing question: Atoms containing radioactive nuclei are called radioactive. And that, my friends, is just the beginning of a truly fascinating adventure!
Don't be afraid of the word "radioactive." It just means an atom has a nucleus that's a little bit buzzy and likes to share its energy!
It’s like a tiny, self-contained cosmic event happening all the time. The universe is full of these little wonders, and understanding them helps us understand everything around us.
The energy they emit, radiation, can be detected and measured. This has led to incredible advancements in fields ranging from archaeology to medicine. It’s a testament to how much we can learn by observing the natural world.
So, next time you’re looking up at the stars or holding a pebble, remember that you’re interacting with a universe filled with these energetic, radioactive building blocks. They’re a reminder that even the smallest things can hold immense power and unlock incredible secrets. It's a constant source of wonder.

The sheer variety of radioactive isotopes is mind-boggling. Each one has its own unique decay rate and energy signature, making them useful for different purposes. It’s like a toolbox full of specialized atomic tools.
So, the simple answer to our initial question unlocks a universe of complexity and wonder. Atoms containing radioactive nuclei are indeed called radioactive. And that’s just the start of a journey into the fascinating world of nuclear science.
It's this inherent instability, this tendency to transform, that makes them so special. They are not static; they are dynamic, constantly evolving at the most fundamental level. This is the essence of being radioactive.
The scientific exploration of radioactive elements has led to some of the most significant discoveries of the modern era. From understanding the age of the Earth to developing new medical treatments, their impact is undeniable. It's a story of scientific curiosity and its profound rewards.
So, go ahead, get curious! The world of radioactive atoms is a universe waiting to be explored. It’s a reminder that science is not just about formulas; it’s about the amazing, energetic, and sometimes surprising nature of reality.
They are a cornerstone of our understanding of physics and chemistry. Their properties allow us to probe the very fabric of matter. It’s a constant source of inspiration for new research and technologies.
In essence, the term radioactive is a gateway to a world of scientific marvels. It represents a fundamental characteristic of matter that has led to profound insights and transformative applications. It's a term that signifies power, transformation, and endless possibility.
So, the next time you think about atoms, remember the ones with the energetic nuclei. They are the radioactive ones, and they are a crucial part of our amazing universe. Their existence is a constant invitation to learn more.
It’s the energy they release that makes them so valuable and so intriguing. This energy is the key to unlocking their secrets and harnessing their power for the benefit of humanity. It’s a delicate balance of understanding and application.

So, what are atoms containing radioactive nuclei called? You’ve got it: radioactive! And that’s just the tip of the iceberg in a world of atomic wonders.
This constant decay and emission of energy is not a flaw, but a feature. It’s what makes these atoms unique and incredibly useful. It’s the universe showing its dynamic, energetic side.
The study of radioactive materials has opened up entirely new fields of science and technology. It’s a testament to human ingenuity and our relentless pursuit of knowledge. The journey of discovery continues.
It’s a name that hints at something powerful, something that can change and emit. And that’s exactly what these atoms do. They are active participants in the grand cosmic dance.
So, embrace the wonder! The world of radioactive atoms is a vibrant and essential part of our existence. It’s a reminder of the incredible forces that shape our reality, one energetic nucleus at a time.
They are the universe’s little firecrackers, constantly giving off light and energy. It’s a fascinating aspect of matter that continues to be explored and understood. The universe is full of surprises.
So, let’s celebrate these energetic atoms. They are the radioactive ones, and they are an integral part of our world and our understanding of it. Their story is far from over.
Ready to dive deeper?
The world of radioactive atoms is vast and full of amazing discoveries. You can find out more about radioactive decay, different types of radiation, and how they are used in medicine and industry. It’s a journey that will expand your understanding of the universe!
Consider looking up some fun facts about famous radioactive elements like Uranium or Radium. They have incredible stories behind them. You might even discover how your own environment contains these fascinating atomic building blocks.
So, the answer to atoms containing radioactive nuclei are called ________ is radioactive. Now you know a little bit more about these special atoms and why they’re so interesting. The universe is full of wonders, and these energetic nuclei are a big part of that!
