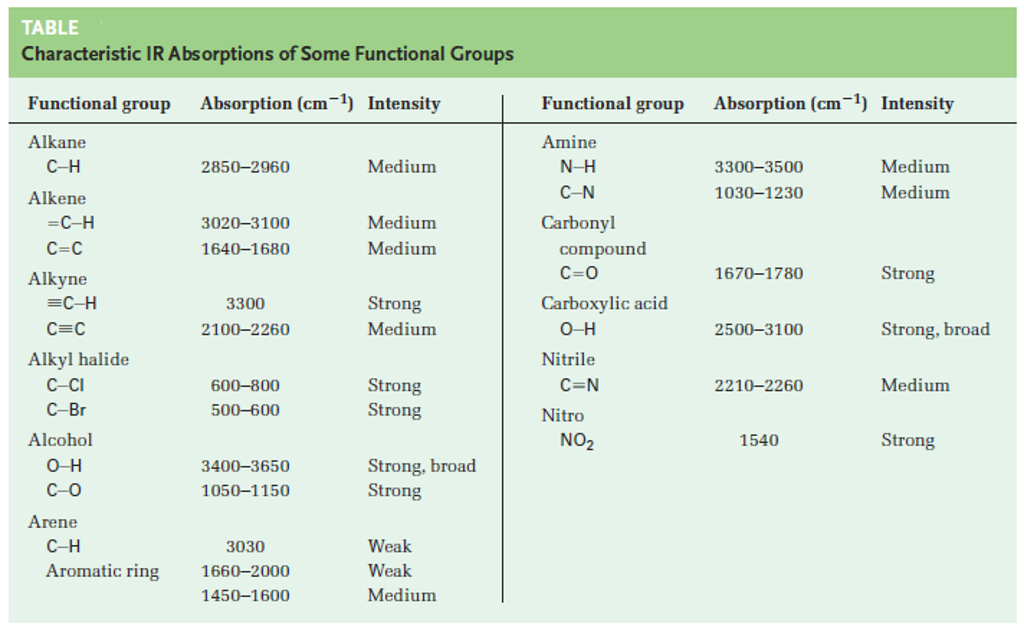
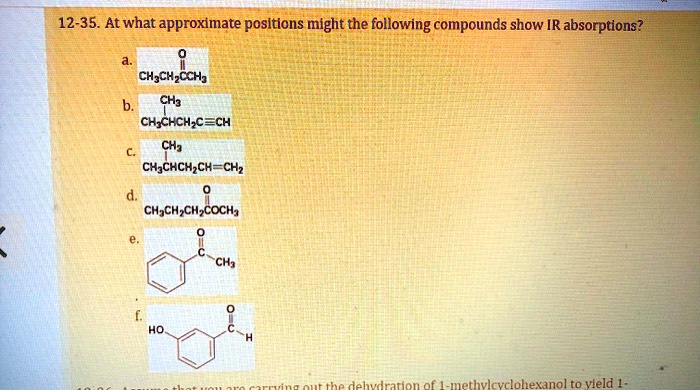
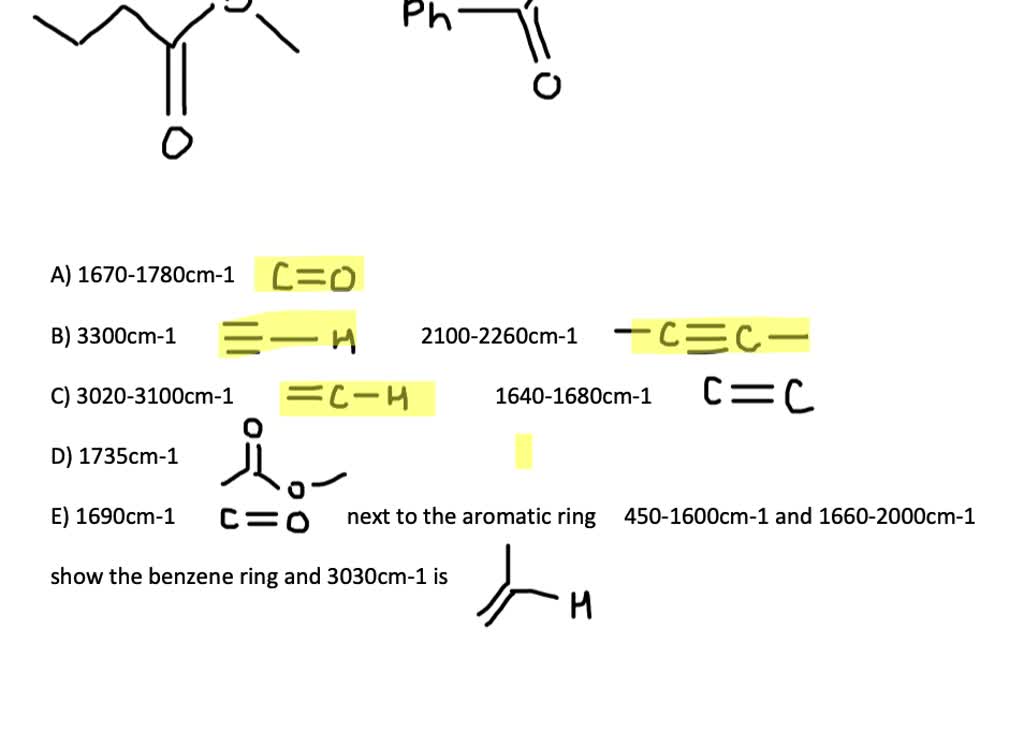
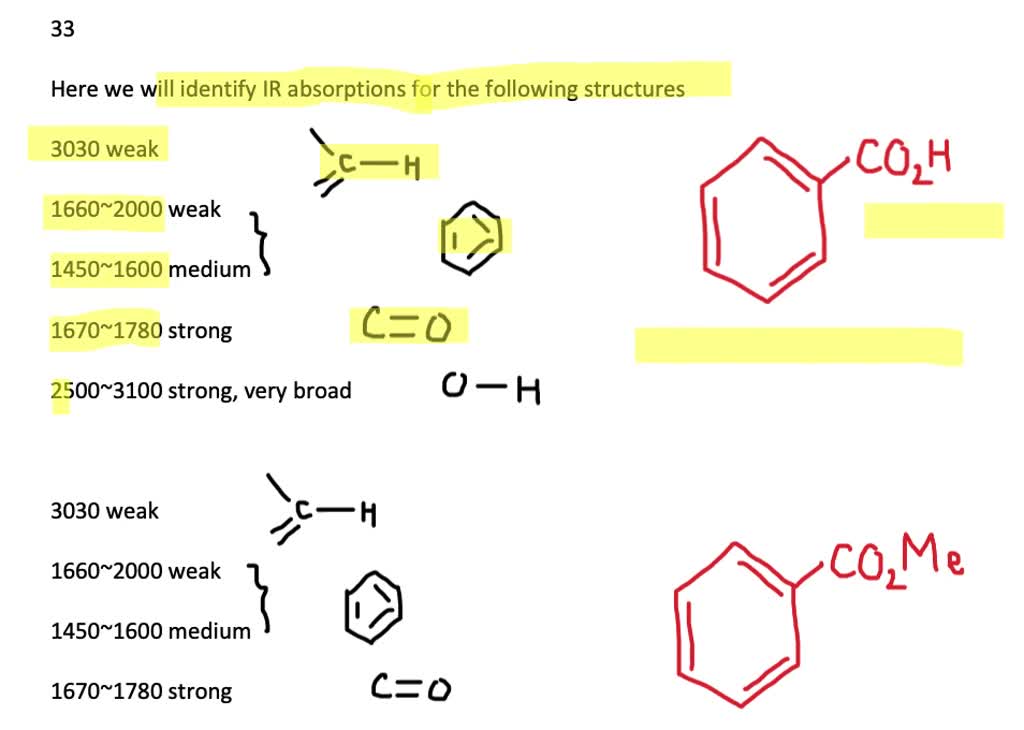
At What Approximate Positions Might This Compound Show Ir Absorptions
Ever stared at a bunch of squiggly lines on a graph and wondered, "What in the science-y world is going on here?" You're not alone! We're talking about Infrared (IR) spectroscopy, which sounds super fancy, but let's be honest, it’s basically a way for molecules to tell us their secrets through a special kind of light. Think of it like a cosmic karaoke session for atoms. They sing their little hearts out, and we listen in.
Now, the big question, the one that keeps aspiring chemists up at night (or maybe just them, who knows?), is: "Where will these singing molecules actually be found on our squiggly line graph?" We're talking about positions, or what the cool kids call wavenumbers. These are usually measured in cm-1, which sounds like a unit of measurement for tiny, invisible hats. But it's not. It's just how we measure how much the molecules are jiggling and wiggling when hit with infrared light.
So, let's play a little game of "Guess Where the Funk Will Be Found." It's a bit like trying to find your keys. You know they're somewhere, but pinpointing the exact spot can be a challenge. And sometimes, the spots are just plain weird. Unpopular opinion incoming: sometimes the most interesting IR absorptions show up in places that make absolutely no sense at first glance. Like that one time I was convinced my cat had learned to play the piano. Turns out, it was just a rogue radiator. Similar energy, right?
Let's start with the absolute superstars of the IR world. These are the groups that are so common, they practically have their own fan clubs. If you see a molecule with a carbonyl group (C=O), get ready for some action! These guys are usually found chilling out in the 1650 to 1800 cm-1 region. It’s like their favorite hangout spot. Think of it as the prime real estate for double bonds doing their carbonyl thing. It’s a strong signal, a loud singer. They don’t do subtle.
Then there are the hydroxyl groups (O-H). These are the gossipy ones. They can be found in a couple of places, depending on their mood and who they're hanging out with. If they're feeling all independent and stretching their legs, you might find them doing their thing around 3200 to 3600 cm-1. That's a pretty wide range, like trying to find a specific shade of beige. But if they decide to get cozy with their neighbors, maybe forming a hydrogen bond, they can get a bit broader and shift down a bit. They become a bit more of a mumbled, "Oh, hey there..." instead of a clear shout.
What about the humble C-H bond? Everyone has those, right? Well, almost everyone. These are the background singers. They’re everywhere, and they tend to cluster in the 2800 to 3100 cm-1 region. It’s a bit like a crowded party – lots of chatter, hard to pick out one voice. But if it’s a double bond (C=C), they’re a bit more excited, usually showing up a little higher, around 1600 to 1680 cm-1. They're not as loud as a carbonyl, but they're definitely there, giving the song some rhythm.
And let’s not forget the amines (N-H). These are the ones with a bit of a unique sound. If you have a primary amine (two N-H bonds), you might see two little peaks around 3300 to 3500 cm-1. It's like they're having a duet! If it's a secondary amine (one N-H bond), it's more of a solo performance, usually a single peak in that same ballpark. They’re a bit more shy than the hydroxyls, but still noticeable.
Now, here’s where things get fun. Sometimes, you see a peak that’s just… off. Maybe it's a bit too strong, or in a place you wouldn't expect. That’s the molecule playing a prank. It’s like when you’re absolutely sure you put your phone on the counter, but it’s actually in the fridge. These unexpected peaks could be due to unusual bonding, strain in the molecule, or even just the molecule being extra dramatic. It’s the jazz improvisation of the molecular world!
For instance, a triple bond (C≡C or C≡N) tends to be a bit more reserved, often showing up around 2100 to 2260 cm-1. They're not as enthusiastic as the double bonds or carbonyls, more of a quiet hum. But they're important! And sometimes, you might see a weird shoulder on a peak, or a broad hum where you expected a sharp note. That's the molecule whispering secrets, and it’s our job to decipher them.
The truth is, while there are general "hot spots" for different functional groups, the exact position can wiggle around. It’s like trying to give directions to a very particular cat. "He's usually by the window, but sometimes he’s under the bed, and on Tuesdays, he’s definitely in the laundry basket." It’s a bit of an art and a bit of a science. And honestly, the unpredictable nature of it all is part of the charm. It keeps things interesting, and prevents us from getting too complacent. So next time you see those IR lines, don’t just see squiggles. See the singing molecules, telling their stories, one wavenumber at a time. And maybe, just maybe, share my unpopular opinion that the best absorptions are the slightly mischievous ones.
