At Constant Pressure And Temperature Which Statement Is True

You know, I was staring at a perfectly innocent cup of tea the other day, steam lazily curling upwards, and it got me thinking. It was one of those moments, right? You’re just… existing. And then your brain decides to go on a little tangent. This particular tangent involved the tea itself. If I were to, say, leave this cup on the counter, would the amount of tea in it change? Obviously, some of it will evaporate, making the liquid level go down. But what if I prevented evaporation? What if I somehow sealed it off from the world? That’s when the real nerdy thoughts kicked in.
Suddenly, my quiet kitchen felt like a tiny, self-contained laboratory. I started wondering about the stuff inside that sealed cup of tea. Is it just… sitting there, being tea? Or is there some subtle, invisible dance happening at the molecular level? And more importantly, if I kept everything constant – the temperature, the pressure – what could I definitively say about this little universe I’d created?
This is where science, my friends, gets really, really interesting. It’s not just about memorizing formulas in school (though, let’s be honest, that’s part of it). It’s about understanding the fundamental rules that govern everything, from our steaming cups of tea to the vastness of space. And today, we're going to dive into one of those fundamental rules. Specifically, we're going to talk about what happens when you decide to keep things nice and stable: constant pressure and temperature. Because, believe it or not, this seemingly simple condition unlocks some pretty profound truths about gases.
Think about it. When you’re cooking, you often adjust the heat, right? You crank it up, you turn it down. And sometimes, you’re dealing with a pot with a lid on it, which creates a certain amount of pressure. But what if you just… decided to fix those things? No more fiddling with the knob, no more worrying about the lid. Just a steady state. What does that tell us about the gas molecules zipping around inside that sealed pot?
This is the realm of what scientists call thermodynamics, and it's basically the study of heat and its relationship to energy and work. It sounds super academic, I know. But at its core, it’s about understanding how energy flows and transforms. And under the specific conditions of constant pressure and temperature, some really neat relationships emerge. Especially when we’re talking about gases. They’re the most enthusiastic movers and shakers in the molecular world, always bumping into each other and the walls of their container.
So, let's get down to brass tacks. If we have a certain amount of gas, and we’re chilling out in a situation where the pressure isn't changing and the temperature isn't changing, what can we confidently say is also staying the same? Or, perhaps more accurately, what fundamental relationship is revealed?
The Grand Unveiling: What's True at Constant P and T?
Alright, drumroll please! When you’re dealing with a gas under conditions of constant pressure and constant temperature, the statement that rings truest, the one that’s like the bedrock of gas behavior in this scenario, is this: the amount of gas is directly proportional to the volume it occupies.
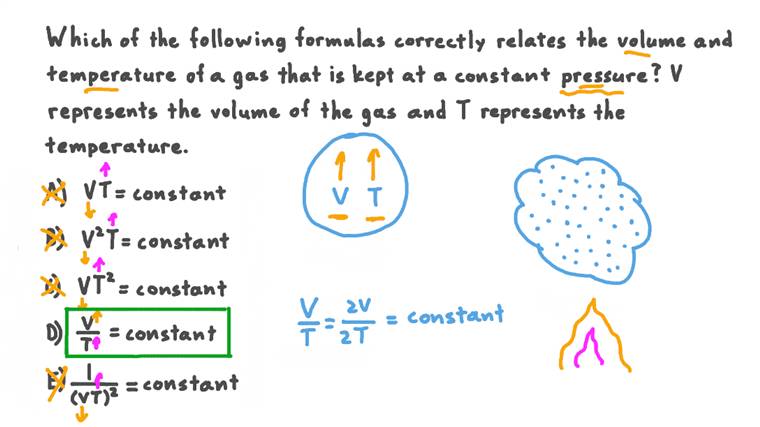
Woah, okay. Let's unpack that a bit. What does "directly proportional" even mean in plain English? It means that if you double the amount of gas, the volume it takes up will also double. If you halve the amount of gas, the volume will halve. They move in lockstep, like a perfectly choreographed dance duo. Pretty neat, huh?
This might sound almost too obvious when you first hear it. Like, "Duh, of course more gas takes up more space!" But the scientific beauty here is in the rigor of it, and the fact that it holds true across a wide range of conditions, as long as our pressure and temperature are behaving themselves.
This relationship is actually a part of a larger family of gas laws. You might have heard of Boyle's Law (which deals with constant temperature and changing pressure and volume) or Charles's Law (constant pressure, changing temperature and volume). Well, this one we're talking about is often attributed to a fellow named Amedeo Avogadro, and it's called Avogadro's Law. And it’s a pretty big deal.
Avogadro's Law: The Unsung Hero
So, why is Avogadro's Law so important, especially when we're holding pressure and temperature steady? Because it tells us something fundamental about the quantity of gas. It's not just about the jiggling molecules themselves, but about how many of them there are.
Imagine you have a balloon. If you keep blowing air into it at the same temperature and with the same external air pressure (more or less), the balloon will get bigger. The more air (which is made of gas molecules, obviously) you add, the more volume it takes up. Simple as that. And the reverse is true too. If you let some air out of the balloon, it shrinks.

This is the essence of Avogadro's Law in action. Under constant pressure and temperature, equal volumes of all gases, at the same temperature and pressure, contain the same number of molecules. This was a revolutionary idea! Before this, people thought different gases, even in the same volume, might have different numbers of "particles." Avogadro basically said, "Nope, nope, nope. If the conditions are the same, the number of little guys is the same."
Now, the statement we’re focusing on for this article is a slightly different, but closely related, consequence of Avogadro's Law when we're talking about a fixed amount of gas changing its volume. It's about how the number of moles (which is a way of counting molecules) relates to the volume when P and T are fixed. And that's where we get our main statement: the amount of gas (number of moles) is directly proportional to the volume.
Let's dig a little deeper into why this is true. Think about the gas molecules. They're constantly moving, colliding with each other and the walls of their container. The pressure we feel is essentially the force of these collisions against the container walls. The temperature is a measure of the average kinetic energy of these molecules – how fast they're moving and jiggling.
Now, if we decide to keep the temperature the same, it means the average speed of our gas molecules isn’t changing. They’re still zipping around with the same gusto. If we also keep the pressure the same, it means the overall effect of their collisions on the container walls is constant. They're hitting the walls with the same intensity and frequency, on average.
So, what happens if we introduce more gas molecules into this system? Well, with more molecules zipping around at the same speed, they're going to bump into the walls more often. To maintain that constant pressure, the container has to expand. It needs more space to accommodate the increased number of collisions without the overall force on the walls increasing. It’s like a crowded room. If you keep the energy level (temperature) the same but add more people, they’re going to bump into each other and the walls more. To keep the "pressure" of the crowd at a certain level, the room needs to get bigger.
Conversely, if we remove some gas molecules, there will be fewer collisions with the walls. To maintain the same pressure, the volume must decrease. The gas molecules will have less space to roam, and the collisions will become more concentrated, but the overall force per unit area (pressure) will remain constant because there are simply fewer of them bouncing around.
It’s a beautiful equilibrium. The gas is essentially saying, "Okay, you want me to maintain this pressure and temperature? Then I need this much space to do my molecular thing. More of me? I need more space. Less of me? I’ll take up less space."
Why This Matters (Beyond Tea and Balloons)
This isn’t just some abstract scientific curiosity. This relationship has real-world implications everywhere. Think about:
- Industrial Processes: In chemical plants, where gases are often handled under controlled conditions of temperature and pressure, understanding this proportionality is crucial for calculating reaction yields, designing storage tanks, and ensuring safety.
- Weather Forecasting: While the atmosphere is a complex beast, the principles of gas behavior, including this relationship, underpin many meteorological models. Changes in the amount of gases in the atmosphere and how they occupy space affect air pressure and weather patterns.
- Breathing: Our own lungs are a biological example. When we inhale, we increase the volume of our chest cavity, drawing air (a mixture of gases) in. When we exhale, we decrease the volume, expelling gases. While temperature and pressure aren't perfectly constant in our bodies, the fundamental principle of volume changing to accommodate different amounts of gas is at play.
- Scuba Diving: Divers need to understand how gases behave under changing pressure. While the temperature might not fluctuate wildly, the pressure changes dramatically with depth. This law, along with others, helps explain why your tank pressure drops as you use air and how gas volumes change in your lungs at different depths.
It’s this idea of conservation of conditions that makes these gas laws so powerful. When you lock down certain variables, others reveal themselves in predictable ways. It’s like putting on blinders and suddenly being able to see the intricate patterns in the path ahead.

Let’s consider another angle. What if we had a fixed volume and constant temperature, and we changed the pressure? According to Boyle's Law, the pressure would be inversely proportional to the volume. But that's not our scenario here. What if we had constant volume and pressure, and changed the temperature? That's a different story too. The beauty of our current discussion is isolating that specific relationship: more gas = more volume, less gas = less volume, when P and T are held captive.
It's also worth noting that this law is an ideal gas law. Real gases, especially at high pressures and low temperatures, can deviate from this ideal behavior. They have actual molecular volumes and intermolecular forces that start to matter. But for most practical purposes, and especially at conditions far from condensation, the ideal gas model and Avogadro's Law give us an incredibly accurate picture.
The Takeaway: Simple but Profound
So, to bring it back to our initial musings about the cup of tea, if we could somehow keep the temperature and pressure of the air above our sealed tea perfectly constant, and more tea molecules decided to jump into the gaseous phase (evaporate), the volume of that gaseous space above the tea would have to increase to accommodate them, if the container allowed for expansion. If the container was rigid and sealed, the pressure would increase, which isn't the scenario we're discussing.
The core truth we're left with is that under conditions of constant pressure and temperature, the amount of a gas (how many moles are present) is directly proportional to the volume it occupies. It’s a fundamental building block in our understanding of matter and energy, a testament to the order that exists in the universe, even at the microscopic level. It’s a reminder that even in the seemingly chaotic dance of gas molecules, there are elegant, unbreakable rules governing their behavior.
So next time you’re enjoying a hot beverage, or even just breathing in the air around you, take a moment to appreciate these invisible laws at play. They're the silent architects of our world, and understanding them, even a little bit, is a pretty cool thing. It’s not just about science class; it’s about understanding the very fabric of reality. And honestly, that’s something worth pondering, even over a simple cup of tea.
