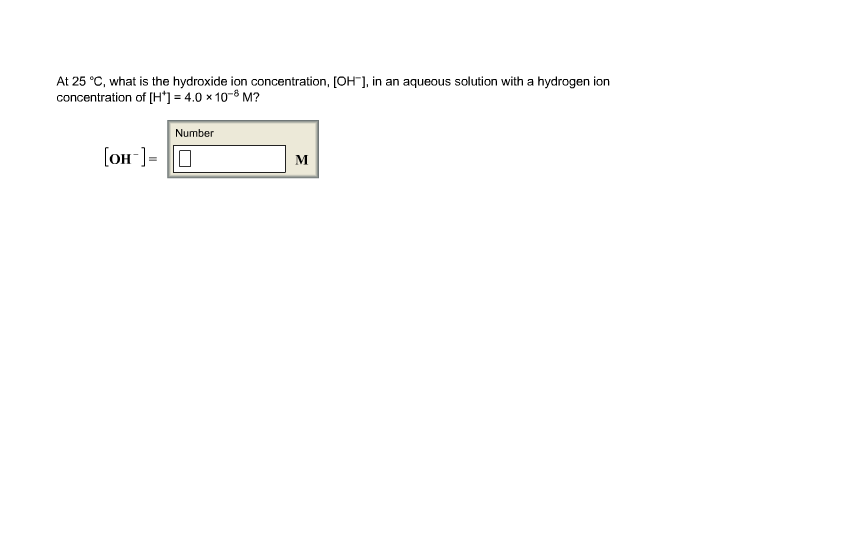
At 25 C What Is The Hydroxide Ion Concentration
Ever wondered what makes water, well, watery? Or why that fizzy drink tickles your nose? It’s all about tiny, invisible players dancing around. Today, we're going to peek behind the curtain and talk about one of these fascinating characters: the hydroxide ion! Specifically, we’re looking at its concentration at a perfectly pleasant 25°C. Sounds a bit science-y, right? But trust me, it's more fun than you might think!
Think of it like this: water is made of tiny little bits. Some of these bits are water molecules (H₂O, if you're feeling fancy). But sometimes, just by chance, these molecules bump into each other and break apart. When they do, they can form two new things: a hydrogen ion (H⁺) and a hydroxide ion (OH⁻). It's like a tiny, spontaneous dance party happening all the time in your water!
Now, the hydroxide ion is the star of our show today. It's an electrically charged little fellow, and it plays a huge role in whether something feels slippery or sour. When you have lots of these hydroxide ions hanging out, things tend to feel a bit more… well, basic. Think of that slippery feel of soap. That’s the magic of hydroxide ions at work!
So, what’s its concentration at 25°C? This is where it gets a little bit like a secret code. In pure water, at this very standard temperature (which is pretty much room temperature for most of us), the concentration of hydroxide ions is a very specific number: 1 x 10⁻⁷ moles per liter. Woah, that looks complicated! But let's break it down.
The "1 x 10⁻⁷" part is a way scientists write really, really small numbers. It means you have a 1 followed by seven zeros after the decimal point, then a 1. So, it’s 0.0000001. That’s a tiny amount! And “moles per liter” is just the scientific way of saying how many of these little guys are packed into a certain amount of liquid. So, in a liter of pure water at 25°C, there are only 0.0000001 moles of hydroxide ions. Pretty minuscule, right?
But here's the cool part: even though it's a tiny number, it's incredibly important. This concentration is the benchmark for neutral. It means there's an equal number of hydroxide ions and their buddies, the hydrogen ions. It’s a perfect balance, a state of chemical zen!
Imagine it like a perfectly balanced seesaw. On one side, you have the hydrogen ions, and on the other, the hydroxide ions. At 25°C in pure water, this seesaw is perfectly level. It's not tipping to the acidic side (too many hydrogen ions) or the basic side (too many hydroxide ions). It’s just right!
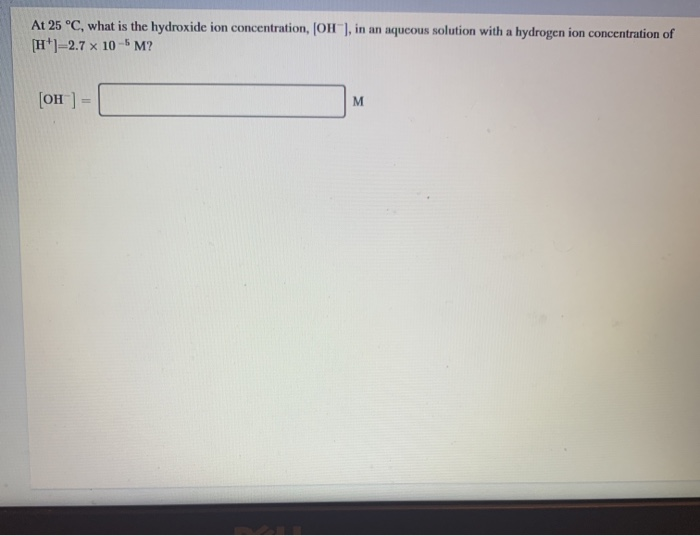
Solved At 25 °C, what is the hydroxide ion concentration, | Chegg.com
Why is this so entertaining? Because it’s the foundation of so much we experience! This little number, 1 x 10⁻⁷ mol/L, dictates the pH of everything around us. pH is just a simpler way to talk about how acidic or basic something is, and it’s directly linked to these ion concentrations. A pH of 7, which is neutral, happens when the hydroxide ion concentration is exactly what we’re talking about.
Think about it: your shampoo is carefully balanced to be a little bit basic, so it cleans your hair without drying it out too much. Your coffee is acidic, making it taste a bit bitter. Even the water you drink has this delicate balance. This concentration is the invisible hand guiding all these experiences!

What makes it special is its universality. At 25°C, this is the magic number for pure water, no matter where you are. Whether you’re in a bustling city or on a remote island, the fundamental chemistry of water at this temperature behaves the same way. It’s a constant in a world of change.
And the best part? You don’t need a fancy lab coat to appreciate it. You can start thinking about the world in terms of these tiny, invisible particles. When you feel that tingle from a lemon or the smoothness of lotion, you’re interacting with the results of these ion concentrations. It’s like having a superpower to understand why things feel and taste the way they do!
So next time you’re sipping on some water, or using soap, take a moment to think about those hardworking hydroxide ions. Their concentration at 25°C is a small number, but it’s a gateway to understanding the chemistry that shapes our everyday lives. It’s a little peek into the amazing, invisible world that makes everything work. Isn’t that just delightfully intriguing?

