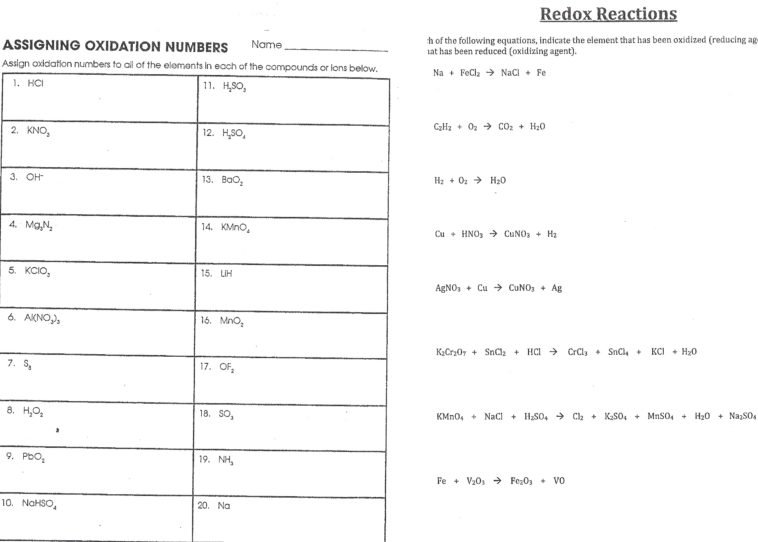
Assigning Oxidation Numbers Worksheet Answer Key
So, you’ve been staring at a pile of those dreaded “Assigning Oxidation Numbers Worksheet” pages. Maybe your kiddo brought them home, or perhaps you’re revisiting some dusty old chemistry memories yourself. Whatever the reason, that innocent-looking worksheet can feel like a cryptic puzzle, can’t it?
But what if I told you that behind those numbers and chemical formulas lies a hidden world of playful characters and unexpected alliances? It’s like a tiny, invisible drama unfolding on your page, and once you know the secret handshake, you can’t help but smile.
Think of the worksheet as a guest list for a very exclusive, very tiny party. Everyone has a role, and their oxidation number is their party outfit – it tells you a bit about who they are and what they’re up to. It’s not always obvious, and sometimes they’re trying to be something they’re not, but that’s where the fun begins.
The Unsung Heroes of the Worksheet
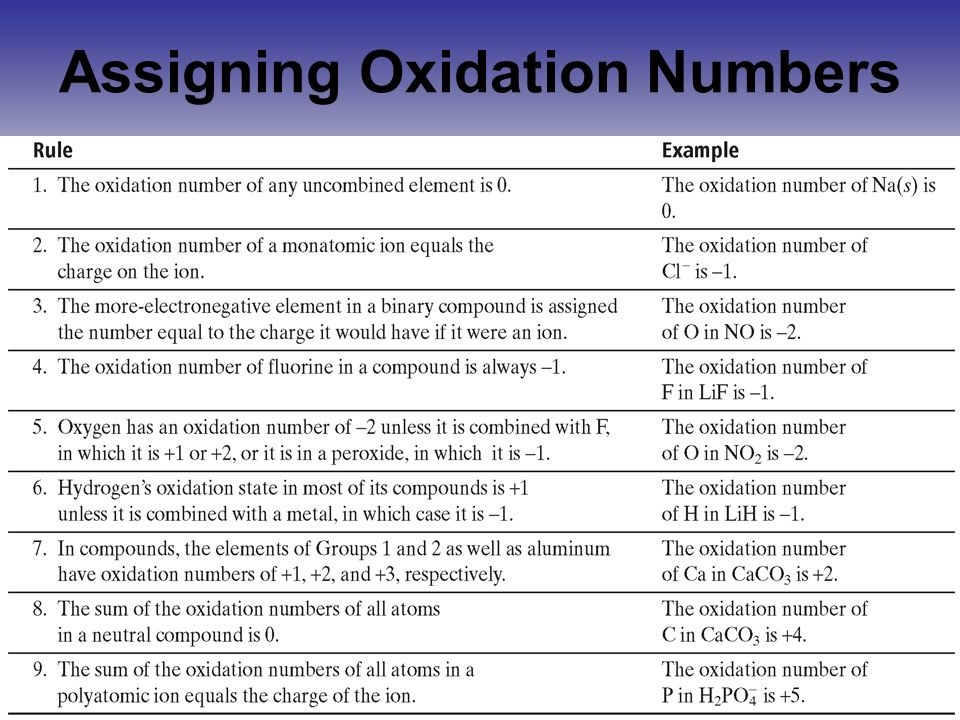
Let’s start with the undisputed stars, the ones who always, no matter what, show up in their signature attire. We’re talking about elements like oxygen and hydrogen. They’re the reliable friends, the ones you can always count on.
Oxygen, bless its little heart, usually struts around with a -2. It’s like its favorite sweater, the one it always wears. Unless, of course, it’s feeling a bit feisty, which is a whole other story we won’t get into right now.
And then there’s hydrogen! Usually, it’s rocking a cheerful +1. It’s the friendly neighbor, always willing to lend a hand. It’s the life of the party, generally speaking.
When Things Get Complicated (and Funny)
But then, you have the elements that are a bit more… eccentric. These are the ones that keep us on our toes. They’re the rebels, the rule-benders, the ones who make you pause and think, “Wait a minute…”

Take fluorine, for example. This little guy is SO electronegative, it practically bullies everyone else. It almost always wears a -1. It’s the bossy one, but in a way that’s almost endearing.
And what about those mysterious ions? They’re like the guests who show up with a plus or minus sign attached to their name, making them instantly more interesting. A sodium ion (Na⁺) is like a sodium atom that’s decided to give away a bit of its energy – it’s feeling generous and has a +1.
The chloride ion (Cl⁻) is the opposite. It’s eagerly accepted an extra bit of energy, sporting a -1. It’s like that friend who always borrows your pen and never gives it back.
The Mystery of the Monatomic Guest List
Now, here’s a little secret that might tickle your funny bone. When you have a single atom all by itself, with no friends or family around, it’s considered a monatomic ion. And in these cases, its oxidation number is simply its charge. It’s like showing up to the party with your name tag clearly stating your mood: happy (+2), grumpy (-3), or just chill (0).

So, a lone sodium atom (Na) chilling by itself? It’s got an oxidation number of 0. It’s perfectly content in its own company.
But a sulfate ion (SO₄²⁻)? Now that’s a whole different story! It’s a group of atoms that have decided to stick together, and they have an overall charge. Figuring out the individual oxidation numbers within that group is where the detective work really kicks in.
The ‘Sum of Their Parts’ Party Rule
Here’s a rule that’s so simple, it’s almost heartwarming. In any neutral compound (one that doesn’t have an overall charge), the oxidation numbers of all the atoms have to add up to exactly zero. It’s like everyone at the party agreeing to keep the total chaos at bay.
If there’s a charged compound, a polyatomic ion, then the sum of the oxidation numbers equals the charge of that ion. It’s like a slightly rowdier party, where the overall vibe is a bit more… energetic.
This rule is your superpower when you’re tackling those worksheets. You’ve got some knowns, and the rest can be figured out by making sure the numbers balance. It’s a bit like a seesaw – if one side goes up, the other has to adjust.
The ‘Worksheet Answer Key’ – Your Friendly Guide
And that’s where the “Assigning Oxidation Numbers Worksheet Answer Key” comes in. Think of it not as cheating, but as having a wise, patient mentor. It’s there to guide you, to show you the way when you’re feeling lost in the chemical wilderness.
Sometimes, when you’re struggling with a problem, the answer key is like a friendly whisper, saying, "Remember that rule about oxygen? Or that fluorine always wins?" It’s the gentle nudge you need to see the pattern.
It’s amazing how a simple answer key can transform frustration into a “aha!” moment. That feeling of understanding, of finally cracking the code, is incredibly rewarding. It’s like finding a hidden treasure on your worksheet.

Beyond the Numbers: A Microscopic Waltz
So, the next time you see an assigning oxidation numbers worksheet, don’t just see numbers. See the personalities, the little dramas, the constant dance of electrons. See the teamwork and the occasional sibling rivalry among the elements.
It’s a reminder that even in the smallest corners of science, there’s a vibrant, dynamic world at play. And with a little bit of imagination and the helpful guidance of an answer key, you can find the fun and the wonder in it all.
It's a microscopic waltz, a silent ballet, and you've just learned a few of the steps! So go forth, and assign those oxidation numbers with a smile.
The true beauty of science is that it's not just about memorizing facts, but about understanding the stories behind them.
