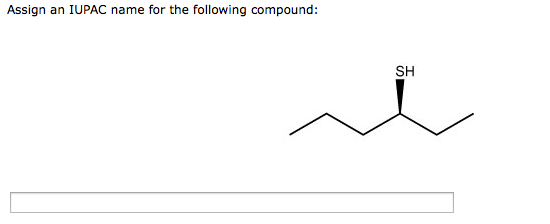
Assign An Iupac Name For The Following Compound:
Imagine you're at a cosmic naming ceremony for a brand new molecule. It’s not just any molecule; it’s one that might end up in your favorite ice cream, your super-strength shampoo, or even the paint on your walls. The chemists, like celestial cartographers of the tiny, have discovered it and now it’s time to give it a proper, official name. But this isn't like naming your pet dog "Fido" or your pet cat "Whiskers." Oh no, this is a name with a whole system behind it!
Think of the International Union of Pure and Applied Chemistry (IUPAC) as the universe's ultimate naming committee. They’ve got rules, like a super-strict kindergarten teacher, but these rules are designed to make sure everyone, everywhere, understands exactly which molecule you’re talking about. It’s a global agreement, so a chemist in Tokyo and a chemist in Toronto can both look at the same name and picture the exact same tiny structure. Pretty neat, right?
So, our little molecule is waiting patiently, vibrating with potential, for its official moniker. It's a bit like waiting for your baby’s name to be announced, but instead of a cute nickname, it gets something… well, a bit more scientific. But don't let that fool you! There's a hidden charm in these long, sometimes tongue-twisting names. They tell a story, a microscopic biography of the compound.
Let's peek at our specific guest of honor. We’re not going to get too deep into the nitty-gritty of electron orbitals or bond angles, that would be like trying to explain quantum physics to a goldfish. Instead, we’re going to focus on the essence of its name. Think of it as looking at a fancy cake and not just seeing frosting, but imagining the delicious layers and flavors beneath.
The Root of the Matter
Every IUPAC name starts with a foundation, a core. For organic molecules, which are the building blocks of life and so many everyday things, this foundation is usually a chain of carbon atoms. The length of this chain tells us the basic “parent” of the molecule. It’s like saying “this is a seven-bedroom house” before you start describing the paint colors and the furniture.
So, we'll find out how many carbons are in a row. Is it a tiny, cozy studio apartment of a carbon chain? Or a sprawling mansion with hundreds of carbons stretching out? This first part of the name is the parent hydrocarbon, the simple skeleton upon which everything else is built.
It's like getting to know the family name. Is it the "Smiths" or the "Joneses"? This tells you the basic lineage. For our molecule, this family name will be something like "methane," "ethane," "propane," or going up the scale, "hexane," "heptane," or even much, much longer ones! Each one signifies a different, fundamental carbon backbone.
Branching Out!
Now, what if our carbon chain isn't perfectly straight? What if it has little “arms” or branches sticking out? This is where the name gets a little more interesting, and frankly, a lot more like a real-life family tree. These branches are like cousins hanging out with the main family, or maybe adventurous kids who’ve built a treehouse off the main trunk.
The IUPAC system has a way of accounting for these branches. They get their own little names, usually ending in "-yl," which is like giving those branches their own mini-family names, like "methyl" or "ethyl." These are the cheerful, often smaller groups that attach themselves to the main carbon chain.
We have to figure out where these branches are attached. This is where numbers come in. Think of it like giving an address to each branch. So, you might have a "2-methyl" group, meaning a methyl branch is hanging off the second carbon atom of the main chain. It’s like saying, “the red door is on the second floor.”
Adding Some Flavor
But wait, there’s more! Molecules often have special “functional groups” attached to them. These are like the spices that give a dish its unique flavor, or the decorations that make a house feel like a home. These groups are the real workhorses, determining how the molecule behaves and what it can do.
Are we talking about an alcohol, which might have an "-OH" group (and might make things bubbly or act as a solvent)? Or perhaps a carboxylic acid, with its distinctive "-COOH" group (which can make things sour)? Maybe it's an ether, an aldehyde, a ketone, or an amine? Each of these functional groups has its own specific suffix, which gets tacked onto the end of the name.
This suffix is like the final flourish on a painting or the signature on a contract. It tells you the molecule’s main personality trait. For example, if it’s an alcohol, the name will end in "-ol," like ethanol (the kind in your hand sanitizer, and yes, in some drinks too!). If it’s a carboxylic acid, it might end in "-oic acid."
Putting It All Together
So, we have our parent chain, our branches with their positions, and our functional groups. Now, the IUPAC system has a set of rules to arrange all this information into a single, coherent name. It’s like a perfectly choreographed dance where every dancer knows their part and their cue.
We'll list the branches alphabetically (imagine them lining up for a photo shoot!), followed by the parent chain and the functional group suffix. It might seem like a lot, but once you get the hang of it, it's like solving a delightful puzzle. Each piece fits perfectly into place.
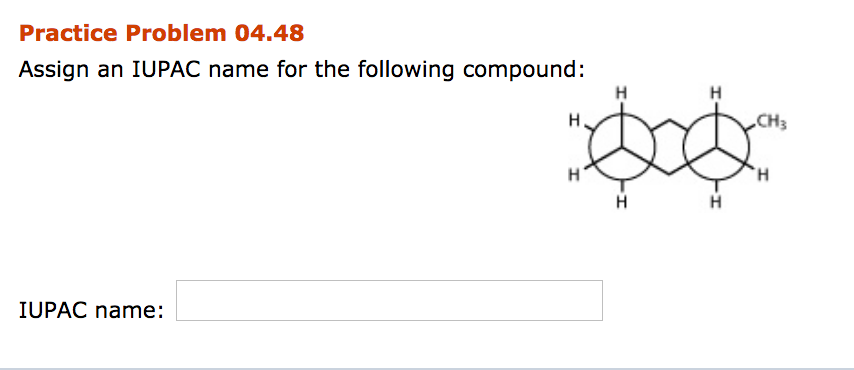
Let’s imagine our compound. Maybe it has a nice, long chain of seven carbons. That’s our heptane backbone. Then, it has a little methyl branch hanging off the third carbon. So, we have "3-methyl." And to top it all off, it has a functional group that makes it an alcohol, so it gets the "-ol" ending.
So, the name might be 3-Methylheptan-1-ol! (We’ll keep the actual name a surprise for now, but this gives you the flavor!). It’s a name that might sound a bit intimidating at first, but it’s actually a detailed blueprint. It tells us exactly how many carbons are there, where the branches are, and what the important functional group is.
The next time you encounter a long, complex chemical name, try to break it down. See if you can find the parent chain, the branches, and the functional groups. It’s like uncovering a secret code, a hidden story of molecular structure. And who knows, you might find yourself appreciating the elegance and precision of these names, even if they are a bit of a mouthful! It’s a whole new way to see the chemistry all around us, turning potentially dry science into a fun linguistic adventure.
