Assign A Systematic Name For The Following Compound:
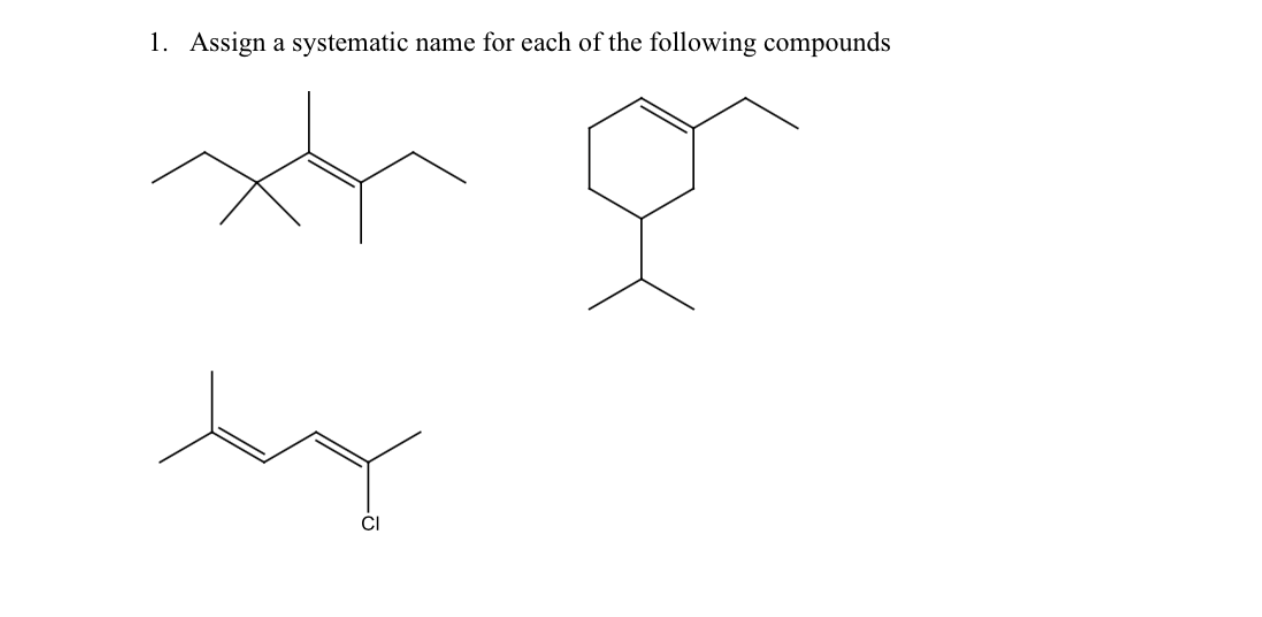
So, we’ve got a mystery on our hands, folks. A chemical conundrum. A molecular masterpiece, if you will. We’re looking at a certain compound, and it’s our job, our noble quest, to give it a name. A proper, official, totally-not-made-up-on-the-spot name. This is where the fun – and perhaps a little bit of head-scratching – begins.
Now, I know what you’re thinking. “Naming chemicals? That sounds… thrilling.” And to that, I say, you’re absolutely right! It’s a thrill ride of prefixes, suffixes, and seemingly random numbers. It’s like deciphering an ancient code, but instead of treasure, you get… a chemical compound. Exciting stuff, am I right?
The thing is, these names aren't just for show. They're like tiny little instruction manuals. They tell you exactly what’s going on in that little chemical world. It’s all very organized, very systematic. Or at least, that’s the idea. Sometimes it feels more like a playful game of Jenga, where one wrong move and the whole thing collapses into a pile of unintelligible syllables.
Let’s just say, for the sake of argument, that our mystery compound looks a little something like this. Imagine, if you will, a chain. A carbon chain, to be precise. Think of it like a string of tiny, hungry little carbon atoms, all linked up and holding hands. This is the backbone of our operation.
Now, these carbon atoms aren't content with just holding hands. Oh no. They've got to have some friends. Some other atoms that like to hang out with them. Like hydrogens, for instance. Those little guys are everywhere, aren't they? Like tiny, energetic toddlers at a party, always sticking to everything.
But it doesn't stop there! Our compound has even more interesting guests. We’ve got some elements that are a bit more… flamboyant. A bit more attention-grabbing. These are the ones that give our compound its unique personality. They’re the quirky aunts and uncles of the molecular family.
So, we start with our longest chain of carbons. This is the foundation. This is the main event. We count them, carefully, precisely. Because in the world of chemical naming, every single carbon counts. It’s like counting your blessings, but with more potential for explosion.
Once we’ve got our chain length sorted, we need to figure out where everything else is attached. Think of it like decorating a cake. You’ve got the cake itself (the carbon chain), and then you’ve got the frosting, the sprinkles, the little edible figurines. Those are our other atoms and groups.
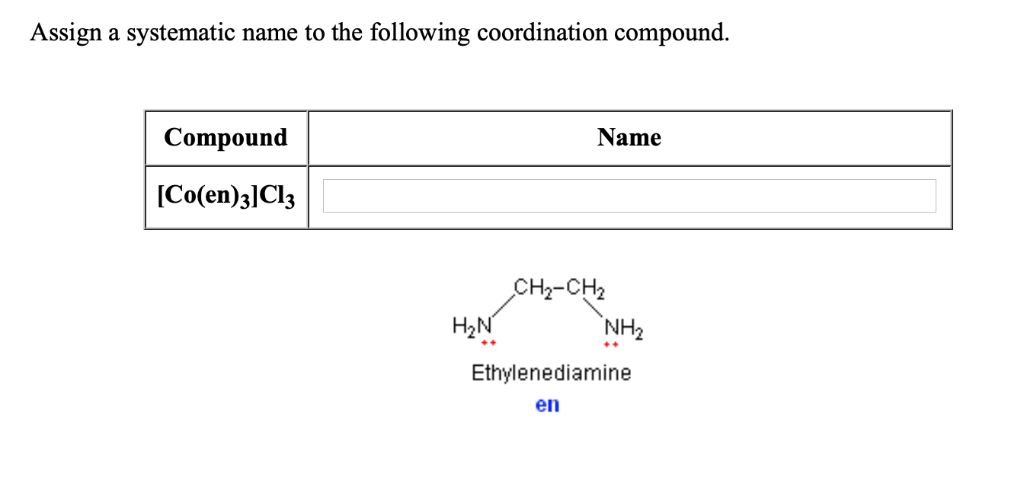
We assign numbers to the carbon chain. It’s like giving seats at a very important dinner party. We want to make sure everyone knows where they belong. And importantly, where our special guests are sitting. This is crucial for the official name.
Now, let’s say we have a functional group. This is like the star of the show. The diva. It’s a specific arrangement of atoms that gives our compound its special powers. Like, is it an alcohol? That means it’s got an -OH group hanging around. Very important. Very, shall we say, hydrated.
Or perhaps it’s a carboxylic acid. That’s like having a little chef’s hat on the end of the chain. It’s got that distinctive -COOH business going on. Makes things a bit tangy, you know?
We also have to consider any branches. Imagine our carbon chain is a main road. The branches are like side streets. Little detours. We need to name these branches too, and tell everyone exactly which exit they’re coming off.
This is where the prefixes come in. You’ve got things like ‘methyl’ for a one-carbon branch, ‘ethyl’ for a two-carbon branch, and so on. It’s like giving nicknames to the little offshoots. “Hey, there’s a little methyl guy chilling on carbon number three!”
And the positions of these branches? We use those numbers we assigned earlier. So, it might be ‘2-methyl’. This means a methyl group is attached to the second carbon in our main chain. It’s all very precise, like a chemical GPS.

What about double bonds or triple bonds? Those are like superhighways in our carbon chain. They make things a bit more energetic. We need to tell people where these are too. ‘Ene’ for double bonds, ‘yne’ for triple bonds. Sounds like a funky dance move, doesn't it?
Then there are the things that are a bit more… unusual. Elements that aren’t carbon or hydrogen. Things like chlorine, bromine, or oxygen atoms hanging off in places they might not be expected. We have to name those too, with prefixes like ‘chloro-’ or ‘bromo-’ or ‘oxo-’.
It’s like a scavenger hunt for names. You find a group, you name it. You find a bond, you name it. You find a branch, you name it. And then you string it all together, in the correct order, of course. Because order is everything.
And what if there are multiple of the same thing? We don’t just say ‘methyl methyl methyl’. Oh no. We get fancy. We use ‘di-’ for two, ‘tri-’ for three, ‘tetra-’ for four. It’s like having a chemical counting system that’s more efficient than trying to shout all the numbers yourself.
So, let’s look at our hypothetical compound. We’ve identified the longest carbon chain. Let’s say it’s five carbons long. That’s a pentane. But wait! It’s not just a plain old pentane. It’s got some friends tagging along.
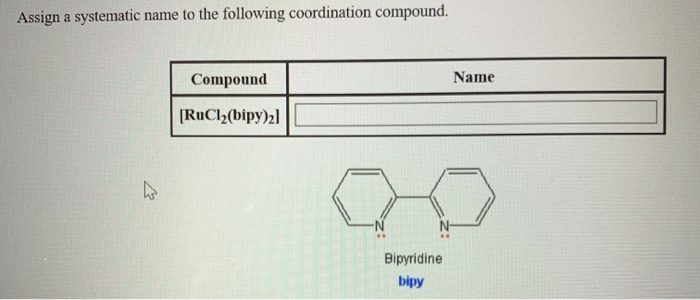
We’ve found a methyl group on the second carbon. So, that’s a ‘2-methyl’. And on the third carbon, we’ve got another methyl group. So that’s ‘3-methyl’. And, oh look, there’s a double bond starting on the second carbon. That’s our ‘2-ene’.
Now, we have to put it all together. We arrange the substituents alphabetically. So, the ‘methyl’ groups come before the ‘ene’. And the numbers tell us where everything is.
So, for our little compound, the name might start to look something like this: 2,3-dimethylpent-2-ene. See? It’s like a chemical puzzle solved. It tells you there are two methyl groups, one at position 2 and one at position 3, on a five-carbon chain, with a double bond starting at position 2.
It’s a bit of a mouthful, I admit. It sounds like a secret handshake for chemists. But beneath the seemingly complex jargon is a beautiful logic. It’s a language that speaks volumes about the structure of these tiny, invisible worlds.
And my unpopular opinion? Sometimes, just sometimes, these names are so wonderfully absurd, they deserve a bit of a cheer. They’re the unsung heroes of scientific communication. The slightly awkward, but ultimately brilliant, individuals who make sure we’re all talking about the same thing. So, next time you see a chemical name, give it a nod. It’s earned it.
Because, at the end of the day, assigning a systematic name is more than just a bureaucratic exercise. It’s an act of creation. It’s giving form and identity to the unseen. It’s like naming a pet, but with more precise instructions on how not to accidentally set your house on fire. And that, my friends, is a kind of magic.

So, there you have it. The thrilling, the terrifying, the utterly systematic world of chemical nomenclature. It’s a journey, and we’ve just scratched the surface. But hopefully, you’ve learned to appreciate the intricate dance of atoms and the eloquent, if slightly verbose, language used to describe them. And who knows, maybe one day you'll be confidently rattling off names like 2,3-dimethylpent-2-ene at your next dinner party. Or maybe not. That’s okay too. Just smile and nod.
The process of assigning a systematic name is like a detective story. You’re looking for clues. The longest chain is your main suspect. The branches and functional groups are the accomplices or key witnesses. And the numbers? They’re the timestamps and locations. You gather all the evidence, and then you build your case – your name.
It’s a skill that takes practice, and sometimes even the most seasoned chemists will pause and count on their fingers. But the satisfaction of correctly identifying and naming a compound is like solving a really good crossword puzzle. It’s a moment of clarity, of order imposed upon complexity. And in the vast, often chaotic, universe of molecules, that’s a pretty wonderful thing.
So, while it might not be as flashy as a science fiction movie, the naming of chemical compounds is a critical and surprisingly entertaining part of chemistry. It’s the foundation upon which all further understanding is built. Without these systematic names, we’d be lost in a sea of unlabeled substances, each with its own unique set of properties and potential surprises.
Let’s just hope our compound isn’t one of those ridiculously long, tongue-twisting ones that takes ten minutes to say. Those are the ones that make you question your life choices. But even those have a certain charm, don’t they? A testament to the endless creativity of nature, and the ingenuity of humans in trying to make sense of it all.
Ultimately, the goal is clarity. To be able to communicate, precisely and unambiguously, about the molecular world. It’s a shared language, a scientific Esperanto, if you will. And our little compound, with its carefully constructed name, is now a recognized member of this global chemical community.
