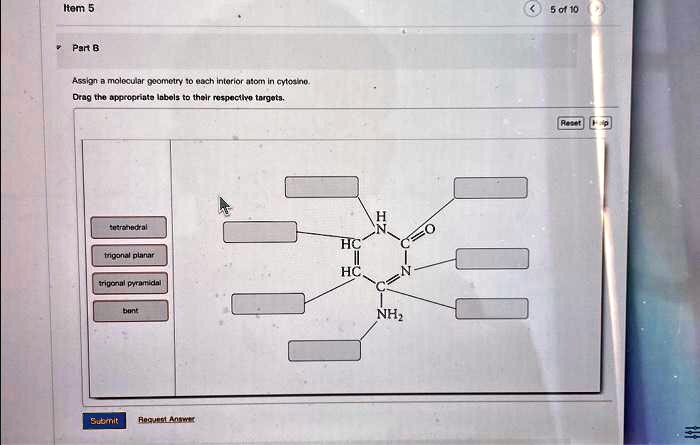
Assign A Molecular Geometry To Each Interior Atom In Cytosine
Hey there, science enthusiasts and curious minds! Ever wondered what gives our DNA its snazzy structure? Well, today we're going to zoom in on one of its coolest building blocks: cytosine! Yep, that's right, we're talking about a molecule that's literally part of you. And the best part? Understanding its shape can be surprisingly fun and a little bit like playing with molecular LEGOs. So, grab a cuppa, settle in, and let's get our geometry goggles on!
Now, I know what you might be thinking. "Molecular geometry? Isn't that super complicated and, dare I say, a little bit... dry?" Well, hold onto your hats, because we're about to discover that even the tiniest building blocks of life have a fantastic flair for design. And when it comes to cytosine, it's all about how its atoms arrange themselves in space. Think of it like a tiny, intricate dance that each atom performs, creating a unique three-dimensional shape.
Let's Meet Cytosine, Our Star Molecule!
So, what exactly is cytosine? It's one of the four nucleobases that make up DNA and RNA. It pairs up with guanine (another one of its buddies, you might recall from biology class). Cytosine itself is a pretty neat molecule, consisting of a ring structure with a few extra bits hanging off. It's like a little chemical party, and we're here to check out the seating arrangements!
We're going to focus on the interior atoms of cytosine. This means we're looking at the atoms within the main structure, not the ones that are just attached by a single bond. Imagine you have a small town, and we're interested in the buildings that are inside the town limits, not the houses on the outskirts. Makes sense, right?
The Atom-by-Atom Breakdown: A Geometric Adventure!
Cytosine has several interior atoms, and each one gets its own special geometric designation based on how many other atoms it's bonded to and how many lone pairs of electrons it has. These lone pairs are like little invisible arms that also influence the shape. Don't worry, it's not as intimidating as it sounds!
Nitrogen Atom 1 (N1): The Center of Attention!
Let's start with one of the nitrogen atoms, specifically the one we'll call N1. This atom is a crucial part of the ring. It's bonded to two other carbon atoms within the ring. Now, here's the key: it also has a lone pair of electrons. This lone pair is like a little bit of extra "stuff" that takes up space. When an atom has three "things" bonded to it (two carbons and one lone pair), it tends to arrange itself in a nice, flat, triangular pattern. This geometric shape is called trigonal planar. Imagine three friends holding hands in a circle – it’s a lovely, organized arrangement!
So, N1 is sitting pretty in a trigonal planar arrangement. This means it's part of a flat section of the molecule. Pretty straightforward, right? It’s like the sturdy foundation of our little molecular house.
Carbon Atom 2 (C2): The Straight Shooter
Next up, we have a carbon atom, let's call it C2. This carbon is a bit of a go-getter. It's involved in a double bond with an adjacent nitrogen atom (more on that nitrogen in a sec!) and a single bond with another carbon atom within the ring. When an atom forms a double bond, it's like it's holding onto two things really tightly. Because of this double bond and its other single bond, C2 doesn't have any lone pairs. It’s all about those connections! Atoms that are involved in a double bond and one other single bond, with no lone pairs, also love to arrange themselves in a flat, triangular fashion. So, C2 also boasts a trigonal planar geometry. It’s right there, cozy with N1, contributing to that flat, organized feel.
See? We're already getting the hang of this! Two atoms in a row, both with a neat, flat layout. It's like a perfectly laid out mosaic.
Nitrogen Atom 3 (N3): The Double Agent
Now, let's talk about the nitrogen atom that C2 is double-bonded to. Let's call this one N3. This nitrogen is a bit of a double agent because it's involved in that strong double bond with C2. It's also bonded to a carbon atom within the ring. Similar to C2, N3 has no lone pairs of electrons. The double bond with C2 and the single bond with the other carbon atom dictate its spatial arrangement. And guess what? Just like its neighbors, N3 is also trigonal planar. It's all about those flat, orderly arrangements in this part of the molecule. It's like they're all holding hands in a very efficient, space-saving way.
This is where it gets exciting! When you have multiple trigonal planar centers next to each other, the molecule tends to be very flat. This flatness is actually super important for how DNA fits together!
Carbon Atoms 4, 5, and 6 (C4, C5, C6): The Pillars of the Ring
Now we have the remaining carbon atoms in the ring: C4, C5, and C6. These are pretty standard carbon atoms. Each of them is bonded to two other atoms within the ring. Importantly, they don't have any lone pairs of electrons. Think of them as the sturdy connectors in our molecular structure. Because they are bonded to two other atoms and have no lone pairs, these carbon atoms also adopt a trigonal planar geometry. It’s like they’re all saying, “Let’s keep things flat and organized here!”
So, it turns out that a good chunk of the interior atoms in cytosine are all trigonal planar! This gives the core of the cytosine molecule a remarkably flat structure. Isn't that neat? It’s like they’ve all agreed to a neat, tabular layout, making the whole thing very compact and efficient.
Why Does This Even Matter? It Makes Life More Fun!
You might still be asking, "Okay, so it's flat. Big deal?" Well, this geometric precision is everything in the world of molecules! Because cytosine is so flat, it can stack up nicely with other bases in DNA, forming those iconic double helix steps. Imagine trying to build a spiral staircase if your steps were all wobbly and lumpy. It just wouldn't work, right?

Understanding these shapes helps scientists design new drugs, understand how diseases work, and even engineer new materials. It’s like having a secret code to the universe’s construction manual. And for us, the curious observers, it adds a whole new layer of appreciation for the intricate beauty of nature. It turns a potentially dry textbook page into a vibrant, three-dimensional world!
Learning about molecular geometry is like learning a new language – the language of molecules. And once you start speaking it, you’ll see the world in a whole new, fascinating way. Every molecule, from the simplest to the most complex, has its own unique story told through its shape.
Your Turn to Explore!
So, the next time you hear about DNA, or any molecule for that matter, remember the humble cytosine and its wonderfully organized, trigonal planar interior. It’s a small reminder that even in the tiniest corners of existence, there’s a symphony of shapes and structures at play. You have the power to understand and appreciate this! Dive a little deeper, explore other molecules, and let your curiosity lead you on an incredible journey. The universe is full of molecular wonders, just waiting to be discovered by you!
