Arrangement Of Electrons In Atoms Chapter 4 Review
Ever wondered what makes things tick? Like, really, really tick? We're not talking about your alarm clock (though that's important too!). We're diving deep into the teeny-tiny world inside atoms, and trust me, it's way cooler than you might think. Forget dusty textbooks; this is like a cosmic detective story, all happening right under our noses.
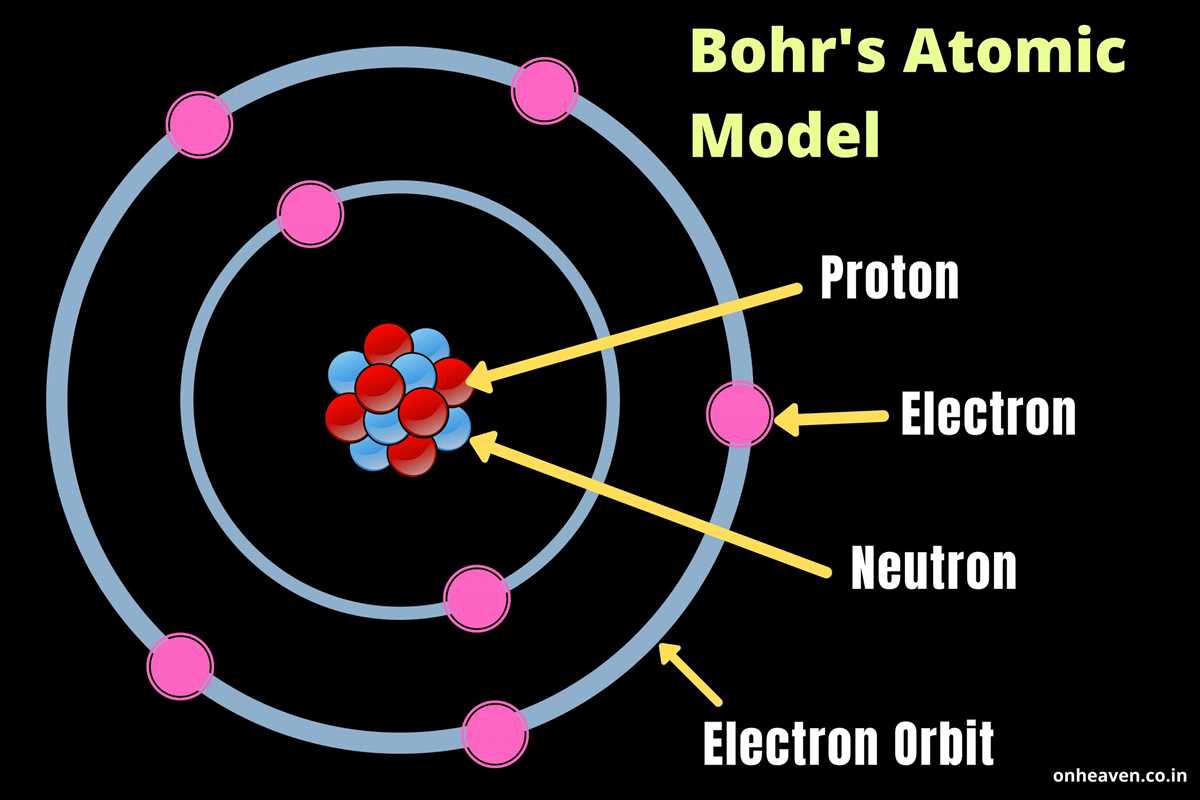
Chapter 4, "Arrangement of Electrons in Atoms," is basically the backstage pass to the atomic show. It's where we unlock the secrets of why some things are shiny and others are dull, why water flows and rocks stay put, and a million other everyday wonders. It’s all about how the electrons, those zippy little particles, are organized. Think of it like a quirky apartment building for electrons. They don't just hang out anywhere; they have specific spots, like different floors and apartments, that they prefer. And how they're arranged dictates everything about the atom.
This isn't just for science geeks, nope! It's for anyone who's ever looked at a rainbow and wondered "how?" or been amazed by electricity. This chapter breaks down the seemingly complex dance of electrons into something you can actually grasp. It's like learning the secret handshake of the universe.
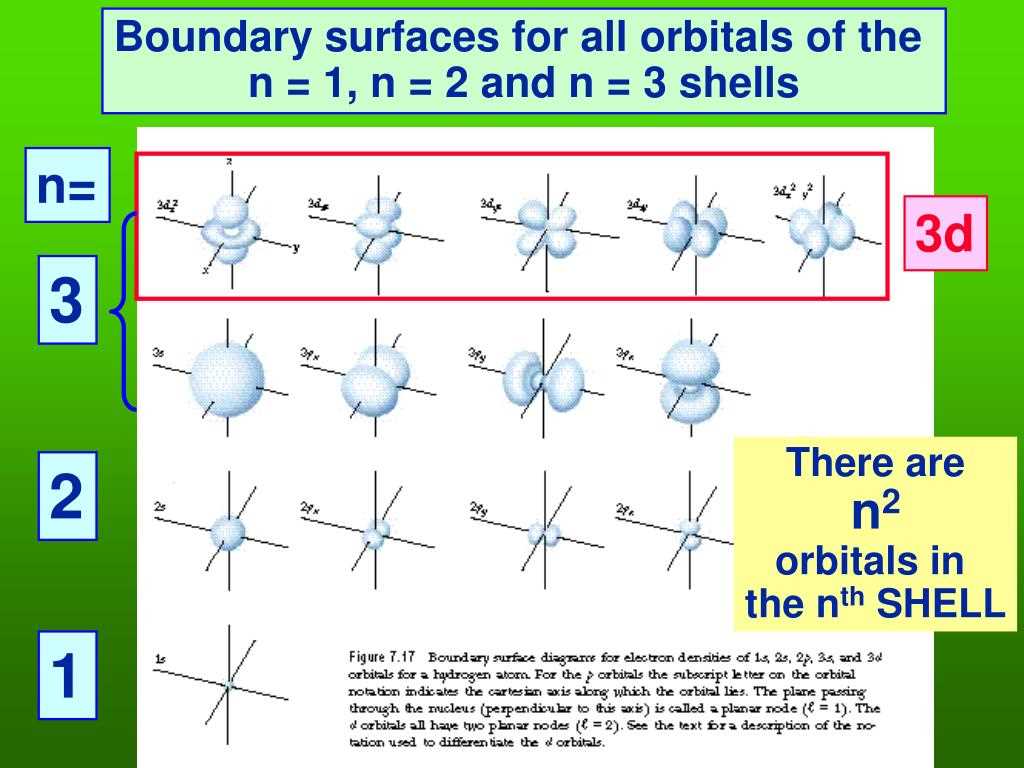
One of the coolest parts is learning about these things called orbitals. Now, don't let the fancy name scare you. Imagine them as fuzzy clouds or specific zones where electrons love to hang out. They’re not like planets whizzing around a sun in a neat little circle. It's more like a probability game. The electron is somewhere in that cloud, and the shape of the cloud tells us a lot. We’ve got these neat little s orbitals that are spherical, like tiny, perfect bubbles. Then there are the more elaborate p orbitals, which look a bit like dumbbells or propellers. It gets even wilder with d orbitals and f orbitals, which are like cosmic abstract art. Each shape is a unique "neighborhood" for electrons.
And the way these orbitals are filled? That’s where the magic really happens. It’s like a game of musical chairs, but with strict rules. Electrons fill up the lowest energy levels first, just like you’d grab the comfiest chair in the room. This is governed by something called the Aufbau principle. Think of it as "building up" the atom, layer by layer, with electrons finding their most stable spots. Then there’s the Pauli exclusion principle, which is like a strict "no roommate" policy for individual electron spots. Each orbital can only hold a maximum of two electrons, and they have to be spinning in opposite directions. It’s like a polite agreement to not bump into each other too much!
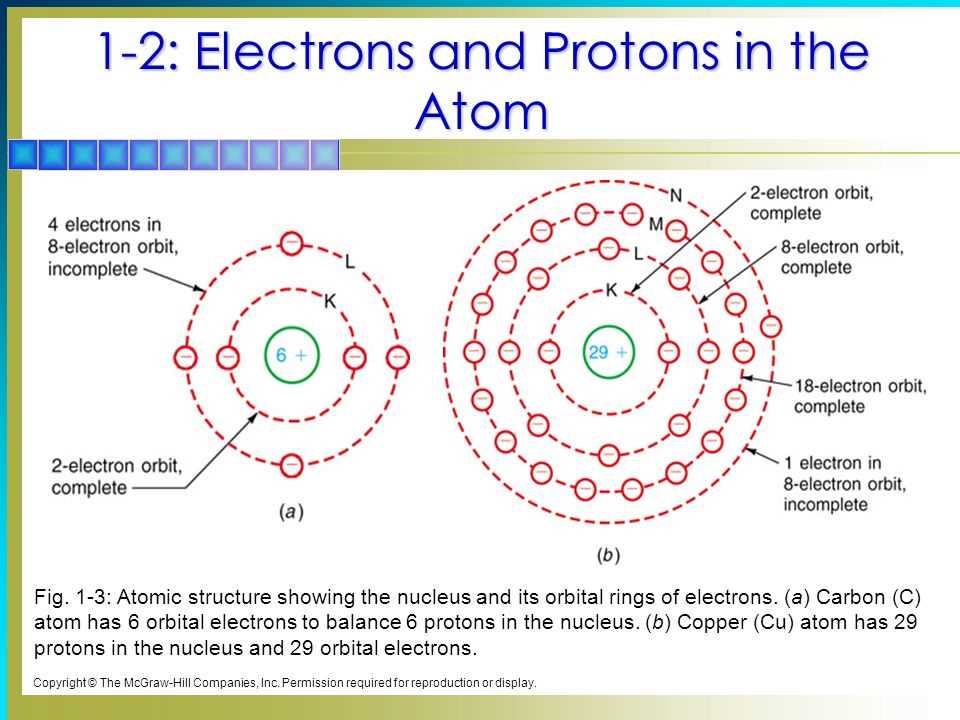
And let’s not forget Hund's rule! This one's a bit more about fairness. If there are multiple identical "rooms" (orbitals) at the same energy level, electrons prefer to spread out and occupy each room singly before they start pairing up. It’s like saying, "Let’s each get our own space before we have to share." This distribution of electrons is what gives atoms their unique personalities. It's why a carbon atom behaves so differently from a gold atom. Their electron "apartments" are arranged in entirely different ways.
What makes this chapter so engaging is that it connects these abstract ideas to the real world. Suddenly, you start seeing the fingerprint of electron arrangements everywhere. The way metals conduct electricity? That’s due to their loosely held outer electrons. The vibrant colors of fireworks? That's the electrons in different atoms getting excited and then calming down, releasing energy as specific colors of light. It’s like the atom is singing its own color song!
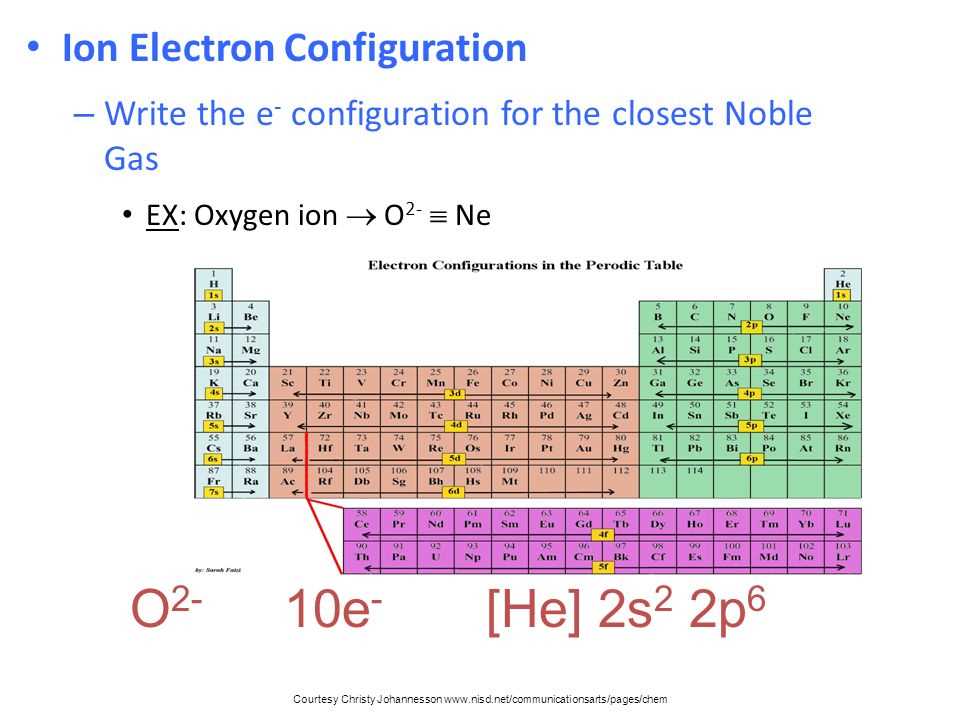
You’ll also get introduced to the concept of electron configurations. This is basically a shorthand way of writing down where all the electrons are. Instead of drawing out all those fuzzy clouds, we use a code. For example, you might see something like 1s²2s²2p⁶. It looks a bit like a secret code, but once you understand the rules, it’s incredibly powerful. It's like having a cheat sheet for understanding an atom's behavior. This code is key to understanding the periodic table, which is arguably one of the most brilliant organizational tools ever invented. The periodic table isn't just a random collection of elements; it’s a map, and electron configurations are the GPS coordinates on that map.
Learning about how electrons are arranged is like peeking behind the curtain of reality. It’s understanding the fundamental building blocks of everything you see, touch, and experience. It's about uncovering the elegant simplicity and surprising complexity that governs the universe. This chapter makes you feel like you're holding a decoder ring for the natural world. It's empowering and, frankly, a little bit addictive!
So, if you've ever been curious about the invisible forces that shape our world, or if you just want to feel like you're in on a cosmic secret, Chapter 4 is your ticket. It’s a journey into the heart of matter, and it’s an adventure that’s surprisingly fun and incredibly rewarding. You’ll start seeing atoms not as just tiny dots, but as intricate, dynamic systems with their own personalities and behaviors. It’s a whole new way to look at the world around you!
