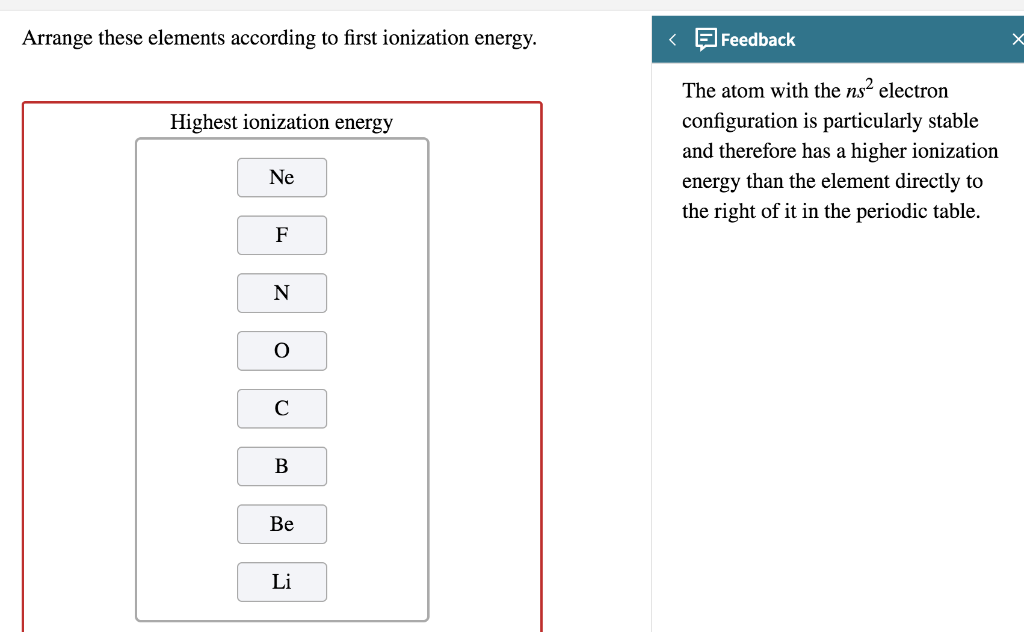
Arrange These Elements According To First Ionization Energy.
Ever feel like you're playing a cosmic game of musical chairs? Well, in the world of chemistry, things can get pretty exciting when you start arranging elements. Forget the periodic table for a moment, because we're diving into a much more dynamic challenge: First Ionization Energy!
Now, don't let the fancy name scare you. Think of it like this: every atom has electrons buzzing around its center, like tiny planets orbiting a sun. The first ionization energy is basically the amount of energy it takes to gently nudge one of those outermost electrons completely off its orbit. It's like giving an electron just enough of a push to say, "See ya!"
And here's where the fun begins. We've got these awesome elements, and we need to figure out which ones are super easy to convince their electrons to leave, and which ones hold on to them like a superhero guards their cape. It’s a real test of your elemental intuition!
Imagine you have a bunch of friends, and you're trying to get them to give you a dollar. Some friends are super generous and will hand it over in a heartbeat. Others? Well, they're a bit more attached to their money. They might ask, "Why do you need it?" or "What are you going to do with it?" They require a bit more convincing, a bit more energy!
That's exactly what's happening with these atoms. Some, like the alkali metals (think Lithium, Sodium, Potassium), are practically begging their electrons to leave. They have only one electron in their outer shell, and it's pretty loosely held. Give them a little nudge, and poof, the electron is gone. These guys have a low ionization energy. They're the generous friends!

On the other hand, you have elements like the noble gases (think Helium, Neon, Argon). These guys are like the super-hoarders of the electron world. Their outer shells are completely full, making them incredibly stable and content. It takes a TON of energy to pry an electron away from them. They are the toughest nuts to crack, and they have a high ionization energy. They're the friends who make you work for that dollar!
So, when you’re asked to arrange elements by their first ionization energy, you're essentially playing a game of "Who's Easiest to Convince?" You're looking at how tightly those outer electrons are held.
Let's consider a few examples. Imagine you're given Sodium and Chlorine. You know Sodium is an alkali metal, with that one lone, easily-lost electron. Chlorine, on the other hand, is a halogen, and it's desperately trying to gain an electron to fill its outer shell. It's going to cling to its own electrons much more tightly. So, Sodium will have a much lower ionization energy than Chlorine.
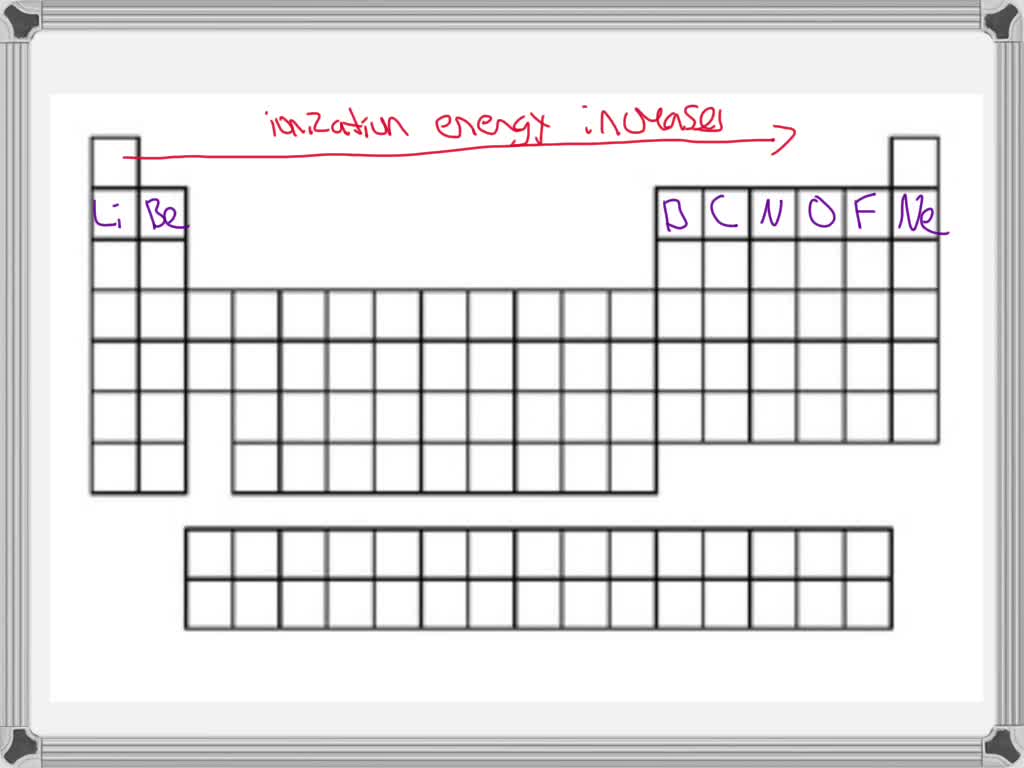
What about Oxygen and Fluorine? Both are in the same period, meaning they have the same number of electron shells. But as you move across a period from left to right, the positive charge in the nucleus gets stronger, pulling those electrons in tighter. Fluorine is further to the right than Oxygen. That means Fluorine's nucleus has a stronger grip on its electrons. So, it will take more energy to remove an electron from Fluorine. Fluorine wins the "Toughest Grip" award in this pairing!
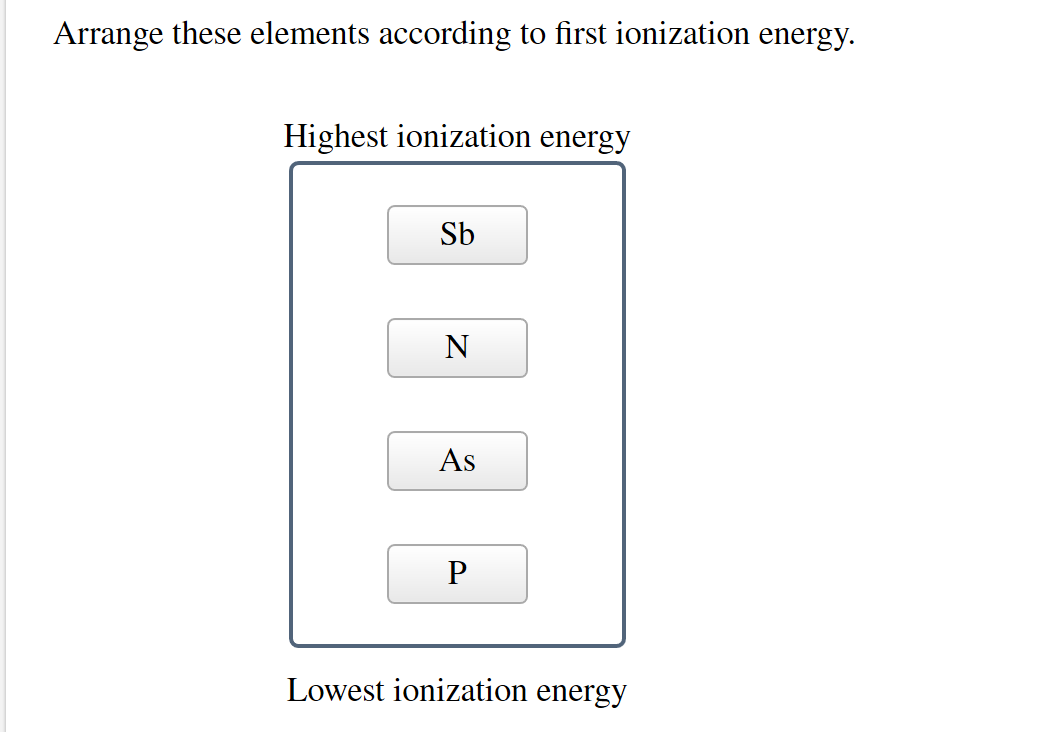
And then there's the vertical aspect, the columns of the periodic table. As you go down a group, the outermost electrons are in shells that are further and further away from the nucleus. It's like the electron is on a much longer leash. The nucleus’s pull weakens significantly. So, elements at the bottom of a group will have lower ionization energies than those at the top.
Think about Lithium versus Cesium. Lithium is up at the top of the alkali metals. Its outermost electron is relatively close to the nucleus. Cesium, however, is way down at the bottom. Its outermost electron is practically waving goodbye already! Cesium has a super low ionization energy.

The real magic happens when you get a list of elements and have to put them all in order. It’s like a puzzle where each piece has a hidden value. You have to consider its position on the periodic table, its group, its period, and its tendency to either give up or gain electrons. It’s a fascinating interplay of attraction and repulsion, a dance of electrons and protons!
It’s not just about memorizing numbers; it’s about understanding the behavior of these elements. It’s about seeing the patterns emerge, the trends that make the periodic table so incredibly elegant. When you start to grasp first ionization energy, you’re unlocking a deeper understanding of why elements behave the way they do. It's like learning the secret handshake of the chemical world!
So, if you ever get the chance to arrange elements according to their first ionization energy, jump at it! It's a brain-tickling challenge that’s surprisingly fun. You'll be surprised at how much you can learn about the fundamental nature of matter, all by figuring out who's most willing to share their electrons. It’s an entertaining way to appreciate the subtle yet powerful forces that govern our universe, one electron at a time. Give it a try, and you might just find yourself hooked on this atomic game!
