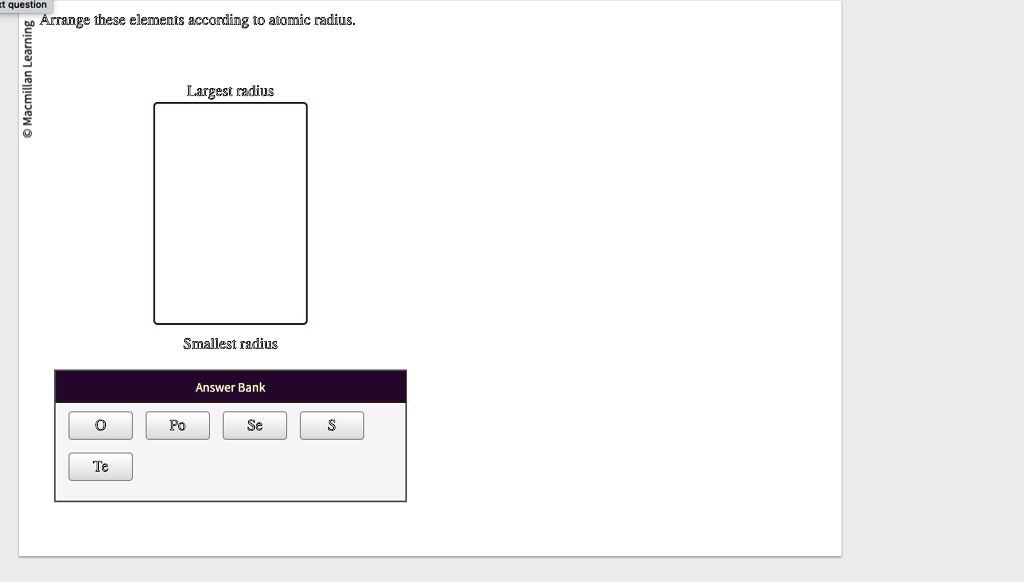
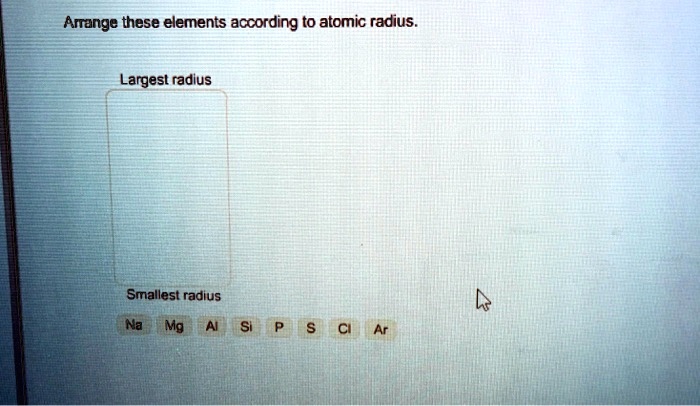
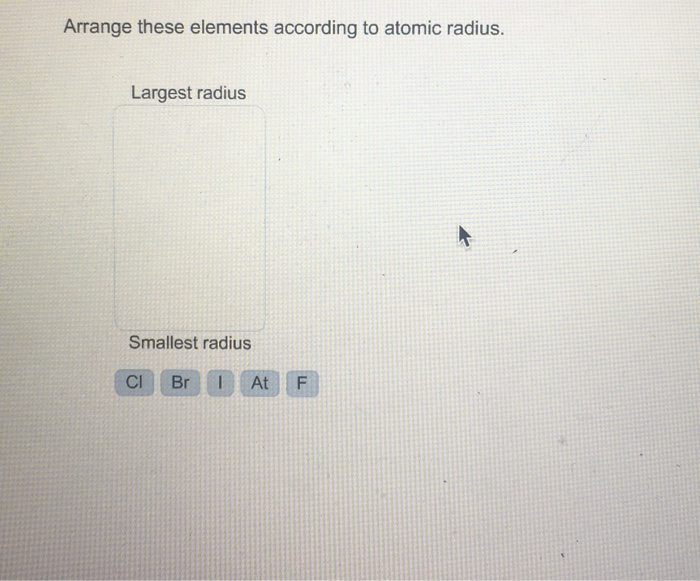
Arrange These Elements According To Atomic Radius

Hey there, science curious folks! Ever found yourself staring at a periodic table and wondering, "What's the deal with these elements?" They're like the ultimate building blocks of everything, right? From the air we breathe to the phones in our hands, it's all made of these tiny, fascinating things. Today, we're going to chat about something super neat: atomic radius. Sounds a bit technical, maybe? But trust me, it's actually pretty cool and makes a whole lot of sense once you get the hang of it.
So, what exactly is atomic radius? Think of an atom like a tiny, fuzzy ball. Atomic radius is basically how big that fuzzy ball is. It's the distance from the center of the atom (the nucleus, with its protons and neutrons) to the outermost edge of its electron cloud. It’s not a super precise measurement because electrons are, well, a bit unpredictable, zipping around like hyperactive toddlers. But we can still get a pretty good idea of their "size."
Now, imagine you have a bunch of these fuzzy balls, and you want to arrange them from smallest to biggest. That's kind of what we're doing with atomic radius! It's not just a random sorting exercise; there are actually some really cool patterns involved. And understanding these patterns can tell us a lot about how elements behave. Pretty neat, huh?
Let's dive into the nitty-gritty of why this size thing matters. Think about it like this: if you're trying to pack a bunch of marbles into a box, their size makes a huge difference. Similarly, the size of atoms affects how they interact with each other. It influences things like how easily they form bonds, their melting points, and even their electrical conductivity. It’s like the foundational characteristic of an element!
The Periodic Table's Secret Code: Trends in Atomic Radius
The periodic table isn't just a colorful chart of elements; it's like a giant cheat sheet for predicting their properties. And atomic radius follows some pretty predictable trends as you move across and down this table. It's almost like the universe left us a little puzzle to solve!
Let's start by looking at the trends as we move across a period (that's the horizontal rows). If you start on the left side of a period and move towards the right, you'll notice that the atomic radius generally decreases. What's going on there?
Imagine you're in a neighborhood, and the houses (atoms) are all on the same street (period). As you move down the street, each house has more people living in it (more protons in the nucleus). These extra protons pull on the electrons with a stronger electrical attraction, like a more powerful magnet. Even though there are more electron "rooms" in the houses as you go along, the stronger pull from the nucleus makes the whole house shrink a bit.
So, element number 3 (Lithium, Li) is bigger than element number 9 (Fluorine, F) in the same row. It's like the Lithium house has fewer strong magnets pulling on its inhabitants, so it can spread out a bit more. Fluorine, with its stronger nucleus, keeps its electron inhabitants closer in.
Now, let's switch gears and look at the trends as we move down a group (that's the vertical columns). Here, things work a little differently, and the atomic radius generally increases. Think of it like going down a skyscraper. Each floor is a new "energy level" for the electrons, kind of like a new floor in our house analogy.
As you go down a group, you're adding more electron shells, or layers, around the nucleus. It's like adding more and more floors to our house. Even though the nucleus is getting stronger with more protons, those new, outer shells of electrons are pretty far away from the nucleus. They're shielded by all the inner shells. It's like the people on the top floors of the skyscraper are much further from the ground floor, and the pull from the ground floor isn't as strong on them.
So, Hydrogen (H) at the very top of group 1 is super tiny. Then comes Lithium (Li), which is bigger. Then Sodium (Na), even bigger. And so on, all the way down to elements like Cesium (Cs) or Francium (Fr), which are quite large atoms. It’s like our neighborhood is expanding upwards, with each new story adding significant height.
Putting It All Together: Arranging Elements
Okay, so we've got the rules: smaller as you go right across a period, and bigger as you go down a group. Now, let's try a little game. Imagine you're given a few elements and asked to arrange them by their atomic radius. This is where the fun really begins!
Let's take a few common elements: Oxygen (O), Sulfur (S), and Selenium (Se). Where do they live on the periodic table? Oxygen is in period 2, group 16. Sulfur is in period 3, group 16. And Selenium is in period 4, group 16. See a pattern? They're all in the same group!
Since they are all in the same group, we know the trend is to get bigger as we go down. So, Oxygen will be the smallest, followed by Sulfur, and then Selenium will be the largest. It's like a little race where the furthest one down the track wins the "biggest atom" prize.
What about Sodium (Na), Magnesium (Mg), and Chlorine (Cl)? These are all in period 3. Sodium is on the far left, Magnesium is next, and Chlorine is towards the right. We know that as we move across a period, the atomic radius generally decreases.
So, Sodium will be the largest, Magnesium will be smaller, and Chlorine will be the smallest. It's like a shrinking contest across the street! The element at the beginning of the row has more "space" to spread out, while the one at the end is being pulled in tighter.
A Little Challenge: Sodium (Na), Potassium (K), and Phosphorus (P)
This one's a bit trickier, as it involves moving both across and down! Sodium (Na) is in period 3, group 1. Potassium (K) is in period 4, group 1. Phosphorus (P) is in period 3, group 15.
First, let's compare Sodium and Potassium. They're both in group 1. Potassium is below Sodium, so Potassium is larger than Sodium. Easy peasy!
Now, let's compare Sodium and Phosphorus. They're both in period 3. Phosphorus is to the right of Sodium. So, Sodium is larger than Phosphorus because the radius decreases as you move across a period.
Putting it all together: Potassium is the biggest. Between Sodium and Phosphorus, Sodium is bigger. So, the order from largest to smallest atomic radius is: Potassium > Sodium > Phosphorus. How cool is that? You just cracked a periodic table code!
The beauty of understanding atomic radius is that it's a fundamental concept that helps us predict a whole bunch of other chemical behaviors. It's like learning the basic rules of a game; once you know them, you can start understanding the more complex strategies.
So, next time you see a periodic table, don't just see a bunch of symbols. See a world of atoms, each with its own unique size, and understand the elegant patterns that govern their existence. It’s a reminder that even in the smallest things, there’s a whole lot of order and a whole lot of wonder. Keep exploring, keep questioning, and keep finding the cool in science!
