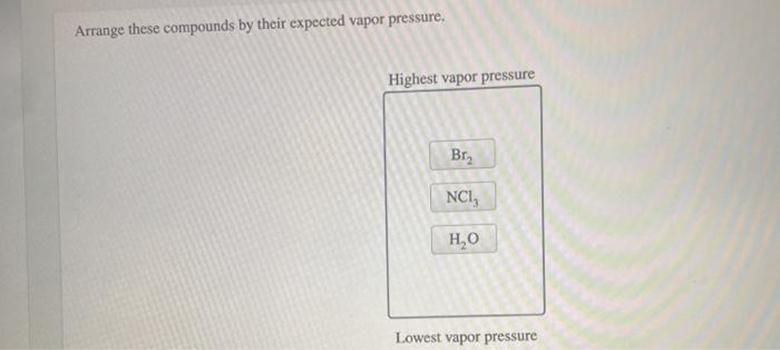
Arrange These Compounds By Their Expected Vapor Pressure
Okay, gather 'round, science curious friends! We're about to dive into a little game. It's like sorting socks, but way more exciting. We're going to take some chemical compounds and decide which one is the biggest escape artist.
Think of vapor pressure like a compound's desire to hit the road, to become a gas and float away. Some molecules are practically packing their bags already. Others are clinging on for dear life, like they just remembered they left the oven on.
We've got a lineup of some interesting characters today. Let's meet our contestants! They're all vying for the title of "Most Likely to Vanish." It's a prestigious award, really.
The Contenders
First up, we have Water. Yeah, the stuff you drink, shower in, and occasionally spill on your keyboard. It's pretty common, right? We know this one well.
Then there's Ethanol. This is the alcohol you find in hand sanitizer and, well, other things. It's got a bit of a reputation.
Next, we have Acetone. You know this one from nail polish remover. It has that distinct, sharp smell. It’s a powerful cleaner, and it likes to get around.
Moving on, we encounter Dichloromethane. This one's a bit more of a mouthful. It's used as a solvent and in some industrial processes. It's not exactly in your kitchen cupboard.
And finally, our last contestant is Sodium Chloride. That’s right, plain old table salt! The stuff that makes your fries taste amazing. It’s a solid, dependable sort of molecule.

The Great Escape: Arranging by Vapor Pressure
Now, the fun part: arranging them! We're putting them in order from the one that's most eager to become a gas (highest vapor pressure) to the one that’s most determined to stay put (lowest vapor pressure). It's a scientific scavenger hunt!
The Wanderer (Highest Vapor Pressure)
At the very top of our list, the undisputed champion of wanting to escape, is Acetone. This stuff is practically vibrating with anticipation to become a vapor. It's like it sees a tiny gas-shaped door and just has to go through it.
If you’ve ever opened a bottle of nail polish remover, you know exactly what I mean. That smell just hits you, and then it’s gone, off on its adventures. It doesn't linger like a shy guest.
Acetone is a true free spirit. It's all about that quick getaway. It’s got that “see ya later, wouldn’t wanna be ya” attitude when it comes to staying liquid.
The Quick Jumper
Next in line, not quite as wild as acetone but still a ready traveler, is Ethanol. It’s got a good dose of wanderlust. It’s not as frantic as acetone, but it's definitely not planning on settling down.
Think about it. When you use hand sanitizer, the alcohol smell is noticeable, and then it evaporates pretty quickly, leaving your hands feeling dry. It’s not sticking around for a prolonged chat.
Ethanol is like that friend who’s always up for a spontaneous road trip. It’s got energy, and it’s not afraid to use it to become a gas. It’s a step up in terms of sticking around, but not by much.
The Reluctant Traveler
Now we get to Water. Water has a bit of a personality. It can be a gentle stream or a raging ocean, but as a pure substance in these comparisons, it's not as eager to become a gas as our first two.
Sure, puddles dry up. That’s water becoming vapor. But it takes its sweet time compared to acetone or ethanol. It’s got some intermolecular forces holding it back.
Water is more like the friend who agrees to a road trip but packs way too much and insists on stopping at every scenic overlook. It’s got its reasons for taking its time. It’s not exactly a couch potato, but it’s no sprint champion either.
The Stubborn Sitter
Getting into the more determined crowd, we have Dichloromethane. This one’s a bit more of a stick-in-the-mud, scientifically speaking. It’s not as readily willing to join the gas party.

It’s got a stronger grip on itself. It’s not going to just up and leave at the drop of a hat. It requires a bit more persuasion, or in this case, energy (heat), to convince it to become a vapor.
Dichloromethane is like the friend who’s happy to stay home and watch a movie. It's not saying no to everything, but it's definitely not initiating the adventure. It’s got a longer leash when it comes to its liquid form.
The Ultimate Homebody (Lowest Vapor Pressure)
And finally, at the very end of our spectrum, the ultimate homebody, the one who is absolutely not going anywhere without a serious fight, is Sodium Chloride. That's right, salt!
Salt is a crystal. It's got a very strong structure. It loves being a solid, and it requires an immense amount of heat to even think about becoming a liquid, let alone a gas.
Trying to get salt to vaporize under normal conditions is like trying to convince your cat to enjoy a bath. It’s not happening. It has incredibly low vapor pressure. It’s the definition of staying put.
So, there you have it! Our lineup, from the flightiest to the most grounded. It’s a bit like a personality test for molecules.
Acetone: "I'm outta here!"

Ethanol: "Yeah, I'm going too, but maybe I'll send a postcard."
Water: "Alright, fine, I'll pack my bags, but I'm taking my time."
Dichloromethane: "You guys go ahead. I'm comfortable."
Sodium Chloride: "Laughable. I'm not moving."
It's a fun way to think about how these different substances behave, isn't it? They all have their own little quirks and preferences. And sometimes, the most common things, like salt, are the most surprisingly resistant to change. Who knew table salt was such a stubborn character?
Next time you're using nail polish remover or just looking at a salt shaker, you can chuckle at their hidden desires to become a gas. Or, in salt's case, their utter lack of desire. Science can be fun, even when it’s about things that don't want to move.
