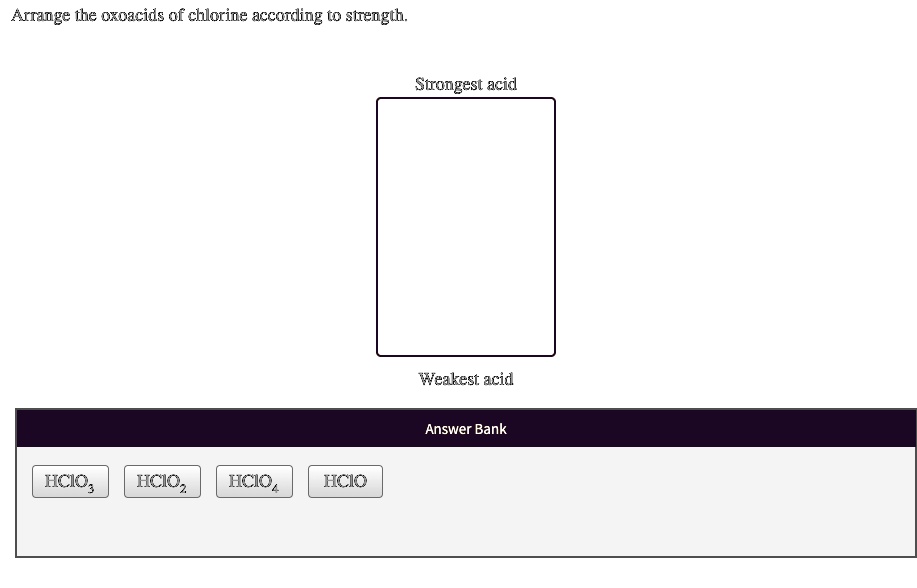
Arrange The Oxoacids Of Chlorine According To Strength
Hey there, fellow curious minds! Ever found yourself staring at a bunch of cleaning products under the sink and wondering, "Which one of these is really the toughest?" Well, today we're going to dive into something a little more scientific, but don't worry, it's going to be as relaxed as a lazy Sunday morning. We're talking about the amazing world of chlorine's oxoacids and how to line them up from weakest to strongest. Think of it as a friendly competition for acidity!
Now, what on earth are "oxoacids of chlorine," you might ask? Imagine chlorine, that element you often hear about in relation to swimming pools and keeping things disinfected, decided to get really friendly with oxygen and hydrogen. When they team up, they form these special compounds called oxoacids. They're like a little family, and just like any family, some members are a bit more assertive than others when it comes to being acidic.
Why should you even care about this, you might wonder? Well, understanding acidity is like having a secret superpower for understanding how things work in the world around you. It helps explain why some things clean better, why certain reactions happen, and even why that lemon juice is so wonderfully tart. Plus, it’s a fun little brain-tickler that makes you feel a bit smarter, right? So, let’s get this party started!
The Chlorine Oxoacid Lineup: From Wimps to Wonders!
Let's meet the contenders in our acidity showdown. We have four main players:
- Hypochlorous acid (HOCl)
- Chlorous acid (HClO₂)
- Chloric acid (HClO₃)
- Perchloric acid (HClO₄)
Now, the order of strength is a bit like arranging your favorite snacks on a shelf. You've got your "grab-and-go" ones at the front, and the ones you save for a special occasion at the back. In our case, the "special occasion" here means more acidic. So, let’s put them in order from weakest to strongest.
The Gentle Giant: Hypochlorous Acid (HOCl)
At the very bottom of our strength ladder, we have hypochlorous acid. This little guy is the most laid-back of the bunch. Think of it as the mildest hand sanitizer you’ve ever used – it does the job, but it’s not going to make you wince.

Why is it the weakest? Well, it all comes down to how easily it can release a hydrogen ion (H⁺). The easier it is to let go of that H⁺, the stronger the acid. Hypochlorous acid is a bit of a clingy parent; it holds onto its hydrogen ion pretty tightly. You might find this in some milder disinfectants or even in your own body’s immune system, working subtly to keep you healthy.
It’s like that friend who offers a polite "Are you sure you don't want another biscuit?" rather than aggressively pushing the whole plate at you. Gentle and effective is its motto.
The Growing Teenager: Chlorous Acid (HClO₂)
Next up, we have chlorous acid. This one is a bit more energetic than its predecessor. It's like a teenager who's started to find their voice – a little more assertive, a little more likely to make a statement. It’s stronger than hypochlorous acid, meaning it's more willing to share its hydrogen ions.
Imagine it’s trying to help clean up a spill. It’s more determined than HOCl, willing to put in a bit more elbow grease. While not as common in everyday household products as HOCl, it plays a role in certain chemical reactions. It’s that middle child who’s still figuring things out but is definitely not as shy as the youngest.

Think of it as stepping up your game from a gentle nudge to a friendly, but firm, tap on the shoulder. It’s getting there, with a bit more gusto!
The Reliable Helper: Chloric Acid (HClO₃)
Now we’re getting to the serious stuff! Chloric acid is significantly stronger. This one is like your go-to, reliable friend who always shows up when you need them. It’s quite a potent oxidizer, which means it’s really good at "stealing" electrons from other substances, which is key to many chemical reactions, including cleaning and bleaching.
Think of it as the cleaner that means business. If HOCl is a gentle wipe, HClO₃ is a good scrub. It's used in some industrial applications and can be quite useful, but you wouldn't want to leave it lying around unattended. It’s got more oomph, more willingness to let go of those H⁺ ions and get things done.
It’s the friend who doesn’t just offer to help move furniture; they show up with the muscles and the plan. Powerful and effective, but requires a bit of respect.

The Undisputed Champion: Perchloric Acid (HClO₄)
And finally, the undisputed champion of our chlorine oxoacid strength competition: perchloric acid! This is the superhero of the group. It’s incredibly strong, one of the strongest acids known. It’s like the ultimate cleaning product that tackles even the toughest grime, or the most potent disinfectant that guarantees germ annihilation.
Perchloric acid is extremely eager to release its hydrogen ions. It’s the most willing to give up its electron buddies. This makes it incredibly reactive and a powerful oxidizing agent. You'll find it used in very specific, high-stakes situations, like rocket fuel oxidizers and in some laboratory procedures where extreme acidity is required. It’s not something you’d find under your kitchen sink, and for good reason – it demands a lot of caution and expertise.
Imagine it as the seasoned professional, the expert surgeon, or the a-team of cleaners. It’s the one you call when you absolutely, positively need the strongest solution. It’s the most acidic and therefore the strongest on our list. Dominant and potent.
Putting It All Together: The Acidity Spectrum
So, to recap, when we arrange these oxoacids of chlorine by their strength, from weakest to strongest, it looks like this:
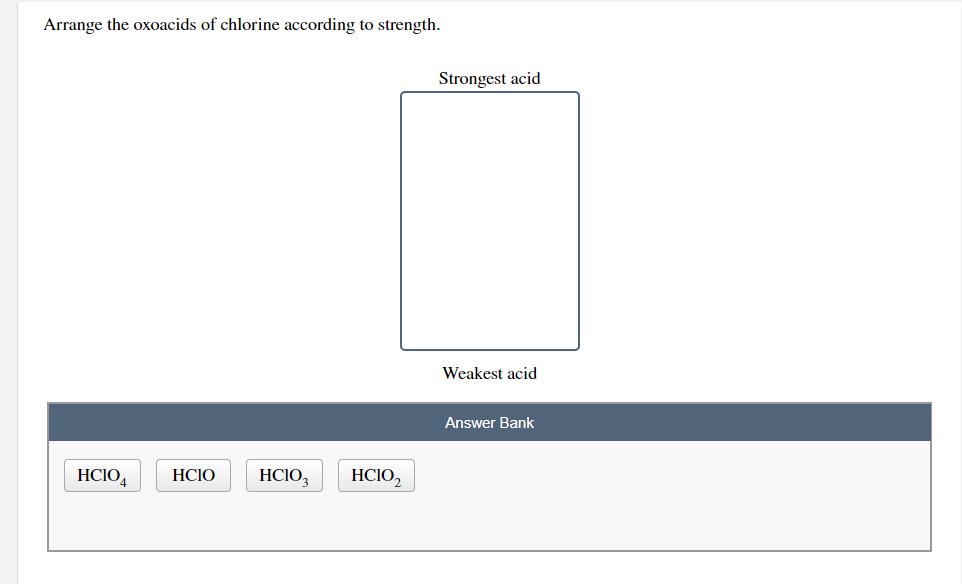
- Hypochlorous acid (HOCl) - The gentle nudge
- Chlorous acid (HClO₂) - The friendly tap
- Chloric acid (HClO₃) - The determined push
- Perchloric acid (HClO₄) - The full-blown charge!
The reason for this order boils down to the number of oxygen atoms bonded to the central chlorine atom. As you add more oxygen atoms, the acid gets stronger. Think of the oxygen atoms as little electrical sponges that pull electron density away from the O-H bond, making it easier for the hydrogen to break free. More oxygen means more pulling, which means easier release of H⁺, which means a stronger acid!
It’s like having more hands helping to push a door open. The more hands you have (the more oxygen atoms), the easier it is to push that door (release the H⁺ ion). Simple, right?
Why This Matters (Beyond Just Being Cool Science!)
Understanding this acidity order isn't just a fun academic exercise. It helps us predict how these substances will behave. For example:
- Cleaning Power: The stronger the acid, the more reactive it is and potentially the better it can break down certain types of dirt or stains. This is why we see hypochlorites (derived from hypochlorous acid) in many household bleaches and disinfectants – they’re effective but manageable.
- Safety: Knowing the strength helps us understand the safety precautions needed. Perchloric acid, being so strong, requires very specific handling and storage. It’s like knowing the difference between handling a feather and handling a heavy dumbbell.
- Industrial Uses: In chemistry labs and industries, knowing the precise acidity of a compound is crucial for carrying out specific reactions. It’s the difference between using a gentle lubricant and a heavy-duty industrial solvent.
So, the next time you're pondering over a cleaning bottle or just enjoying a bit of science trivia, remember the oxoacids of chlorine! They’re a fantastic example of how a small change in chemical structure can lead to a big difference in properties. It’s a reminder that even in seemingly simple elements, there’s a whole spectrum of behavior waiting to be discovered. Pretty neat, huh?
