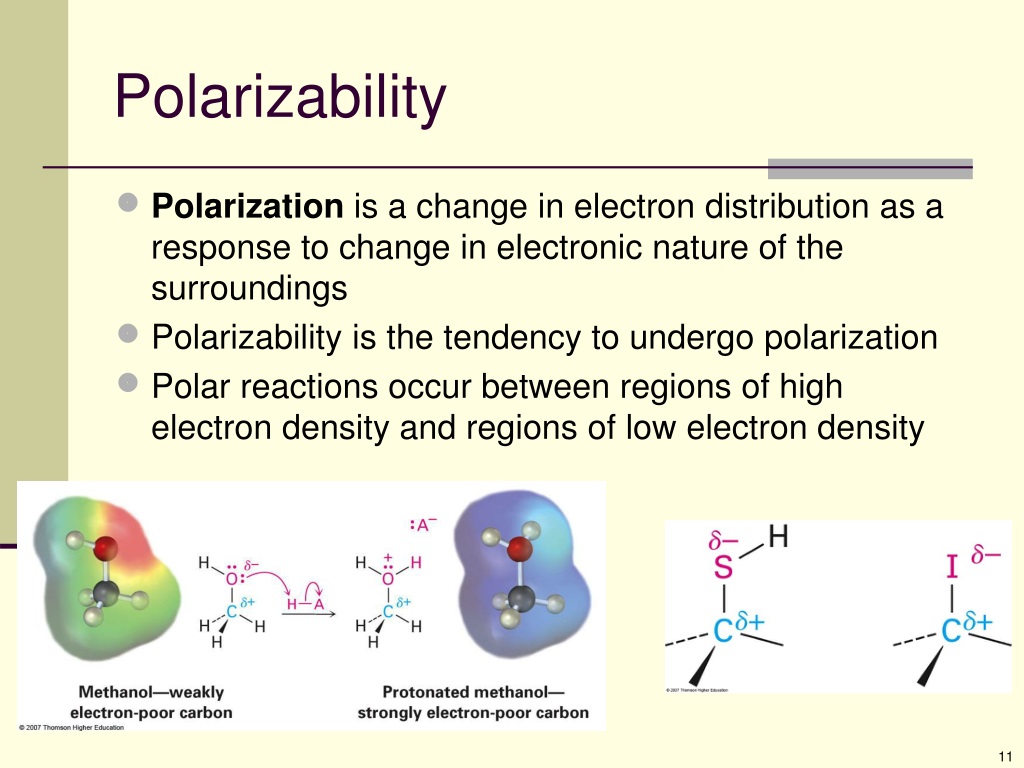
Arrange The Nucleophiles In Order Of Polarizability.

Alright, settle in, folks! Let's talk about something that sounds super fancy but is actually kind of like figuring out which person at a party is going to get the most attention. We're talking about nucleophiles and their magical ability to be, well, polarizable. Now, I know what you're thinking. "Polarizable? Is that even a real word?" Oh, it's real, and it's a doozy!
Think of it this way: imagine a bunch of friendly characters, all ready to lend a hand (or a pair of electrons, if you're being technical). These are our nucleophiles. They're the helpful types, the ones who are drawn to the center of things, the electron-deficient spots. They want to get in there and share their electron love. But not all nucleophiles are created equal, my friends.
Some are like those super spread-out, comfy beanbag chairs. You can just sink right into them. Others are like those tiny, tightly wound springs. They're ready to go, but they're not going to let themselves get too squished. This, my dears, is the essence of polarizability.
So, how do we arrange these helpful little critters in order? It’s a bit like judging a popularity contest, but with more electrons and less awkward dancing. We want to put the ones that are the most "squishy" or "spread-out" at the top of our list. These are the ones that can easily have their electron clouds nudged and stretched. They're the chill ones, the laid-back types, the ones who are totally fine with sharing their electrons in a big, amorphous blob.
Imagine a giant, fluffy cloud. That's a highly polarizable nucleophile. It's easy to imagine that cloud getting a little bit smushed. Now imagine a tiny, hard pebble. That's not very polarizable at all. It's going to take a lot more effort to change its shape, if it even changes at all.

Let's get down to the nitty-gritty, the juicy gossip of the nucleophile world. We're going to put the biggest, baddest electron-cloud carriers right at the top. These are the ones that have more electrons floating around, and those electrons are further away from the nucleus, making them more susceptible to a friendly poke.
First up on our polarizability pedestal, we've got the big guys. Think of atoms that are really far down the periodic table. They've got more shells, more space for those electrons to roam. The larger the atom, the more easily its electron cloud can be distorted. It's like trying to bend a twig versus trying to bend a giant redwood. The redwood, with all its mass, is actually a bit more "bendable" in this electron-y way.

So, if we're talking about elements, things like iodine (I⁻) and bromine (Br⁻) are going to be pretty high up there. They're the gentle giants of the nucleophile universe, happy to let their electrons get a little squished for the greater good. They've got these big, billowy electron clouds that are just begging to be reshaped.
Then we have our mid-range folks. Think of things like chlorine (Cl⁻). It's still pretty good at being squished, but maybe not quite as enthusiastically as iodine. It’s like a slightly smaller, but still very comfortable, armchair. It's got a decent electron cloud, ready to participate, but it's not going to be completely engulfed by the situation.
And then, we get to the little guys. These are the compact, no-nonsense nucleophiles. Think of things like fluorine (F⁻). This little fella is so small and tightly packed, its electrons are practically glued to its nucleus. It's like trying to get a super-compressed spring to bend. It's not going to happen easily.
So, a general rule of thumb, if you want to impress your chemistry friends (or just win at a very niche trivia night), is that bigger is generally better when it comes to polarizability. It's the triumph of the spread-out over the squeezed-in!
Now, it's not just about size, oh no! We also have to consider how many electrons are hanging around. More electrons often means a bigger, fluffier electron cloud. It's like adding more stuffing to that beanbag chair. The more stuffing, the more likely it is to conform to whatever shape you throw at it.

But honestly, for most of us just trying to get through the day (or a particularly tricky chemistry problem), focusing on the size is a pretty good shortcut. Imagine you're choosing a pillow. You want the one that's going to cradle your head perfectly, right? You're not going for the rock-hard, tiny little pebble pillow. You want the plush, accommodating one.
So, when you see a list of these nucleophiles, and you're asked to put them in order of how easily they can be "squished" or "deformed" by an incoming electric field (that’s the fancy term, but "squished" is way more fun, right?), just remember our big, fluffy cloud analogy. The bigger the cloud, the more polarizable the nucleophile.
It’s a strange little corner of chemistry, this polarizability business. But honestly, there's something kind of charming about it. It's about embracing the squish, the willingness to adapt and share. And who among us can't appreciate a good squish now and then? It’s a reminder that sometimes, the most helpful things are the ones that are a little bit spread out, a little bit willing to go with the flow. So next time you see a list of nucleophiles, just picture them as a lineup of helpful, electron-donating characters, and pick the one with the biggest, fluffiest cloud. You’ll be a polarizability pro in no time!
