Arrange The Non Covalent Interactions In Order Of Strength
Ever wondered what makes things stick together? It's not just superglue! In the tiny world of molecules, there are invisible forces at play. These forces are called non-covalent interactions. They're like the polite handshakes and gentle nudges between molecules.
Think of it like a party. You have different types of guests. Some are super chatty and grab everyone's attention. Others are more reserved, offering a quiet nod. These interactions are the molecular equivalent of that. They might not be as strong as a chemical bond, but they're incredibly important.
And guess what? We can actually put them in order! It's like sorting your favorite candies from most delicious to just okay. This ranking isn't just for scientists in lab coats. It helps us understand so much about the world around us.
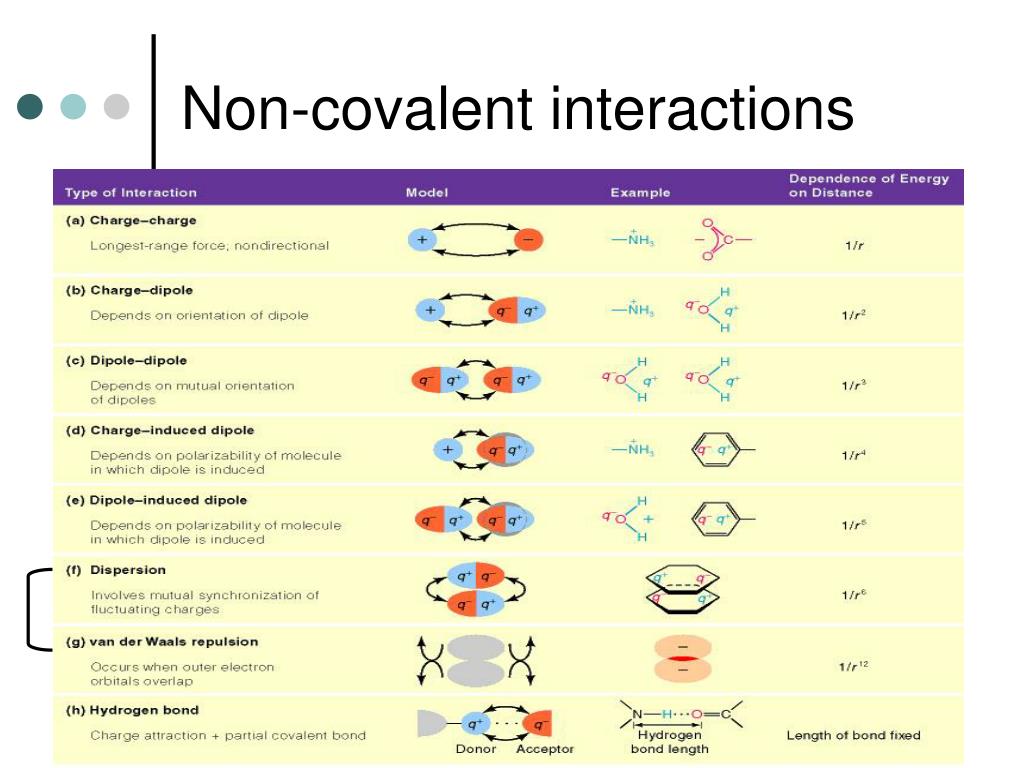
The strongest of these gentle persuaders is the ion-dipole interaction. Imagine a really charged guest at the party, like someone wearing a bright, flashy outfit. They attract everyone around them, like magnets! This happens when a full-on charged ion meets a molecule with a bit of a split personality.
This molecule, the dipole, has a positive end and a negative end. The ion, being fully charged, can't resist this partial charge. It's a powerful attraction, like a direct invitation to dance. These are super important in things like dissolving salt in water. That salty taste? Thank the ion-dipole!
Next up, we have the hydrogen bond. These are like the popular kids at the party, everyone wants to be near them. It's not a full charge, but a strong attraction nonetheless. It happens when hydrogen is bonded to a very electronegative atom, like oxygen or nitrogen.
Think of it as a hydrogen atom with a tiny, positive "halo." This halo is then attracted to the negative part of another molecule. Water molecules, for example, are best friends because of hydrogen bonds. It's what makes water so special and life-giving.
Then come the dipole-dipole interactions. These are the friendly acquaintances. They're not as intense as the ion-dipole or the hydrogen bond, but they definitely add to the vibe of the party. Here, two molecules, each with their own positive and negative ends, find each other.
Their positive ends are drawn to the other's negative ends. It's like a polite wave and a smile across the room. It's not a deep connection, but it's enough to keep them from wandering too far apart. Many organic molecules rely on these to stay together.
After that, we have ion-induced dipole interactions. This is like someone with a really strong personality walking into a room. They don't have a charge themselves, but they can make others feel a charge. It's a bit of subtle influence.
The charged ion comes near a neutral molecule. This can push or pull the electrons in the neutral molecule. This creates a temporary dipole, a little spark of attraction. It's not a conscious effort, but it happens!
And finally, at the very bottom of our strength list, are the London dispersion forces. These are the quietest guests at the party. They're so subtle, you might not even notice them! Even though they're the weakest, they are everywhere.
These arise from tiny, temporary fluctuations in electron distribution. Imagine a group of people all milling about. For a split second, more electrons might gather on one side of a molecule. This creates a fleeting, weak dipole.
This weak dipole can then induce a dipole in a neighboring molecule. It's like a tiny, ripple effect. Even though each individual interaction is weak, when you have billions and billions of them, they add up! They're the glue for nonpolar molecules.

So, why is this so entertaining? Because it’s like solving a molecular puzzle! We're looking at these invisible forces and figuring out their pecking order. It's a bit like a friendly competition, but instead of trophies, we get understanding.
What makes it special? These interactions are the unsung heroes of biology and chemistry. They dictate how proteins fold, how drugs bind to targets, and even why your favorite snack has its texture. Without them, life as we know it wouldn't exist!
It’s a subtle dance, a delicate balance of attractions. Understanding this order gives us a peek into the hidden mechanisms of the universe. It's a bit like learning the secret handshake of the cosmos.
The ion-dipole interaction is the rockstar. It's got that powerful pull. Think of a salt crystal dissolving in water. The water molecules, with their partial charges, are completely captivated by the full charges of the sodium and chloride ions. It’s a dramatic embrace!
Next in line is the hydrogen bond. This is the charismatic leader of the pack. It's responsible for water's amazing properties. It's why ice floats and why water is such a great solvent. It's the backbone of DNA, holding those two strands together.
Following that, we have dipole-dipole interactions. These are the reliable friends. They might not be as flashy, but they're consistent. Molecules that have permanent polarities, like in many plastics and oils, use these to hang out together. It’s the molecular equivalent of a steady relationship.

Then comes the ion-induced dipole interaction. This is the influencer. It can subtly change the behavior of others. It's important when a charged molecule meets a neutral one, and that charge causes a temporary, attractive polarization. It’s like a whisper that gets a whole crowd moving.
And finally, the ubiquitous London dispersion forces. These are the quiet workers in the background. They're the collective power of many tiny, fleeting attractions. Even nonpolar molecules, which seem so aloof, are held together by these. Think of how gases liquefy – that's the dispersion forces doing their work!
The order from strongest to weakest is: ion-dipole > hydrogen bond > dipole-dipole > ion-induced dipole > London dispersion forces. It's a hierarchy of attraction, a fascinating look at molecular charisma!
Learning this order is like unlocking a secret code. Suddenly, you can look at molecules and predict how they might behave. It's empowering and, dare I say, a little bit magical.
It makes you appreciate the intricate beauty of the molecular world. Every interaction, no matter how weak, plays a crucial role. It's a symphony of invisible forces, all playing their part.
So, the next time you marvel at how water behaves, or how a biological process unfolds, remember these non-covalent interactions. They are the unseen architects of so much of what we experience. They are the polite but persistent forces that shape our world.

It’s a bit like a molecular popularity contest, but with real-world consequences. The ones with the strongest attractions get the most attention, and their influence is profound. But even the least popular have their role to play.
So, there you have it! A little peek into the world of molecular attractions, ranked from the most clingy to the most casually acquainted. Isn't it amazing what invisible forces can do? It’s a fascinating journey, and the more you learn, the more you’ll want to explore.
This ordering isn't just a dry list of facts. It's a story. It's about how things connect, how they influence each other, and how these seemingly small interactions lead to incredibly large phenomena. It’s the molecular drama that unfolds all around us, all the time!
The beauty of it is its simplicity and its complexity. Simple concepts, when applied to the vastness of molecules, create incredibly complex and beautiful results. It's like a recipe where a few basic ingredients, when combined just right, make an exquisite meal.
So, don't be shy to dive a little deeper. Explore these interactions. They are the secret language of matter, and understanding them is like learning to speak fluent molecular! It’s a rewarding adventure that opens up a whole new perspective on the world.
The invisible forces that bind the universe together are often the most fascinating.
