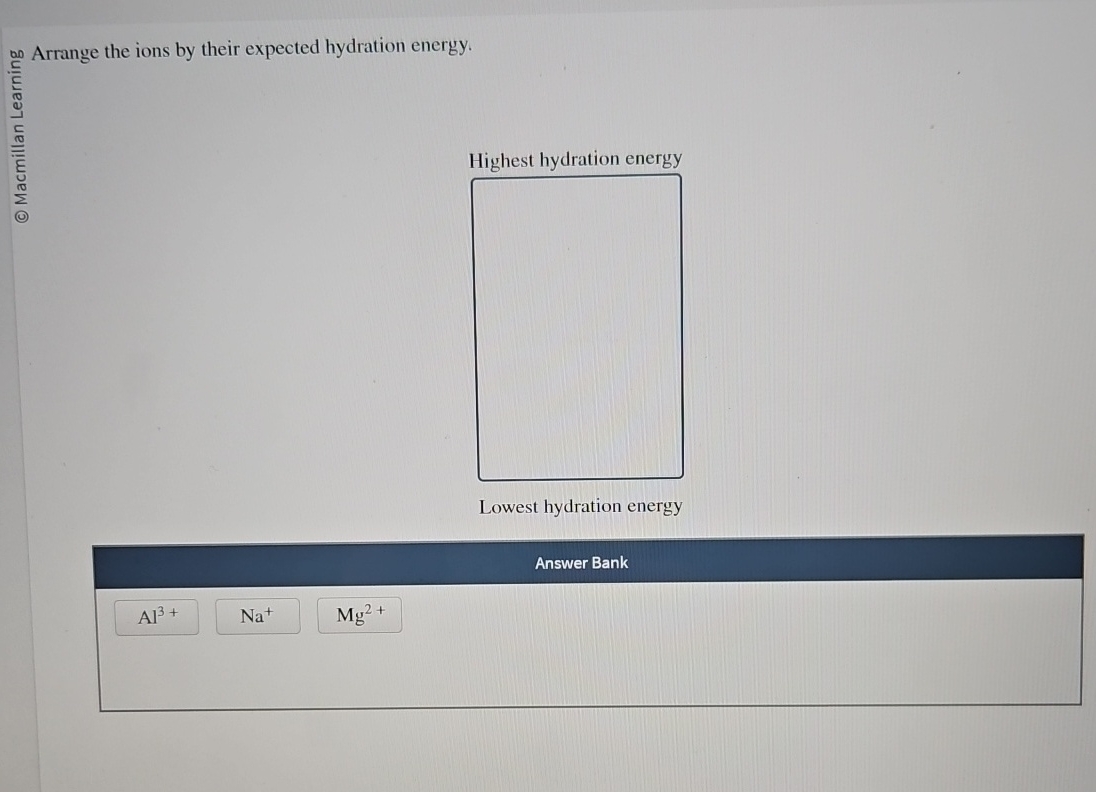
Arrange The Ions By Their Expected Hydration Energy
So, you've got a bunch of these little fellas running around. They’re not exactly people, but they’ve got a charge. You know, like a tiny, zappy personality. We’re talking about ions. And these ions, bless their electrically charged hearts, sometimes decide to take a dip. A big, splashy, watery dip.
Now, this isn't just any old swim. Oh no. This is about how much these ions get themselves all cozy with the water molecules. Think of it like a big hug. Some ions are super popular, and the water molecules go absolutely bonkers for them. Others? Well, they get a polite nod and maybe a lukewarm handshake. It's all about something called hydration energy. Sounds fancy, right? But really, it's just a measure of how much of a fuss the water makes over each ion.
Imagine you’re at a party. Some people walk in, and suddenly everyone’s rushing over, offering them drinks, pulling up chairs. That’s a high hydration energy ion. They’re the life of the party, the celebrity guest. Then there are the folks who just kind of blend into the background. People might say hi, but nobody’s throwing a ticker-tape parade. That’s your low hydration energy ion. Perfectly nice, but not exactly causing a scene.
Now, we’ve got a few contenders for our little hydration energy ranking. We've got the usual suspects, the ones you probably know from chemistry class, even if you tried to forget. We’ve got Lithium, or Li⁺, which is basically the shy but incredibly popular kid. Then there’s Sodium, or Na⁺, the dependable friend, always there. Next up is Potassium, or K⁺, a bit more laid-back, but still gets attention. And finally, we have Cesium, or Cs⁺, the cool, aloof type. They’re here, but they’re not exactly breaking a sweat trying to make friends.
So, how do we arrange these guys? It’s not rocket science, but it does involve a certain je ne sais quoi of ionic charm. We’re looking for the ones that get the most enthusiastic water molecule welcome. It’s like a popularity contest, but with more H₂O.

Let’s start with the one that throws the biggest water party. This ion, you see, is like that one person who walks into a room and instantly sparks a dozen conversations. The water molecules are practically throwing themselves at it. It’s tiny, and that makes all the difference. All that charge packed into a small space? It’s irresistible to those slightly negative oxygen atoms in the water. They’re like little magnets, and this ion is the super-strong magnet.
The most popular ion gets the biggest water hug!
Who is this reigning champion of hydration? Drumroll, please! It’s our little, energetic friend, Lithium (Li⁺)! Yes, that tiny powerhouse. It’s so small and zappy that the water molecules are practically forming a mosh pit around it. They’re clinging on for dear life, giving it the ultimate, intense hydration experience. The energy released when this happens? Off the charts! It's like Liam Neeson in a tiny, charged package, demanding attention and getting it in spades.

Now, who’s next in line for the water embrace? We move up in size a bit. This ion is still quite good at attracting those water molecules. It's not quite the frenzied mob that gathers around Lithium, but it's definitely a lively gathering. Think of it as a moderately popular celebrity at a book signing. People are eager, they're reaching out, but it's not quite a riot. The charge is still strong enough to pull those water molecules in with gusto. This one is a bit bigger than Lithium, so the charge isn't quite as concentrated, but it's still a very attractive ion indeed.
This next contender in our hydration lineup is our old friend, Sodium (Na⁺). Sodium is like the reliable friend who’s always up for an adventure. The water molecules are definitely keen to hang out with Sodium. It’s got a good grip, and the water molecules are happy to oblige. The energy released here is still significant, showing that Sodium is a pretty popular dude in the water world. It’s the friendly wave, the warm smile, the invitation for a coffee. Definitely a strong showing.
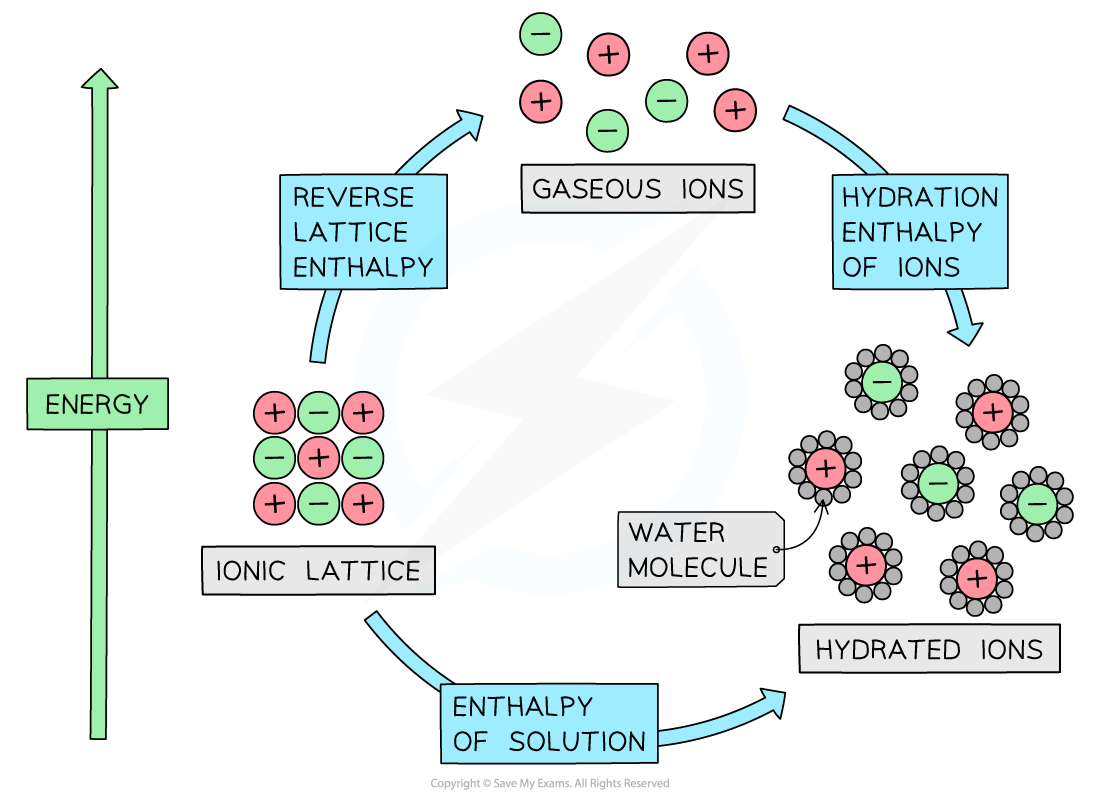
As we continue our journey through the world of hydrated ions, we encounter another player. This one is a bit more relaxed about the whole thing. It's larger than Sodium, so its charge is spread out a bit more. The water molecules still find it attractive, but the intensity of the hug is starting to mellow out. It's like the friend who's happy to chat, but doesn't necessarily need you to hang on their every word. They're enjoying the company, but they've got other things on their mind. The water molecules are still interested, but the urgent need to cling is less pronounced.
Taking the third spot in our surprisingly important hydration hierarchy is Potassium (K⁺). Potassium is like the chill cousin. They’re definitely part of the family, and everyone likes them, but they’re not demanding the spotlight. The water molecules offer a comfortable embrace, a gentle squeeze. The energy released is still there, a testament to their ionic appeal, but it's not the wild party Lithium or even Sodium can command. It’s a pleasant afternoon picnic, not a rave.
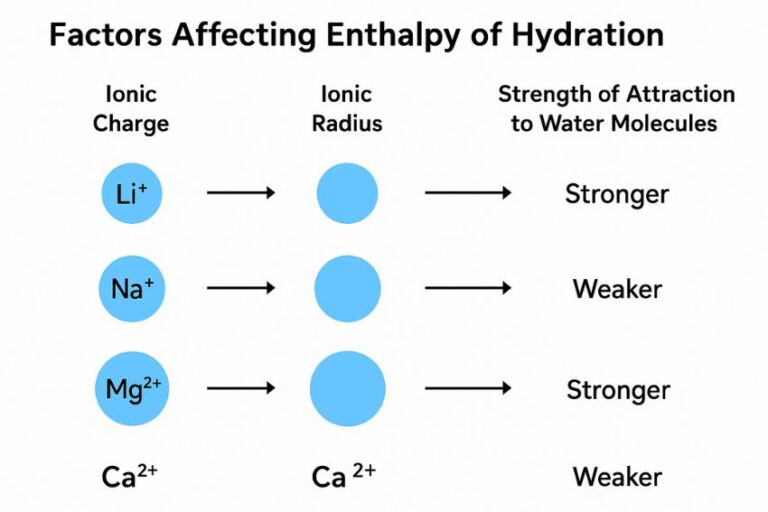
And then, we have our final contestant. This ion is the epitome of laid-back. It’s the biggest of the bunch, and its charge is, shall we say, rather diffused. The water molecules… well, they acknowledge its presence. They might drift by, offer a polite nod, maybe a brief, almost apologetic, wave. But a full-on, enthusiastic hug? Not so much. The water molecules are more like casual acquaintances who bump into each other at the grocery store. There’s a polite smile, a quick “how are you?” and then they both move on with their day.
This ultimate chill-out ion, the one that barely gets a ripple in the water world, is Cesium (Cs⁺). Cesium is the one you invite to a party, and they show up, have one drink, and then politely excuse themselves. The water molecules are just not that bothered. The energy released from hydrating Cesium is the lowest on our list. It’s the quiet hum of existence, the gentle breeze rather than a gust of wind. And you know what? There’s a certain elegance in that, isn't there? Not everyone needs to be the life of the party. Sometimes, just being present is enough.
So, there you have it. The grand procession of ions, ranked by how much the water molecules are willing to throw themselves at them. It’s a testament to size, charge, and the undeniable allure of a good water-based cuddle. Who knew chemistry could be so… huggy?
