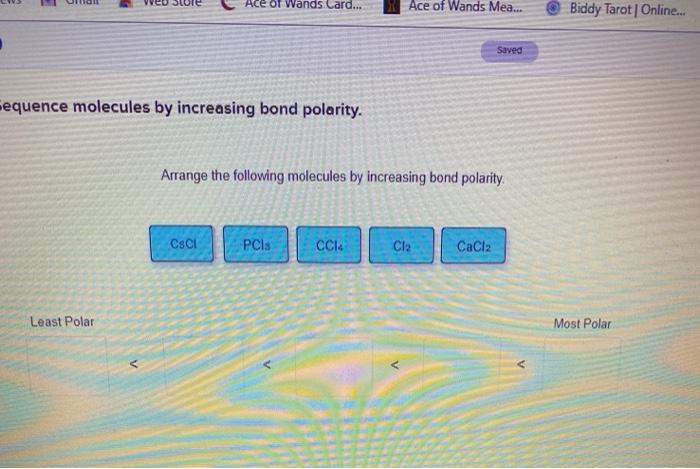
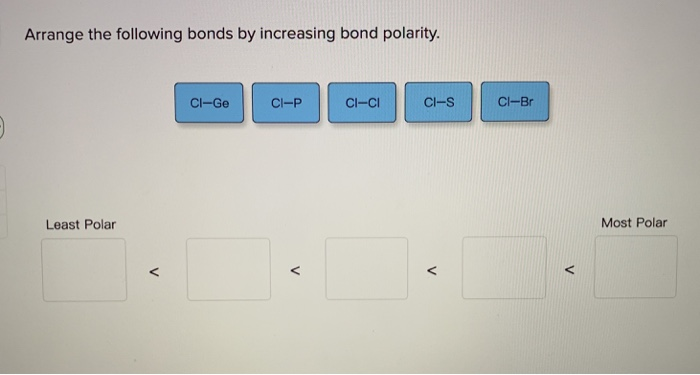
Arrange The Following Molecules By Increasing Bond Polarity.

Hey, you! Grab your mug, let's chat about something super cool, but also maybe a tiny bit nerdy. We're gonna talk about molecules and their bond polarity. Sounds fancy, right? But honestly, it's kinda like figuring out who's the most popular kid at the molecular party. You know, who's hogging all the electron attention?
So, what even is bond polarity? Imagine two atoms are holding hands, right? In a covalent bond, they're sharing electrons. But sometimes, one atom is a little bit of a hog. It pulls those shared electrons closer to itself. That makes it a little bit negative, and the other atom, feeling a bit left out, becomes a little bit positive. It’s all about the electronegativity, baby! Like a chemical popularity contest.
We're gonna arrange some molecules from least polar to most polar. Think of it as a totally chill, science-y ranking. No pop quizzes, I promise! We're just gonna look at these guys and see who's the most… unequal in their electron sharing. It’s like picking the winners and losers in a super low-stakes game of tug-of-war.
First up on our little journey, we have… drumroll please… N₂! Nitrogen gas. You know, the stuff that makes up most of the air we breathe? Yeah, that guy. So, in N₂, we have two nitrogen atoms. And guess what? They're identical. Like twins. So, they're gonna share those electrons perfectly equally. No hogging, no leaving anyone out. It’s the definition of a nonpolar covalent bond. Imagine them both just chilling, perfectly balanced. No charge separation here, folks. This is our baseline. The absolute least polar of the bunch. Think of it as the wallflower of the molecular dance. Perfectly content, not causing any drama.
Next in line, we’re stepping it up a notch. We've got CO₂, carbon dioxide. Now, this one's interesting. You'd think, "Ooh, carbon and oxygen, they're different!" And you'd be right! Oxygen is way more electronegative than carbon. It's like oxygen's got that irresistible charm, pulling those electrons towards itself. So, the oxygens get a slight negative charge, and the carbon gets a slight positive charge. BUT! Here's the kicker, and it’s a doozy. CO₂ is a linear molecule. Think of it like this: O=C=O. The two oxygen atoms are on opposite sides, pulling equally in opposite directions. It’s like two people pulling on a rope, but they’re facing each other and pulling with the same strength. The pull cancels out! So, even though the individual bonds are polar, the overall molecule is nonpolar. Mind. Blown. It’s a masterclass in deception, this one. A true anarchist of the chemical world, making you think it's one way when it's totally another. So, it’s technically more polar than N₂ because of those individual polar bonds, but its symmetrical shape makes it effectively nonpolar. Weird, right?
Alright, moving on to our next contender: NH₃, ammonia. Now we're talking! Nitrogen is more electronegative than hydrogen. So, the nitrogen atom is gonna be pulling those shared electrons from the hydrogens. This creates a partial negative charge on the nitrogen and partial positive charges on the hydrogens. But, unlike CO₂, NH₃ is not symmetrical. It's got a cool pyramidal shape. Imagine the nitrogen is at the top of a little pyramid, and the three hydrogens are at the base. All those little pulls towards the nitrogen are happening on the same side of the molecule, more or less. There's no canceling out going on here, no beautiful symmetry. It’s like a bunch of kids all running towards one ice cream truck. They’re all contributing to a flow in one direction. So, the dipole moment – that’s the fancy word for the overall polarity – is significant. This is where we start seeing some real electron inequality. NH₃ is definitely pulling ahead in the polarity race. It’s like the popular kid who’s actually got the biggest smile, drawing everyone’s attention.
And now, ladies and gentlemen, prepare yourselves for the reigning champ, the undisputed king of our little polarity lineup: HF, hydrogen fluoride! Oh boy. Fluorine is the most electronegative element on the entire periodic table. Seriously, it’s a black hole for electrons. It’s like the ultimate electron magnet. It just devours them. Hydrogen, on the other hand, is like, "Whoa, dude, can I have some electrons back?" So, in HF, the fluorine atom is aggressively pulling the electrons towards itself. This creates a very large partial negative charge on the fluorine and a very large partial positive charge on the hydrogen. The difference in electronegativity between hydrogen and fluorine is huge. It's not just a little nudge; it's a full-on shove. This makes HF a very, very polar molecule. We’re talking a seriously strong dipole moment here. It's like the rockstar of the group, grabbing all the spotlight. If you were to imagine these molecules swimming in a solvent, HF would be the one making the biggest splash, really interacting with other polar molecules. It’s the ultimate loner, in a way, because its own internal tug-of-war is so intense.
So, let's recap our little journey. We went from the perfectly balanced twins of N₂ (nonpolar, zero polarity), to the sneaky symmetrical trickster CO₂ (effectively nonpolar due to symmetry, even with polar bonds), then to the pleasantly lopsided pyramid of NH₃ (moderately polar), and finally to the electron-devouring superstar HF (highly polar). It’s a spectrum, right? A beautiful, messy spectrum of how atoms decide to share (or not share!) their electron toys.
Why does this even matter, you ask? Well, this bond polarity thing? It dictates a ton about how molecules behave. Like, do they dissolve in water? (Water is polar, so it likes to hang out with other polar things. Like dissolves like, remember that little mantra?) Do they have high boiling points? Do they react with other molecules? It's all linked back to how those electrons are distributed. So, understanding bond polarity is like getting a secret decoder ring for the entire world of chemistry. It’s the foundation upon which so much of chemical behavior is built.
Think about it like people again. A bunch of people who are all super similar and get along perfectly? They're probably gonna have a pretty chill, predictable group dynamic. Like N₂. But if you have a group with some really strong personalities, some who pull and some who are pulled? Things get a lot more interesting, a lot more dynamic. That's where our NH₃ and HF come in. The interactions are gonna be way more complex and, frankly, more fun to study. It’s the difference between a quiet afternoon in the library and a wild, unpredictable party. And we know which one is more exciting, right?

So, next time you're looking at a molecule, remember this little chat. Think about those atoms, think about their electronegativity hunger. Are they sharing nicely? Is one being a bit of a diva? The answers to those questions will tell you so much about what that molecule is up to. It’s like being a molecular detective, piecing together clues based on electron distribution. Pretty neat, huh? It’s not just abstract symbols on a page; it’s the fundamental forces that shape everything around us. From the air we breathe to the water we drink, it all comes down to how these tiny particles are playing electron fetch.
And the cool part is, this concept of polarity isn’t just for these specific molecules. It applies across the board! You can look at any bond between different atoms and make a pretty good guess about its polarity. Just pull out your handy-dandy periodic table, find those electronegativity values, and you’re basically a chemistry wizard. It’s like having a superpower, but instead of flying, you can predict how substances will interact. Which, let’s be honest, is way more useful in everyday life. Unless you need to escape a burning building, then maybe flying is better. But for, you know, making tea? Polarity is your friend.
So, there you have it! Our little lineup of molecular polarity. From the calm and collected to the dramatic and intense. It’s a whole universe of electron sharing happening all around us, all the time. And now you’re a part of understanding it. Pretty awesome, right? Now, go forth and be the most chemically savvy person you know. Just don't ask me to explain ionic bonds over coffee next time. That's a whole other can of worms… or, rather, a whole other type of bond. 😉
