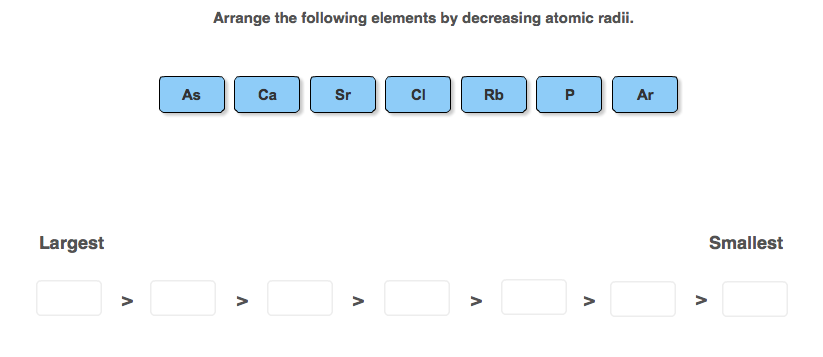
Arrange The Following Elements By Decreasing Atomic Radii
Alright, gather 'round, you magnificent bunch of knowledge-seekers! Today, we're diving headfirst into the wonderfully weird world of atomic radii. Think of atoms as tiny little people, right? And their atomic radii? That’s basically their personal bubble. Some are tiny, shy introverts who like to keep to themselves, while others are big, boisterous extroverts who need a whole lot of elbow room. And guess what? We're going to arrange some of these atomic superstars by how much space they like to take up, from most bubble-having to least bubble-having.
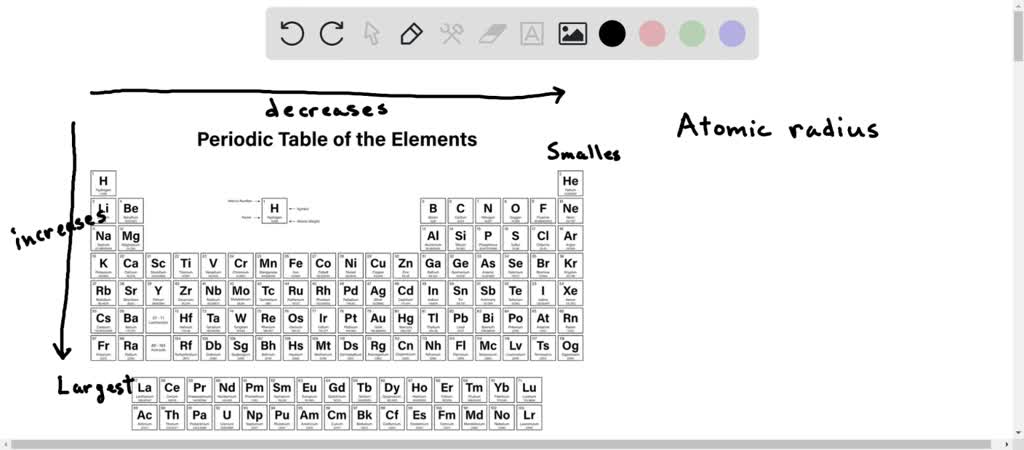
Now, before you picture me with a ruler and a magnifying glass, measuring teeny-tiny atom arms, let's get our bearings. Atomic radius is, in simple terms, the distance from the nucleus (the atom's brainy center) to the outermost electron (the atom's gossipy outer layer). It's a bit like measuring from the center of a very, very fluffy cloud to its edge. And these radii? They're not just some abstract concept; they actually tell us a ton about how an atom behaves. It's like knowing your friend's personal bubble size – you know whether to hug them from across the room or give them a big ol' bear hug!
The Big Kahunas: Where the Atoms Get Their Stretch On
So, let's talk about the elements you've been given. We've got a motley crew: Potassium (K), Lithium (Li), Sodium (Na), and Rubidium (Rb). Imagine them at a party. Who's going to be hogging the dance floor? Who's trying to squeeze into a conversation in a crowded corner?
First up, the reigning champ of sprawl, the king of cosmic chill: Potassium (K)! This guy is like your uncle who arrives at a party and immediately starts telling the same story for the fifth time, taking up all the conversational space. Potassium is a metal, and metals in the same row (period) of the periodic table tend to get bigger as you go from right to left. Why? Because as you add more protons to the nucleus, they pull on the electrons, but when you move left, the number of electron shells (the "layers" of your atom's bubble) increases, giving it more room to expand. Plus, the outer electrons in Potassium are further away from the pull of the nucleus than in its neighbors to the right.
Think of it like this: The nucleus is the DJ at the party, and the electrons are the dancers. In Potassium, the DJ is still trying to control a lot of dancers, but they're spread out over a really big dance floor. So, even though the DJ is powerful, the dancers are pretty independent and can move around a lot. Potassium is huge, folks. It’s practically overflowing its personal bubble!
Next on the expansive journey, we have Rubidium (Rb). Rubidium is like Potassium's slightly more laid-back, but still very spacious, cousin. It’s also an alkali metal, and just like Potassium, it’s rocking a seriously generous atomic radius. If Potassium is your uncle, Rubidium is your dad who’s still telling slightly embarrassing childhood stories. It’s in the same group (column) as Potassium, but it’s even lower down. Why is it bigger? Because it has more electron shells. More shells means more distance from the nucleus. It’s like adding another floor to your house – suddenly, you have way more living space!
So, we have Potassium and Rubidium, both boasting impressive real estate. But who wins the "most sprawling" award between these two? Generally, going down a group on the periodic table means larger atomic radii. This is because each step down adds another electron shell, pushing those outer electrons further and further away from the nucleus’s grip. So, while both are massive, Rubidium, being further down, is generally a smidge bigger than Potassium. It's like comparing a McMansion to a slightly less McMansion-y mansion. Still huge!

The Mid-Sized Movers: Not Too Big, Not Too Small
Now, let's bring in our other contenders. We've got Sodium (Na) and Lithium (Li). These guys are like the dancers who are having a good time but aren't necessarily stage-diving or starting a mosh pit. They’re keeping it cool, occupying their fair share of space.
Sodium (Na) is up next. If Potassium and Rubidium are your sprawling suburban homes, Sodium is your comfortable, well-appointed city apartment. It's still a big deal, but it's more contained. Sodium is also an alkali metal, and it's directly above Potassium in the periodic table. This means it has one less electron shell than Potassium. Fewer shells mean the nucleus can pull on the electrons a bit more effectively, shrinking that atomic radius. It’s still pretty large compared to many elements, but it’s definitely not the king of sprawl anymore.

Imagine Sodium at the party. It's got its own circle of friends, it’s laughing and chatting, but it’s not, you know, blocking the entire hallway. It’s got a good amount of personal space, but it’s not demanding the whole room.
And finally, we arrive at the pocket rocket, the compact powerhouse: Lithium (Li)! Lithium is the smallest of this bunch. Think of Lithium as the tiny studio apartment in a trendy neighborhood. It's got everything it needs, it's functional, but it's definitely not giving you room to swing a cat. Lithium is at the very top of the alkali metal group. It has the fewest electron shells of the group, and the nucleus can really clamp down on those outermost electrons. It’s like the DJ has everyone dancing super close to the booth, and they’re all really feeling the beat together.

So, to recap our partygoers: Rubidium is the one doing the conga line across the entire venue. Potassium is right behind it, a close second. Sodium is in the middle of the dance floor, having a blast but not taking over. And Lithium? It’s busting out some impressive moves, but it’s right there, close to the action, in a neat little package.
The Grand Reveal: The Order of Awesomeness (and Size!)
So, let's arrange our element pals from the biggest atomic bubble to the smallest. Remember, we're talking about decreasing atomic radii. This means we start with the biggest and work our way down.
The Largest to Smallest Lineup:
- Rubidium (Rb): The undisputed champion of space-hogging. It's got the most electron shells and a nucleus that's just a little too far away to keep those outer electrons completely in check. It's practically wearing a "Do Not Disturb" sign made of electrons!
- Potassium (K): A very close second! Just like Rubidium, it’s got a good number of electron shells. It’s the king of its castle, and its castle is pretty darn big.
- Sodium (Na): Our comfortable city dweller. It’s got fewer shells than Rb and K, so the nucleus can exert a bit more influence. It’s still a substantial atom, but it’s more compact.
- Lithium (Li): The petite powerhouse! With the fewest electron shells, its nucleus has the tightest grip. It’s small, but don't let that fool you; it’s still a mighty element!
And there you have it! The grand, slightly exaggerated, and hopefully entertaining, journey through atomic radii. Remember, it’s all about those electron shells and the nucleus’s magnetic personality. Keep your eyes peeled for more elemental adventures – the periodic table is a goldmine of quirky characters and surprising facts, just waiting to be explored!
