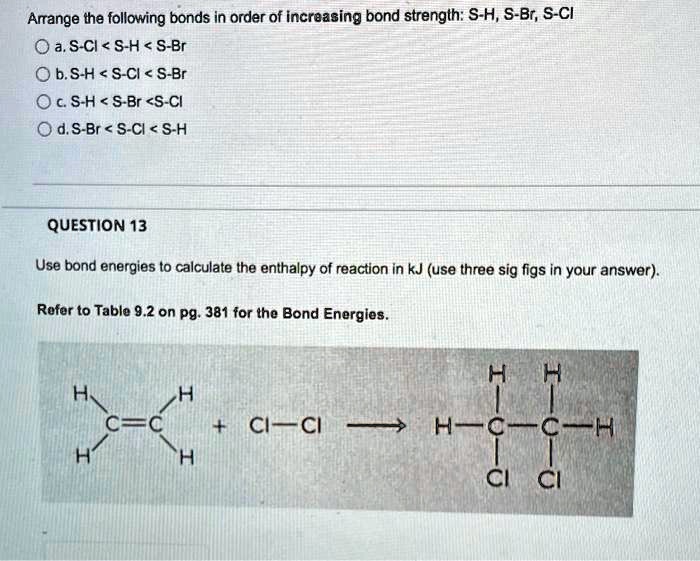
Arrange The Following Bonds In Order Of Increasing Bond Strength

Hey there, science buddies! Today, we're diving into something super cool, but don't worry, it's not going to be like that time I tried to explain quantum physics to my cat. This is way more chill. We're talking about bond strength. Imagine you've got a bunch of tiny little Lego bricks that make up everything in the universe. These bonds are like the connections holding those bricks together. And guess what? Some connections are stronger than others. Who knew the universe had its own hierarchy of clinginess?
So, the big question is, how do we figure out which of these invisible, yet oh-so-important, connections are the toughest? It’s kind of like trying to decide if your toddler’s grip on a cookie is a mild suggestion or a full-on, no-letting-go policy. We need a way to rank them from the "aww, that's cute" to the "whoa, you're gonna need a crowbar for that one."
Let's get right to it. We're going to arrange some common chemical bonds in order of increasing bond strength. Think of it as a popularity contest, but for atoms trying to stick together. The weaker the bond, the lower it ranks. The stronger the bond, the more it’s crushing it at the top.
The Usual Suspects: A Lineup of Bonds
Alright, imagine you're at a party, and you've got a few key players mingling. We’ve got our participants ready to be judged. These are the bonds you'll bump into pretty often in your chemistry adventures. We're talking about bonds between different types of atoms, and sometimes, even the same atoms. It’s a diverse crowd!
First up, we have the Hydrogen Bond. Now, this one is a bit of an oddball. It's not a true chemical bond in the same way as others, but it’s super important. Think of it like a friendly wave or a gentle nudge between molecules, especially in water. It’s there, it’s influential, but it’s not exactly a death grip. We’ll put it down as our starter, the one that’s not quite as “stuck” as the others. It’s the social butterfly of the bond world.
Next, let’s look at some of the single covalent bonds. These are the workhorses of the chemistry world. They involve the sharing of one pair of electrons between two atoms. Super common, like the handshake of the atomic world. We’ll consider a few examples, like a C-H bond (carbon-hydrogen) or an O-H bond (oxygen-hydrogen). These are pretty stable, but, you know, they’re sharing just one pair. So, there’s room for improvement, energetically speaking.
Then, we introduce the concept of multiple covalent bonds. This is where things get more exciting! Instead of just one pair of electrons being shared, atoms can decide to share two pairs (a double bond) or even three pairs (a triple bond). Imagine going from a polite handshake to a full-on hug, and then to a competitive wrestling match for electrons. The more electrons shared, the more “stickiness” you’re likely to find. Double bonds are definitely tougher than single bonds, and triple bonds? Oh boy, they’re the rock stars of covalent bonding.

We’ll be comparing a few specific examples to really nail this down. We’ll look at a few single covalent bonds, a double covalent bond, and a triple covalent bond. And to make things really interesting, we'll throw in some ionic bonds too. Because, let's be honest, ionic bonds are like the strong, silent types of the chemical world – they're all about that electrostatic attraction, like magnets going "nope, not letting go!"
The Ranking System: What Makes a Bond Strong?
So, how do scientists actually measure bond strength? It's not like they have tiny little springs they can attach to atoms. Well, sort of! They usually measure something called bond dissociation energy. This is basically the amount of energy (think calories for atoms, but way, way smaller) it takes to break a specific bond. The more energy it takes, the stronger the bond. Simple, right? It’s like measuring how much effort it takes to pry a toddler away from their favorite toy. High effort = strong attachment!
Several factors influence how strong a bond is. One of the biggies is the size of the atoms involved. Smaller atoms tend to form stronger bonds because their electron clouds are closer to the nucleus, meaning the attraction is more intense. Think of it like trying to pull two people apart who are holding hands versus two people who are holding onto each other's pinkies. The pinky grip is way easier to break!
Another crucial factor is the number of shared electron pairs in covalent bonds. As we touched upon, a single bond shares one pair, a double bond shares two, and a triple bond shares three. More shared pairs mean more electrostatic attraction holding the atoms together. It’s like adding more glue to the Lego bricks – they’re not going anywhere!

Then we have electronegativity differences. This is a fancy word for how much an atom wants to hog electrons. When atoms with very different electronegativities bond, they form a very strong ionic bond. One atom is practically stealing electrons from the other, leading to a powerful electrostatic attraction. Think of it as one extremely generous donor and one very enthusiastic receiver of electrons. They're going to be super attracted to each other!
Finally, the type of overlap between atomic orbitals matters. In covalent bonds, how the electron clouds overlap can influence the bond's strength. Better overlap generally means a stronger bond. It’s like making sure those Lego bricks are clicked together firmly, not just loosely bumping into each other.
Let’s Get This Party Started: The Order of Increasing Bond Strength!
Alright, drumroll please! We're going to take our lineup of bonds and put them in order from the weakest to the strongest. Remember, we're looking for the lowest bond dissociation energy first and working our way up to the highest.
At the very, very bottom, often considered weaker than typical covalent or ionic bonds, we have the Hydrogen Bond. While crucial for many biological processes (like holding DNA together in that famous double helix shape!), it's more of an intermolecular attraction than a true bond within a molecule. Think of it as a polite handshake or a gentle hug between molecules, easily broken. It's like the warm-up act before the main event.
Moving up, we encounter our first true covalent bonds: Single Covalent Bonds. These are generally weaker than their multiple-bond cousins. For example, let's consider a C-H bond (like in methane, CH4). These are quite common and reasonably strong, but not the strongest kid on the block. Breaking them requires a moderate amount of energy. They’re like a firm grip, but if you really try, you can get loose.

Next, we might see other single bonds, perhaps involving larger atoms or atoms with slightly different electronegativities. For instance, a C-Cl bond (carbon-chlorine) might be a bit stronger than a C-H bond, as chlorine is more electronegative and pulls electrons a bit more. But they are still in the single bond category. It's all about the subtle nuances of atomic personalities!
Now, prepare for the jump in strength! We're entering the realm of Double Covalent Bonds. A classic example is the C=C bond (carbon-carbon double bond) found in alkenes. Because there are two shared pairs of electrons, this bond is significantly stronger than a single C-C bond. Think of it as going from two people holding hands to two people in a very tight embrace. It takes more effort to break that embrace! The energy required to break a double bond is substantially higher than for a single bond.
And then, we reach the absolute pinnacle of covalent bond strength (among our common examples): the Triple Covalent Bond. The king of this category is the C≡C bond (carbon-carbon triple bond) found in alkynes, or the N≡N bond (nitrogen-nitrogen triple bond) in nitrogen gas (N2). With three shared pairs of electrons, these bonds are incredibly strong and require a massive amount of energy to break. It’s like trying to pull apart two sumo wrestlers in a serious grappling match. They are holding on with all their might! The nitrogen triple bond, in particular, is one of the strongest chemical bonds out there, which is why nitrogen gas is so stable and unreactive.
So, if we were to arrange these from weakest to strongest, it might look something like this (keeping in mind these are general trends and specific bond energies can vary slightly depending on the molecule):
Weakest to Strongest: The Grand Finale!
- Hydrogen Bond (weakest of the bunch we’re considering as a distinct interaction)
- Single Covalent Bond (e.g., C-H)
- Single Covalent Bond (e.g., C-Cl)
- Double Covalent Bond (e.g., C=C)
- Triple Covalent Bond (e.g., C≡C or N≡N) (strongest)
What about those ionic bonds I mentioned earlier? Well, ionic bonds are a bit of a different beast. They are formed by the electrostatic attraction between oppositely charged ions. The strength of an ionic bond depends on the magnitude of the charges and the distance between the ions. Some ionic bonds can be incredibly strong, often stronger than many covalent bonds. For example, the attraction between Na+ and Cl- in table salt (NaCl) is quite substantial. It’s like comparing a very strong magnetic pull to a really good velcro. They are different mechanisms of sticking, but both can be powerful!
However, when we focus specifically on the progression within covalent bonds and then compare them to the more informal hydrogen bond, the order I’ve laid out gives you a clear picture of increasing bond strength due to the increasing number of shared electron pairs. It’s like a ramp going upwards – each step up requires more energy to climb!
The Takeaway: It's All About the Stickiness!
So, what’s the big lesson here? It’s that the universe is held together by a fascinating array of connections, each with its own personality and strength. Understanding bond strength helps us predict how molecules will behave, what reactions will occur, and why things are the way they are. It’s the secret sauce behind everything from the water you drink to the air you breathe!
Think about it: the incredible stability of nitrogen gas keeps our atmosphere safe for life, while the flexibility of hydrogen bonds allows for the intricate dance of DNA. Each bond plays its part, from the gentle whispers of hydrogen bonds to the thunderous roar of a triple bond.
So, the next time you’re marveling at a chemical reaction or pondering the structure of a molecule, give a little nod to the bonds. They’re the unsung heroes, the tiny, invisible forces that make our universe so wonderfully complex and, dare I say, beautiful. Keep exploring, keep questioning, and keep smiling. The world of chemistry is full of amazing surprises, and you, my friend, are perfectly equipped to discover them!
