Arrange The Fatty Acids In Order Of Increasing Melting Point.

So, picture this: I’m in my late teens, blissfully unaware of the intricate molecular world humming just beneath the surface of everything I touched. My mission? To bake the perfect batch of chocolate chip cookies. My grandma, bless her heart, had a recipe that was legendary. It involved this secret ingredient she’d wink and nod about, which, as it turns out, was just a specific type of shortening. Anyway, I remember meticulously measuring out the ingredients, but I got a little too enthusiastic with the shortening, thinking "more fat equals more deliciousness," right? Wrong. The cookies came out… well, let's just say they had the structural integrity of a deflated balloon and tasted suspiciously like lukewarm butter. It was a culinary disaster, and the culprit? Something about how that shortening behaved at room temperature. Little did I know, I was wrestling with the very concept of melting points.
It’s funny how life throws these little lessons at you, isn’t it? You think you’re just making cookies, and suddenly you’re getting a crash course in physical chemistry. And honestly, once you start noticing it, you see these melting points everywhere. That butter on your toast? Melting point. The wax in a candle? Melting point. The asphalt on a hot summer road? Definitely a melting point (and a rather sticky one, if you’re not careful!).
But today, we’re diving a bit deeper, specifically into the fascinating, and sometimes surprisingly slippery, world of fatty acids. These are the building blocks of fats and oils, and understanding their melting points is key to understanding why some things are solid at room temperature and others are liquid. Think about it: why is olive oil liquid, but butter is… well, buttery (and often solid-ish)? It all comes down to these little molecular structures and how they decide to behave when things heat up or cool down.
The Nitty-Gritty of Fatty Acids
Alright, let’s get a little science-y for a sec, but don’t worry, we’re keeping it friendly. Fatty acids are basically long chains of carbon atoms, with a hydrogen atom attached to each carbon, except for the end one, which has a carboxyl group (COOH). Think of it like a string of pearls, but instead of pearls, it’s carbon atoms, and the string is made of chemical bonds.
Now, here’s where things get interesting. These carbon chains can be either saturated or unsaturated. This is a HUGE deal when it comes to their melting points. It’s like the difference between a perfectly straight piece of spaghetti and one that’s got a few kinks and bends in it. The kinks make a big difference in how they pack together, and that affects how much energy (heat) they need to get them moving and melt.
Saturated vs. Unsaturated: The Molecular Dance Floor
Let’s break down the key players. You’ve got your saturated fatty acids. These guys are pretty straightforward. Every carbon atom in the chain is bonded to the maximum number of hydrogen atoms it can hold. It's like a perfectly packed dance floor where everyone’s got their designated spot and there’s no room to wiggle. They tend to be straight chains.
Then you have your unsaturated fatty acids. These are the rebels of the group. They have at least one double bond between carbon atoms in the chain. This double bond creates a “kink” or a bend in the chain. Imagine our straight spaghetti again; now imagine someone’s gently twisted it in the middle. This kink means these molecules can’t pack together as tightly as their saturated cousins. They’re less orderly, more… fluid. It's like a dance floor where people are doing the cha-cha and occasionally bumping into each other. This makes them more likely to be liquid at room temperature.
Within the unsaturated category, you’ve got further subdivisions: monounsaturated (one double bond) and polyunsaturated (two or more double bonds). The more double bonds, the more kinks, the less tightly they pack, and generally, the lower the melting point. It's a cascading effect, really.
Oh, and a quick word on the cis and trans configurations of those double bonds. Most natural unsaturated fatty acids are in the 'cis' form, which creates a significant kink. 'Trans' fats, often formed through industrial processing, are more linear, making them behave more like saturated fats – hence why they're not so great for you! But that’s a whole other can of worms for another day. Let’s stick to our main mission: melting points!
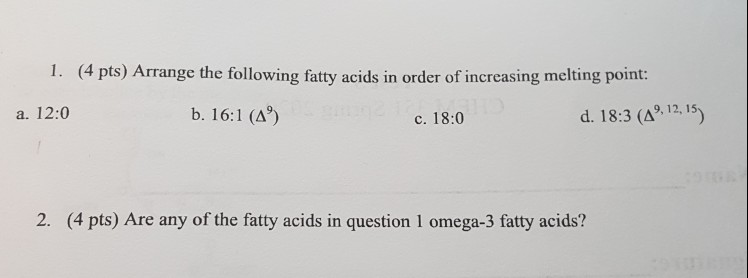
The Art of Arranging: Putting Them in Order
So, the core principle here is that the more kinks (double bonds) and the shorter the chain, the lower the melting point. Conversely, the straighter the chain (saturated) and the longer it is, the higher the melting point.
Let’s think about some common players. You’ve probably heard of many of these in your kitchen or in health food stores.
We need to arrange some fatty acids in order of increasing melting point. This means starting with the one that melts at the lowest temperature and ending with the one that melts at the highest. Imagine you’re at an ice-skating rink, and you want to arrange them from the slipperiest (lowest melting point) to the stickiest (highest melting point).
The Contenders: A Cast of Characters
Here are some fatty acids we might encounter:
- Stearic Acid: A saturated fatty acid. This is a long, straight chain, 18 carbons long (C18:0).
- Oleic Acid: A monounsaturated fatty acid. It’s also 18 carbons long, but it has one double bond (C18:1). Remember that kink!
- Linoleic Acid: A polyunsaturated fatty acid. It’s 18 carbons long and has two double bonds (C18:2). Two kinks for the price of one!
- Palmitic Acid: Another saturated fatty acid. This one is shorter than stearic acid, 16 carbons long (C16:0).
- Myristic Acid: A shorter saturated fatty acid, 14 carbons long (C14:0).
- Lauric Acid: An even shorter saturated fatty acid, 12 carbons long (C12:0).
See how we're playing with both chain length and the presence of double bonds? This is the molecular juggling act we’re doing.
The Grand Unveiling: The Order Revealed!
Let’s put them in order, from lowest melting point to highest:
1. Linoleic Acid (C18:2)
Why this one first? It has the most kinks! Two double bonds mean it's the most "bent" of our 18-carbon fatty acids. These kinks prevent the molecules from packing closely together, requiring less energy (heat) to break free and become liquid. It’s the most fluid, the least rigid. Think of it as the most eager to melt.
2. Oleic Acid (C18:1)
Next up, Oleic Acid. It’s got one double bond, giving it a kink, but it’s not as bent as Linoleic Acid. It’s still more fluid than saturated fats of the same length, but it’s a bit more ordered than Linoleic Acid. It requires a little more heat to melt than Linoleic Acid, but less than a straight, saturated chain of the same length.
3. Stearic Acid (C18:0)
Now we’re moving into the saturated zone. Stearic Acid is a long, straight chain with no kinks. Because it's saturated, the molecules can pack very tightly together. This close packing creates strong intermolecular forces, meaning it takes a significant amount of heat to get them moving and melt. It's the most ordered of the 18-carbon fatty acids and will have a higher melting point than both Oleic and Linoleic Acids.
4. Palmitic Acid (C16:0)

Here's where chain length starts to become the dominant factor among the saturated fats. Palmitic Acid is shorter than Stearic Acid (16 carbons vs. 18). Shorter chains have weaker intermolecular forces because there are fewer points of interaction. Therefore, it takes less energy to overcome these forces and melt. So, even though it's also saturated and straight, it will melt at a lower temperature than Stearic Acid.
5. Myristic Acid (C14:0)
Even shorter! Myristic Acid has 14 carbons. As we’ve seen the pattern, shorter saturated chains mean weaker intermolecular forces, and thus, a lower melting point than Palmitic Acid. It's getting easier and easier to melt!
6. Lauric Acid (C12:0)
And finally, Lauric Acid, with 12 carbons. This is our shortest saturated fatty acid in this lineup. It has the weakest intermolecular forces among the saturated fats here, and therefore, the lowest melting point of this group. It's the easiest to melt of all our contenders.
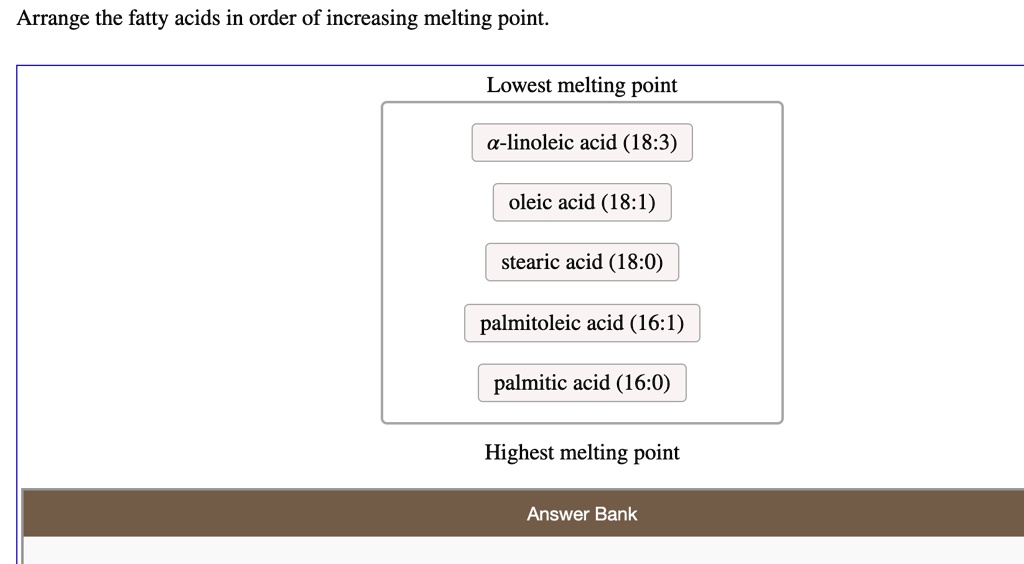
The Complete Order (Increasing Melting Point):
So, if we were to lay them out, from the easiest to melt to the hardest to melt, our lineup would look like this:
![[Solved]: Arrange the fatty acids in order of increasing me](https://media.cheggcdn.com/study/740/740c28d4-5fa5-48d9-be7b-d776a63925c8/Screenshot2025-07-17at10.50.00.png)
- Linoleic Acid (C18:2)
- Oleic Acid (C18:1)
- Stearic Acid (C18:0)
- Palmitic Acid (C16:0)
- Myristic Acid (C14:0)
- Lauric Acid (C12:0)
Pretty neat, huh? You can see how the kinks (unsaturation) lower the melting point compared to saturated fats of the same chain length, and how a shorter chain length also lowers the melting point among saturated fats. It's a delicate balance of molecular structure and how those molecules interact with each other.
Why Does This Even Matter?
Okay, so we've played chemist for a bit. Why should you, dear reader, care about the melting points of fatty acids? Well, it’s actually super relevant to your everyday life, from what you eat to how things are made!
Think about cooking. The type of fat you use affects how your food cooks and the texture it has. A fat with a low melting point, like many vegetable oils (rich in unsaturated fatty acids), will be liquid at room temperature and will melt quickly when heated. This is great for sautéing and frying.
Fats with higher melting points, like butter or lard (often with more saturated fatty acids), will be solid or semi-solid at room temperature. They contribute to the richness and texture of baked goods, like my disastrous cookies. If you use a fat that’s too hard, your baked goods can be dense. If you use one that’s too soft, they might spread out too much.
It also plays a role in food processing. Manufacturers use their knowledge of fatty acid melting points to create specific textures and shelf lives for their products. Ever wonder why some margarines are firmer than others? It’s all about the blend of fatty acids and how they've been processed!
And then there's the health aspect. We talked briefly about trans fats. Their melting point behavior (more like saturated fats) and how they interact in the body are some of the reasons they're considered unhealthy. Understanding these molecular properties helps us make better choices about the fats we consume.
So, next time you’re enjoying a slice of cheese, spreading butter on your toast, or even just looking at a bottle of cooking oil, take a moment to appreciate the invisible dance of fatty acid molecules and their fascinating melting points. It’s a small detail that has a surprisingly big impact on the world around us. And who knows, maybe it’ll even help you avoid another cookie catastrophe!
