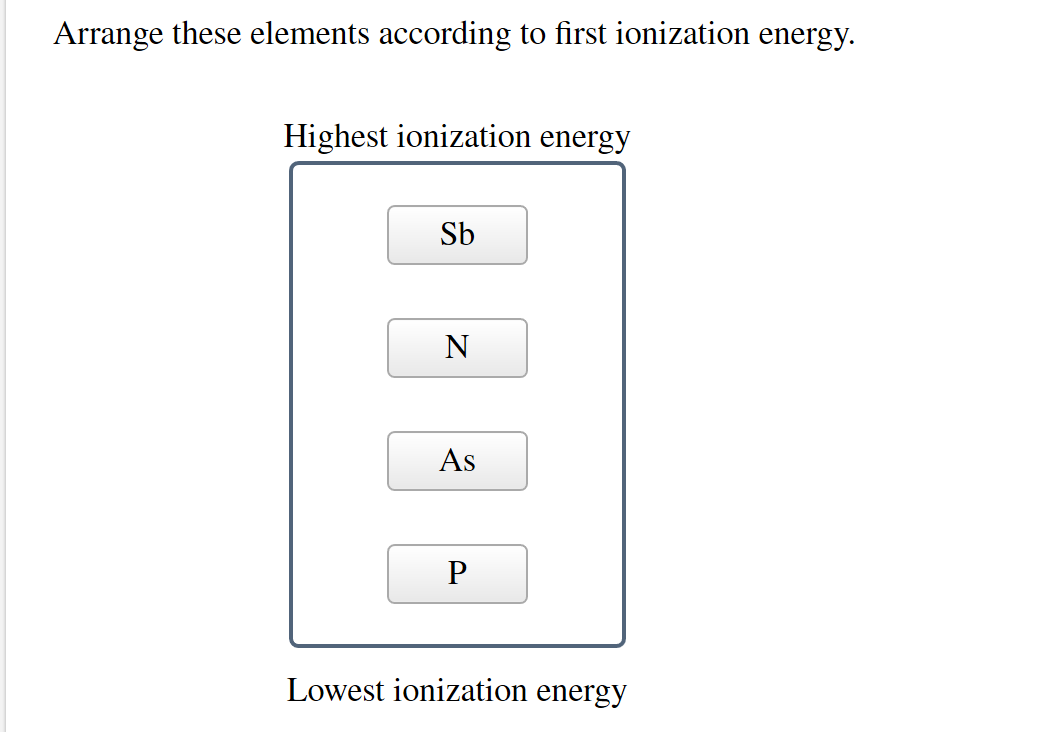
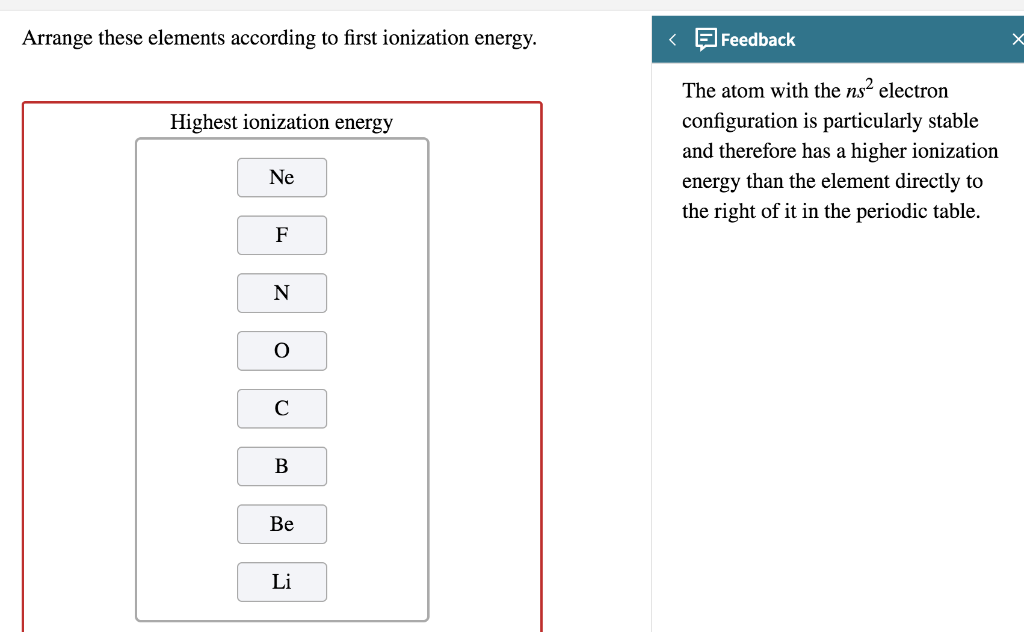
Arrange The Elements According To First Ionization Energy

Hey there, science enthusiast! So, we’re diving into the wonderfully weird world of chemistry today, and specifically, we’re gonna chat about something called first ionization energy. Sounds fancy, right? But honestly, it’s just a way of measuring how much of a pain it is to snatch an electron away from an atom. Think of it like trying to pry a stubborn kid’s toy out of their hands. Some atoms are super chill, others? Not so much!
We're talking about arranging the elements, see? Like a big, cosmic lineup. And we're lining them up based on this ionization energy thingy. It’s not just some random act, oh no. There are patterns, my friend. And that's the really cool part. It’s like solving a puzzle, but with atoms instead of cardboard bits. Wouldn't that be a weird puzzle though? Imagine trying to sort tiny little specks! The mind boggles.
So, what exactly is this first ionization energy? Picture an atom. It’s got this nucleus in the middle, right? All positive and grumpy. Then it’s got these electrons whizzing around, all negative and a little bit flighty. First ionization energy is basically the minimum amount of energy you need to zap an atom to make it lose its outermost electron. That one that’s farthest away, the one that’s maybe thinking about making a break for it anyway. It’s the easiest one to get rid of, relatively speaking, of course. Like the most independent kid in the family, always looking for an escape route.
Why do we care, you ask? Well, knowing this tells us a ton about how elements will behave. It’s like knowing someone’s personality. Are they going to be super reactive, always wanting to bond with someone else? Or are they going to be a lone wolf, happy to just chill by themselves? This little number, this ionization energy, gives us some serious clues. It’s the atom’s personality quiz score, basically.
Let’s start with the big picture. The periodic table, that magnificent chart of all known elements. It’s not just pretty colors, you know. It’s organized for a reason! And those reasons often relate back to things like ionization energy. You’ve got periods, those horizontal rows, and groups, those vertical columns. They’re like the neighborhoods and the streets of the element world. And our ionization energy has a game plan for how it plays out across this whole map.
Going Across a Period: The Trendsetters
Alright, let’s zoom in on a single period. Say, the second period: Lithium, Beryllium, Boron, Carbon, Nitrogen, Oxygen, Fluorine, Neon. What happens to the first ionization energy as we move from left to right? It generally increases. Yep, you heard it right! It gets harder and harder to snatch that electron as you go across. Why? It's all about the nuclear charge. As you move from left to right, you’re adding more protons to the nucleus. More protons mean a stronger positive pull. It’s like the nucleus is getting more and more of a superpower, and it’s holding onto its electrons like a dragon guarding its gold. Seriously, those electrons are getting seriously clung to!
Think about it. Lithium has 3 protons. Neon has 10. That’s a big jump in positive oomph! And the electrons are all in roughly the same energy shell, the second one. So, these guys on the right side of the period? They’re practically glued to the nucleus. Fluorine and Neon? They’re the super popular kids, everyone wants to be their friend, but they’re also really good at keeping their posse together. Especially Neon. It’s like the ultimate introvert, perfectly content and doesn't want anyone messing with its electron setup. It’s got a full outer shell, a complete set, and it’s not about to share. "My electrons? Nope, all mine!"
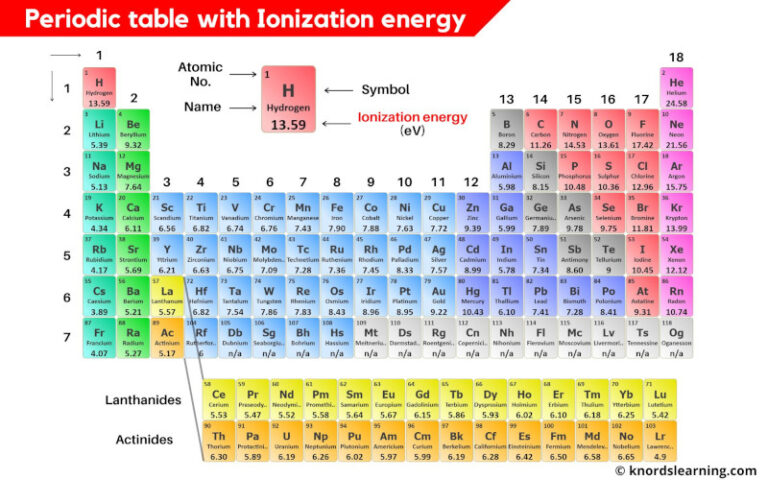
So, Lithium, being on the left, has that one extra electron in its outer shell that it’s kinda, like, "Meh, whatever, you can have it." It doesn't take much energy to get rid of it. It’s practically begging to be removed so it can get to a more stable, lower-energy state. It’s like the one friend who’s always willing to lend you a fiver. Easy peasy.
Beryllium is a little tougher. It’s got a slightly stronger pull. Then Boron… it gets a tiny bit weird here, but we’ll get to the sneaky exceptions later. Carbon, Nitrogen, Oxygen, and then Fluorine. Fluorine is practically screaming "Don't you dare touch my electrons!" It’s got the highest first ionization energy in that period. It’s the fortress of solitude for electrons. And Neon? Even higher! It’s got that perfect, happy electron configuration. It’s like a perfectly organized desk. You wouldn't go messing with that, would you? It’s just… right.
So, the general rule is: left to right across a period, ionization energy increases. Keep that in your mental toolbox. It’s a good one.
Going Down a Group: The Distant Cousins
Now, let’s shift gears and look at a group, those vertical columns. Take Group 1, the alkali metals: Lithium, Sodium, Potassium, Rubidium, Cesium, Francium. What happens to ionization energy as you go down? Surprise, surprise, it generally decreases. Lower and lower and lower. Why is this happening? It’s all about distance and shielding. As you go down a group, you’re adding new electron shells. The outermost electron is getting further and further away from the nucleus. Imagine the nucleus is like the sun. The electrons are like planets. The ones in the first shell are like Mercury, super close and feeling the heat. The ones in the seventh shell? They’re like Neptune, way out there, barely feeling the sun’s warmth.
Plus, you’ve got all these inner electrons, like little bodyguards, shielding the outer electron from the full force of the nucleus’s attraction. It’s like having a bunch of friends in between you and someone you’re trying to grab. They’re blocking the way, weakening the connection. So, even though the nucleus is getting stronger with more protons as you go down, the effect is overridden by the increased distance and shielding. It’s like trying to shout a secret to someone across a football stadium. The farther away they are, and the more people are in between, the harder it is for them to hear you.
Lithium is the feisty one in the top row. It’s got that one electron relatively close. But Sodium, the next one down? That outermost electron is in the third shell. It’s much further away. And Potassium? Even further! By the time you get to Cesium, that electron is practically waving goodbye from orbit. It takes very little energy to tell it, "See ya!" It's practically begging to be set free. It's the ultimate free spirit of the atom world.
So, the alkali metals are notorious for being super reactive. They love to lose that one electron. That’s why they’re all soft, silvery metals that tarnish quickly in air. They’re just itching to react with something! They’re the social butterflies of the periodic table, always looking for a dance partner. And the bigger they get, the easier it is for them to find one.
The general rule for groups: down a group, ionization energy decreases. Another gem for your chemistry crown!
The Sneaky Exceptions: Nature Loves a Curveball!
Now, I wouldn't be a good coffee-chat buddy if I didn't mention that chemistry is never quite as straightforward as the basic rules. Nature, bless its heart, loves to throw in a few curveballs. There are some sneaky little exceptions to these trends, and they usually pop up when we’re talking about electron configurations, specifically those filled or half-filled subshells. It’s like when your friend tells you a simple story, but then adds, "Oh, and by the way..."
Remember Boron in the second period? We said ionization energy generally increases. Well, Boron's first ionization energy is actually lower than Beryllium’s. What gives? Beryllium has a full 2s subshell (2s²). That’s a nice, stable configuration. Boron has one electron in the 2p subshell (2s²2p¹). That single electron in the 2p subshell is actually a bit easier to remove than one from a full 2s subshell, even though Boron has more protons. It's like that one single sock in a drawer full of pairs. It's not part of a cozy unit, so it's a bit more susceptible to being grabbed. The full subshell is just too darn comfortable.
Then you have the jump from Nitrogen to Oxygen. Nitrogen has a half-filled 2p subshell (2p³), which is quite stable due to electron repulsion being minimized. Oxygen, however, has one more electron in the 2p subshell (2p⁴). This extra electron has to pair up with an existing electron, creating a bit more electron-electron repulsion. This makes it slightly easier to remove one of Oxygen's outer electrons compared to Nitrogen's. It’s like having two people trying to sit in the same chair – a little awkward, right? That awkwardness makes one of them more likely to get up and leave. So, Nitrogen is like, "Nope, I'm good, happy half-full house." Oxygen is like, "Ugh, this is crowded, maybe one of us should go?"
These little bumps and dips, these deviations from the smooth trends, are what make chemistry so fascinating. They show us the subtle interplay of forces at the atomic level. It's not just brute force; it's also about how the electrons themselves are arranged and how they feel about each other. They’re like tiny little social dynamics playing out in space!
So, while the general trends are super useful for predicting behavior, always keep an eye out for these specific cases. They’re the proof that even in the seemingly rigid rules of chemistry, there’s always a bit of nuance and a lot of clever electron wrangling going on.
Putting It All Together: The Grand Arrangement
So, how do we actually arrange these elements then? We’ve got our two main rules: increases across a period, decreases down a group. That gives us a pretty good starting point. Imagine a topographical map of ionization energy. The mountains are on the right side of each period, and the valleys are at the bottom of each group.
The elements with the highest first ionization energy are generally found in the top right corner of the periodic table (excluding the noble gases, which are usually considered separately because they're so stable). Think Fluorine, Chlorine, Oxygen. These guys are electron hoarders. They hold onto their electrons with a vice-like grip. They're the ones you'd have the hardest time trying to get an electron from. They're like the most guarded secrets.
On the flip side, the elements with the lowest first ionization energy are found in the bottom left corner. Think Cesium, Francium, Potassium. These are your electron givers. They’re practically throwing electrons at you. They're the most reactive metals, always ready to form positive ions. They're the open books of the element world.
The noble gases (Group 18), like Helium, Neon, Argon, Krypton, Xenon, and Radon, are a special case. They have extremely high first ionization energies. Why? Because they have a full outer electron shell. They’re already perfectly happy and stable. They’ve achieved electron nirvana. It takes a massive amount of energy to even think about messing with their electron setup. They are the ultimate chillers, perfectly content in their electron bubbles. They’re the monks in their secluded monasteries. You don't bother them, and they don't bother anyone else. Unless you really want to spend a fortune trying to convince them otherwise, which some scientists do, leading to the formation of some very interesting noble gas compounds.
So, if you were to arrange them from lowest ionization energy to highest, you'd start with those bottom-left alkali metals and alkaline earth metals, then move across the periods and up the groups, ending with those super-stable, electron-clinging halogens and finally the incredibly reluctant noble gases. It's a beautiful dance of electron attraction and repulsion, all dictated by the number of protons, neutrons (well, not directly for ionization energy, but they affect atomic size), and the way the electrons are arranged.
It's like a puzzle where each piece (element) has a specific characteristic (ionization energy), and the periodic table is the board that shows us how they relate. Understanding these trends helps us predict how elements will react, what kinds of bonds they’ll form, and what properties they'll exhibit. It’s fundamental chemistry, and once you get it, a whole world of understanding opens up. So next time you look at the periodic table, don't just see a bunch of boxes; see a family portrait, with each member having its own unique energy signature!
And that, my friend, is the lowdown on arranging elements by their first ionization energy. It's a bit of a journey, with some predictable paths and a few surprising detours. But ultimately, it’s all about how much an atom wants to keep its electrons. Pretty cool, huh?
