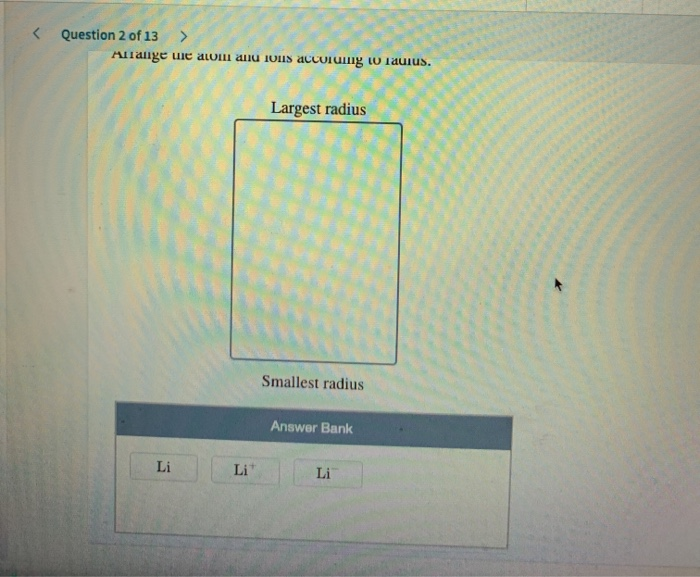
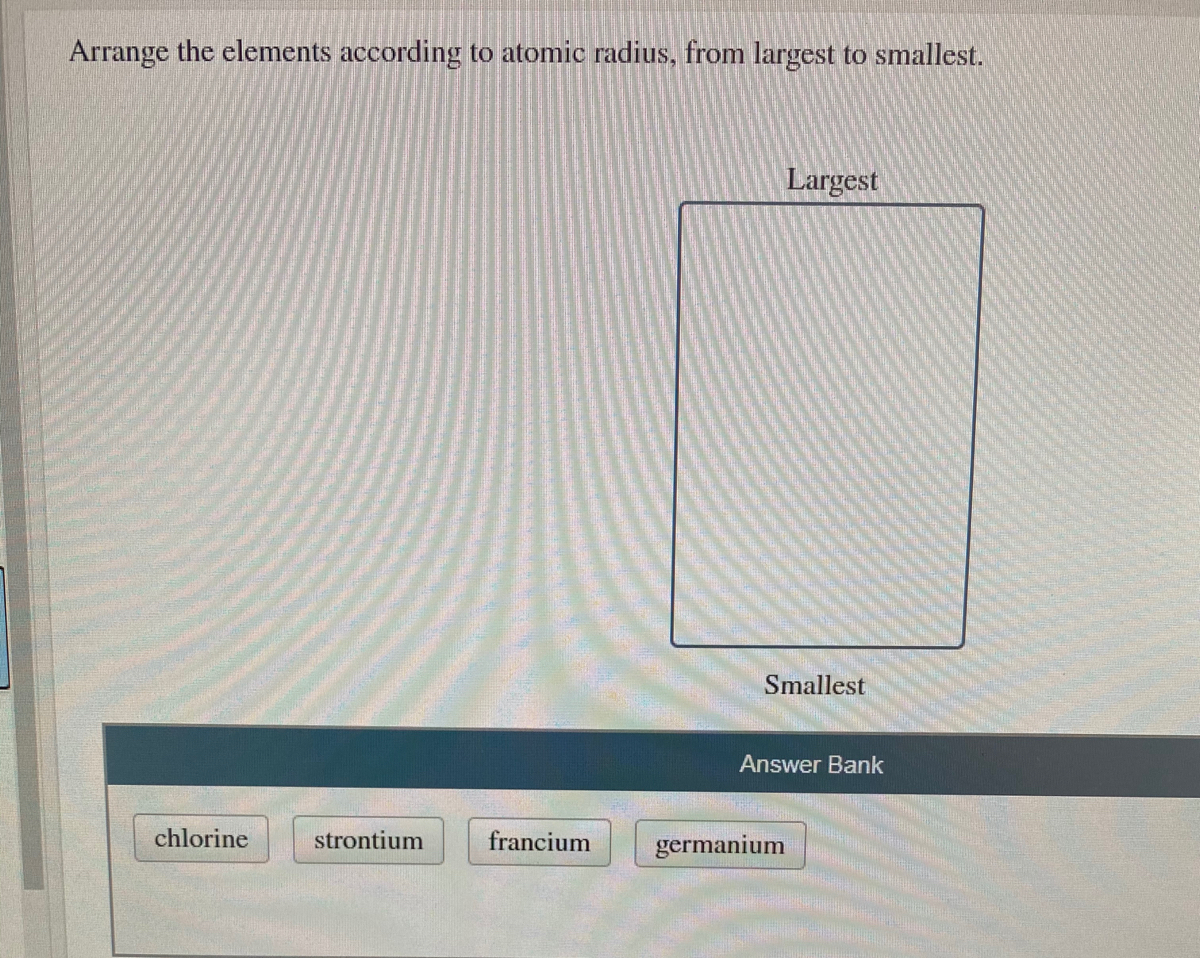
Arrange The Elements According To Atomic Radius
Hey there, science curious pals! Ever looked at a chemistry textbook and thought, "Whoa, all these element names are kinda wild"? Like, what even is an element? Think of them as the ultimate building blocks of, well, everything. You, me, your cat, that pizza you inhaled last night – all made of these tiny, invisible dudes.
But here’s where things get really interesting. These elements aren't just chilling in a giant blob. They're organized! And one of the coolest ways to sort them is by their atomic radius. Basically, it's how big an atom is. Imagine a tiny, tiny balloon. That's kind of what we're talking about.
So, how do we arrange them? It's like a cosmic game of Tetris, but with atoms. And guess what? There's a pattern. A totally predictable, super-satisfying pattern. It's like finding out your favorite socks always match or that you can, in fact, eat that entire bag of chips. Pure joy!
The Periodic Table: More Than Just a Pretty Chart
You've probably seen it. The big, colorful grid. That's the Periodic Table of Elements. It's basically the VIP list of all known elements. And it's not just for show. Every element has its own little spot, and where it sits tells you a TON about it. It's like a superhero roster, but with less spandex and more protons.
When we talk about atomic radius, we're looking at how atoms shrink and grow as you move across and down this amazing table. It’s not random, folks. It’s a whole system. A beautiful, ordered chaos.
Think of it like this: you're building a LEGO castle. You wouldn't just grab random bricks, right? You'd pick the ones that fit, the ones that make sense. The Periodic Table is like the LEGO catalog for the universe.
Going Big: The Left and Down Rule
So, ready for the big reveal? Where do you find the biggest atoms? Drumroll, please… they're usually found on the left side and towards the bottom of the Periodic Table. It's like the elemental version of "go big or go home!"

Why? It's all about the electrons. These are the little guys buzzing around the atom's core. The more electron shells (think of them as layers of an onion), the bigger the atom gets. And the further down you go, the more shells you add. Simple, right? Well, sort of. It's like adding extra rooms to your house – it just keeps getting bigger!
And on the left side? These elements are like the chill neighbors. They don't hold onto their outermost electrons that tightly. So, those electrons can spread out a bit, making the atom puff up. It’s like they're saying, "Yeah, you can borrow my electrons. No biggie."
So, if you're looking for the king-sized atoms, head to the bottom left. You'll find elements like Francium and Cesium chilling there, practically yawning with their massive atomic girth.
Shrinking Down: The Right and Up Trend
Now, what about the tiny tots? The elements that are more like sprigs than oak trees? They're over on the right side and towards the top of the table. It's the opposite vibe. Think less "king-sized" and more "snack-sized."
Why the drama? As you move from left to right, the elements are holding onto their electrons with a stronger grip. It's like they're saying, "Nope, these are my electrons, and I'm not letting them go easily!" This pull makes the atom scrunch in on itself, becoming smaller. It's the atomic version of a protective hug.
And at the very top? You've got the smallest of the small. Take Helium, for example. It's so tiny, it's practically a whisper. It's like the element that just rolled out of bed and hasn't had its coffee yet, so it's all curled up.
So, if you're ever playing "guess the element size," remember the opposite rule: right and up means small. It’s a handy trick for impressing your friends at parties. Or just for winning trivia nights. No judgment here!
The Noble Gases: The Tiny, Happy Hermits
Speaking of the top right, we have a special group: the Noble Gases. These guys, like Neon and Argon, are the ultimate homebodies. They've got a full outer shell of electrons, meaning they’re super stable and don't want to share or steal electrons from anyone. They're perfectly content in their own little world.
And because they're so stable and don't interact much, their atomic radius tends to be quite small. They're the independent artists of the element world, doing their own thing.
It's kind of funny, right? They're so reactive… by not reacting. It's the ultimate act of defiance. They're the quiet ones who secretly have all the power.
The Weird Exceptions and Quirky Details
Now, science is never that simple, is it? There are always little hiccups, little moments where you have to pause and say, "Wait a minute…" Atomic radius isn't a perfectly smooth gradient. There are some funky trends, especially when you get into the middle parts of the table.
For instance, as you go down a group (that’s a column on the table), the atomic radius almost always increases. But as you go across a period (that’s a row), it usually decreases. Usually. There are a few elements that do their own thing, just to keep us on our toes. It's like a surprise plot twist in your favorite book!
Think of the transition metals – the ones in the middle block. They can be a bit weird. Their electrons are doing some complex dances, and it can mess with the nice, clean size trend. It's like trying to herd cats, but with electrons.

And have you heard of Lanthanum and Actinium? These guys are kinda the gatekeepers to the special "lanthanide" and "actinide" series at the bottom. Their sizes are a little tricky to pin down perfectly, adding to the fun complexity of it all.
Why Should You Care About Atomic Size?
Okay, okay, I hear you. "Atomic radius, sounds important, but why should I care?" Well, my friend, it's more than just a nerdy fact for a chemistry exam. Understanding atomic radius helps us predict how elements will behave.
It affects how they bond with other elements. It influences their physical properties, like how they conduct electricity or what color they are. It's the foundation for so many chemical reactions and material science breakthroughs.
It's like knowing the personality of your elements. Are they big and generous (giving away electrons)? Or small and clingy (holding onto them tight)? This dictates how they'll play together.
So, the next time you see the Periodic Table, don't just see a bunch of boxes. See a living, breathing map of the universe. See a story of shrinking and growing, of giving and taking, all told through the sizes of atoms. It's a simple concept, but it unlocks a whole world of understanding. Pretty cool, huh?
