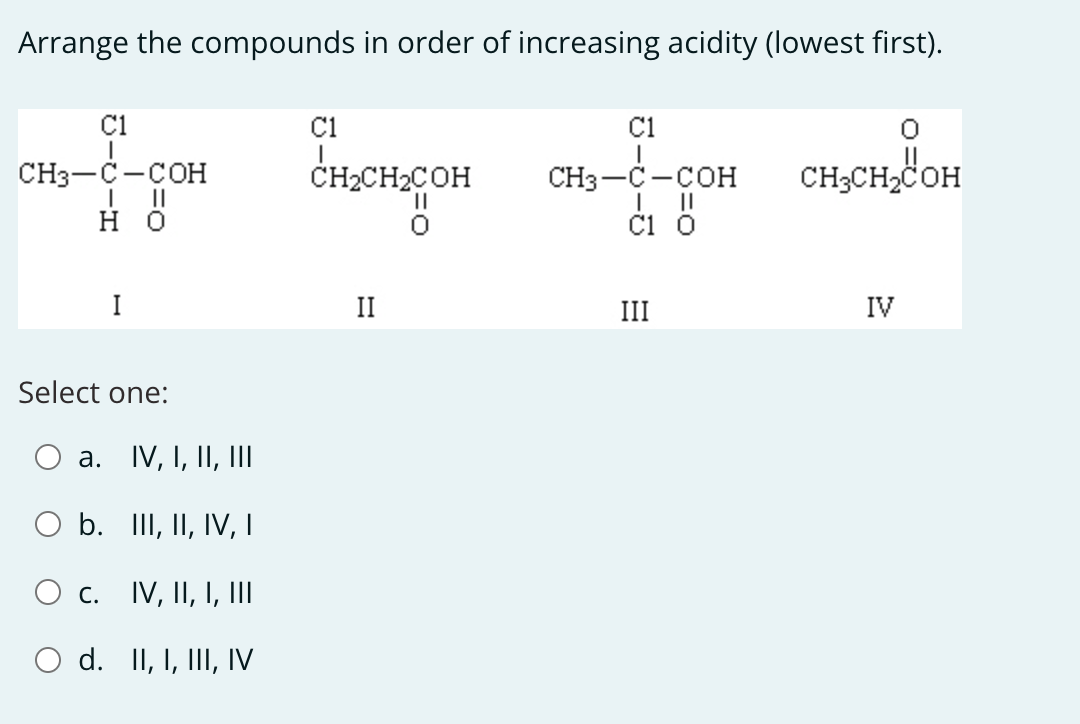
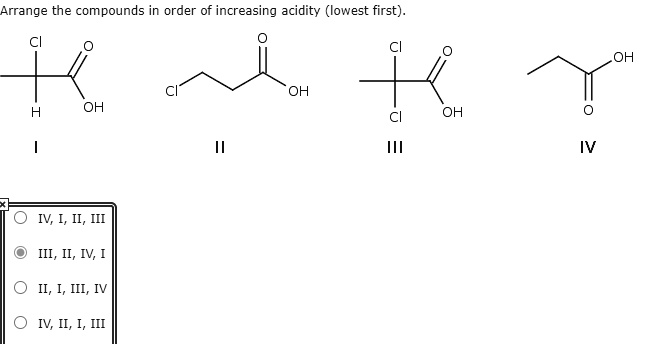
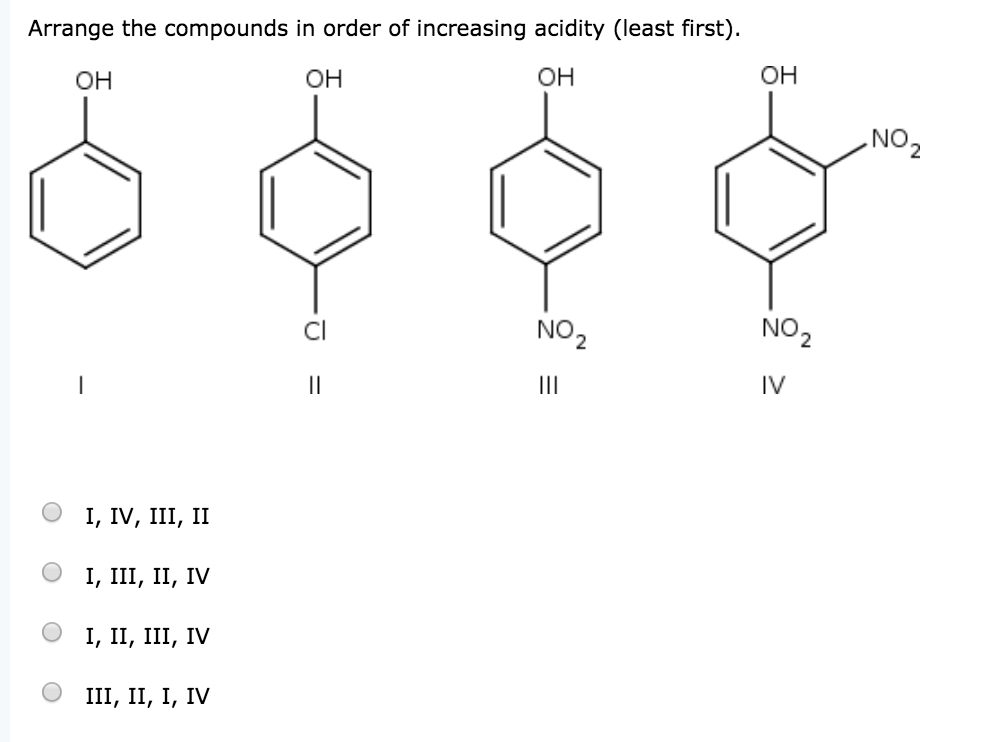
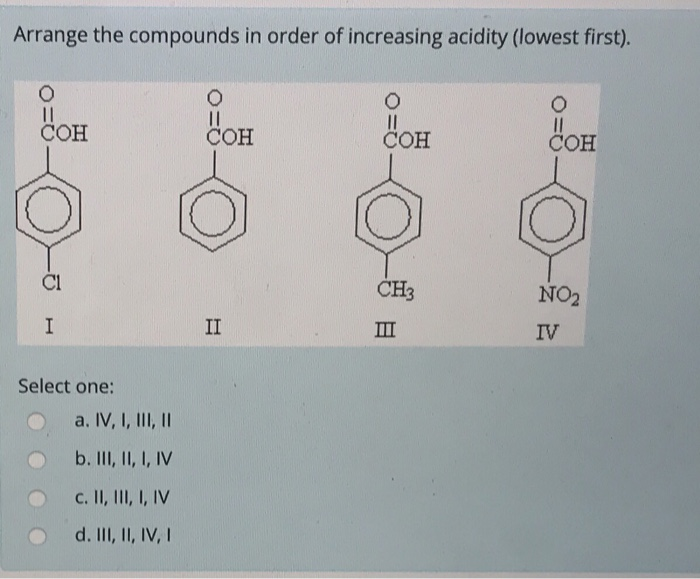
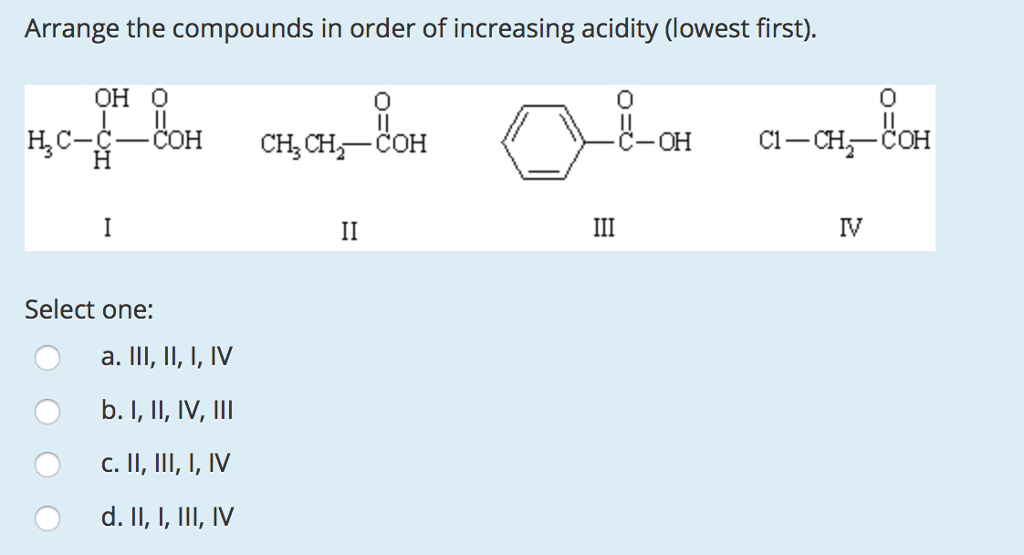
Arrange The Compounds In Order Of Increasing Acidity
So, you wanna talk about acidity, huh? Sounds kinda science-y, right? But it's actually pretty cool! Think of it like a picky eater contest for molecules. Who's the most likely to give up a proton? That's our acidic champ!
We're gonna arrange some compounds, from "meh, I guess" to "OH DEAR LORD, TAKE IT!" It's like a pH-powered rollercoaster. Buckle up!
The Acidity Adventure: A Molecular Smackdown!
Imagine you've got a bunch of molecules chilling. Acidity is basically their willingness to share. Specifically, a proton. That's a tiny, positively charged particle. Like a little hydrogen guy who just wants to bail.
The stronger the acid, the more eager that little proton is to jump ship. It's all about stability. Where does that proton go? To a lonely, electron-hungry base. They're like the molecule dating scene.
We're going to look at some common culprits. Acids, bases, and things in between. Get ready for some serious molecular drama!
Let's Meet Our Contestants!
First up, we've got good ol' water. H2O. It's like the neutral, chill friend. It can be an acid, it can be a base. It's just... there. Not super eager to donate or accept protons. Kinda the wallflower at the molecular party.
Then there's ammonia. NH3. This guy is a base. It loves to grab protons. It's the life of the party, always looking for a proton to cling to. Imagine it with little grabby hands, shouting "Mine!" at any passing proton.
Now, let's spice things up with some organic molecules. Meet ethanol. CH3CH2OH. It's got an -OH group, just like water. But is it as acidic? We'll see!
And here's where it gets interesting. We've got some carboxylic acids. Think acetic acid. CH3COOH. This is the stuff in vinegar! It's got that signature -COOH group. This one's definitely gonna be more acidic than water or ethanol. It's got a reputation!
And for our final boss? Trifluoroacetic acid. CF3COOH. Three fluorine atoms are hanging out. Fluorine is super electronegative. It's like a proton magnet! This one is going to be a beast.
The Big Reveal: Ordering the Acids!
So, how do we order them? From weakest to strongest. The ones that barely want to give up a proton to the ones practically begging to ditch it.
We start with our least acidic. Who's the most reluctant proton donor? It's our friend, the base. In this scenario, ammonia (NH3) is the clear winner for least acidic. It's a base, remember? It wants to take protons, not give them away. So, it’s at the very bottom of our acidity scale.
Next up, we have water (H2O). It's amphoteric, meaning it can act as both an acid and a base. But it's not exactly raring to go as an acid. It's more like, "Eh, maybe I'll share, maybe I won't." It's a very weak acid. So, it comes after ammonia in our order.
Now, let's look at ethanol (CH3CH2OH). It's an alcohol. Alcohols have an -OH group, just like water. But the rest of the molecule (the ethyl group) makes the proton a little more willing to leave than in plain water. It's not a huge jump, but it's there. Ethanol is slightly more acidic than water. So, it slots in after water.
Then we hit the big leagues: acetic acid (CH3COOH). This is a carboxylic acid. That -COOH group? It's designed for acidity. The oxygen atoms are good at stabilizing the negative charge that's left behind when the proton leaves. It's like a comfy couch for the leftover anion. Acetic acid is a much stronger acid than ethanol.
And our ultimate acid champion? Trifluoroacetic acid (CF3COOH). Remember those fluorine atoms? They're super greedy for electrons. They pull electron density away from the rest of the molecule, making that proton practically leap off the oxygen. It's like the proton has a jetpack and is ready for takeoff. Trifluoroacetic acid is a seriously strong acid. It's way, way more acidic than acetic acid. The fluorine atoms are like tiny, electron-stealing ninjas.
The Grand Finale: The Order Revealed!
So, put it all together! From least acidic to most acidic:
1. Ammonia (NH3): The proton-grabbing base!
2. Water (H2O): The neutral, go-with-the-flow molecule.
3. Ethanol (CH3CH2OH): A slightly more eager proton donor than water.
4. Acetic Acid (CH3COOH): The classic carboxylic acid, ready to share.
5. Trifluoroacetic Acid (CF3COOH): The electron-pulling, proton-shedding powerhouse!
Isn't that fun? It's like a little molecular popularity contest, but for giving stuff away. Who's the most generous? Who's the stingy one?
Why Is This Even A Thing?
Okay, so maybe you're thinking, "Why do I care about this?" Well, this stuff is everywhere!
From the way your stomach digests food (hello, hydrochloric acid!) to the way paints dry and medicines work, acidity plays a huge role.
Understanding this order helps chemists predict how reactions will happen. It's like knowing who's going to win a race before it even starts. Super useful!
Plus, it's just neat to think about molecules having personalities, right? Some are eager, some are shy, some are absolute drama queens.
So next time you hear about an acid, think of this little molecular lineup. Think of the protons making their great escape. It's a whole universe of tiny interactions, and it's pretty darn fascinating.
Keep exploring! There's always something new and fun to discover in the world of chemistry. Even if it sounds a bit intimidating at first, it's often just a playful dance of atoms and molecules.
