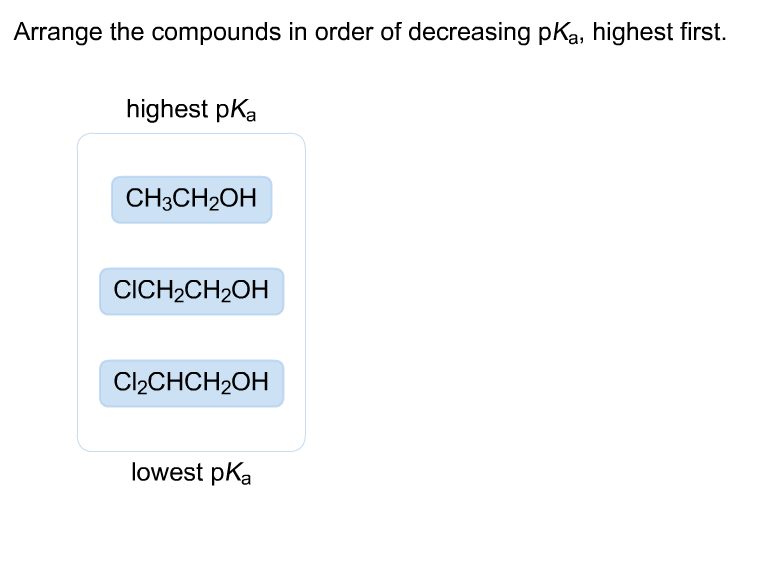
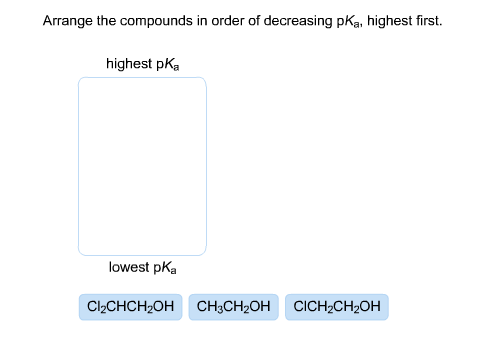
Arrange The Compounds In Order Of Decreasing Pka Highest First
Ever wondered why some things are more acidic than others? Like, why does that splash of lemon juice make your tongue tingle, but a gulp of milk just feels... milky? It all comes down to the hidden superpower of molecules: their acidity, and a handy way we scientists talk about it called pKa. Think of pKa not as a stuffy scientific term, but as a fun little ranking system, a leaderboard for how eager a compound is to give away a proton (a tiny positively charged particle, basically a hydrogen atom without its electron). The higher the pKa, the less eager it is to let go. So, if you’re curious about the world around you, from the fizz in your soda to the ingredients in your favorite face cream, understanding pKa is like unlocking a secret language of chemistry. It’s a surprisingly practical skill that pops up in all sorts of everyday scenarios, making you a bit of a chemistry whiz without even trying!
So, what's the big deal with this pKa business? Well, it’s essentially a numerical value that tells us exactly how strong an acid is. The lower the pKa, the stronger the acid. It’s like a competition, and the compounds with the lowest pKa are the champions of donating protons. This is super useful because it helps us predict how different substances will behave when mixed together. Imagine you’re cooking and you need to know if adding a certain ingredient will make your dish too sour, or if it’ll help neutralize some unwanted flavors. Or perhaps you're curious about why some cleaning products are more effective than others. That’s where pKa shines! It allows us to make informed decisions in the kitchen, in the lab, and even when choosing the right products for our homes. It’s a tool that bridges the gap between abstract chemical concepts and tangible, real-world applications. By understanding the relative strengths of acids and bases through their pKa values, we can better control reactions, optimize processes, and even develop new materials and medicines. It’s all about predicting and manipulating chemical behavior, and pKa is our secret weapon for doing just that.
Arranging the Compounds: A pKa Power Ranking!
Let's dive into the fun part: ranking some common compounds by their acidity, from the most eager proton-donator (highest pKa) to the least. Remember, a higher pKa means a weaker acid. We're going on a journey from the super-acids to the almost-neutral.
First up, leading the pack with a significantly higher pKa, we have Water. Its pKa hovers around a rather mellow 14. Compared to many other compounds, water is pretty reluctant to give up its protons. This is why water is often used as a solvent for all sorts of reactions; it’s not overly reactive itself, providing a stable environment. Think of it as the calm and steady friend in a group of energetic youngsters.
Next in line, with a slightly lower pKa, we find Ammonia (NH3). For ammonia, we're looking at a pKa in the ballpark of 33 (when considering its conjugate acid, the ammonium ion). This means ammonia is an even weaker acid than water. In fact, it's primarily known for its basic properties, readily accepting protons rather than donating them. This difference in behavior is crucial in understanding how these molecules interact in various chemical settings. Its high pKa value when acting as an acid highlights its strong tendency to act as a base.

Now, let’s bring in a compound that’s a bit more in the middle of the acidity spectrum: Ethanol. This common alcohol has a pKa of about 16. So, it's a slightly stronger acid than water, meaning it's a little more willing to part with a proton, but still far from being a strong contender. You’ll often see ethanol used in organic chemistry, and its moderate acidity makes it a versatile player in many reactions. It's like a versatile tool in a toolbox, useful for a variety of tasks without being too extreme in its properties.
Moving further down the list, we encounter Acetic Acid, the key component that gives vinegar its characteristic sourness. Acetic acid boasts a pKa of approximately 4.75. This makes it a significantly stronger acid than water, ethanol, or ammonia. Its acidity is what allows it to perform its functions, from adding tang to food to its role in various industrial processes. This is where we start to see more noticeable acidic properties in everyday life.
As we continue our descent, we meet Hydrofluoric Acid (HF). With a pKa around 3.17, hydrofluoric acid is considerably stronger than acetic acid. Despite being an acid composed of hydrogen and a halogen, it’s surprisingly weaker than other common hydrohalic acids like HCl, HBr, and HI. This is due to the strong bond between hydrogen and fluorine, which makes it less likely to dissociate and release a proton. Its strength lies in its ability to form strong hydrogen bonds, which influences its reactivity and physical properties. It’s a good example of how seemingly small differences in molecular structure can lead to significant changes in acidity.
Finally, at the very bottom of our pKa ranking, meaning it's the strongest acid on our list, we have Hydrochloric Acid (HCl). Its pKa is a staggeringly low number, often cited as around -6 or even lower! This means HCl is incredibly eager to donate its proton. It’s a powerful acid used in everything from industrial cleaning to the digestive processes in our stomachs. Its low pKa signifies a near-complete dissociation in water, making it a potent force in chemical reactions.
So, to recap our fun little power ranking, from highest pKa (weakest acid) to lowest pKa (strongest acid), the order is:
Ammonia (acting as an acid, conjugate acid pKa ~33) > Water (pKa ~14) > Ethanol (pKa ~16) > Acetic Acid (pKa ~4.75) > Hydrofluoric Acid (pKa ~3.17) > Hydrochloric Acid (pKa ~-6)
It's fascinating how these numbers can paint such a clear picture of a compound's chemical personality! This ranking helps us understand why certain substances react the way they do and allows us to predict their behavior in different situations. It’s a testament to the elegance and predictability of chemistry!
