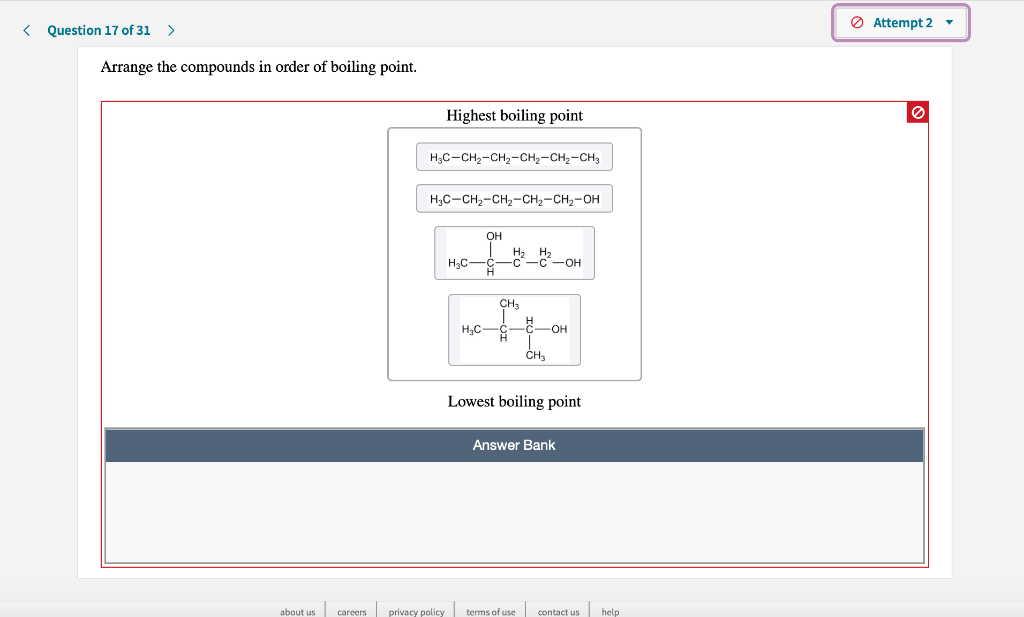
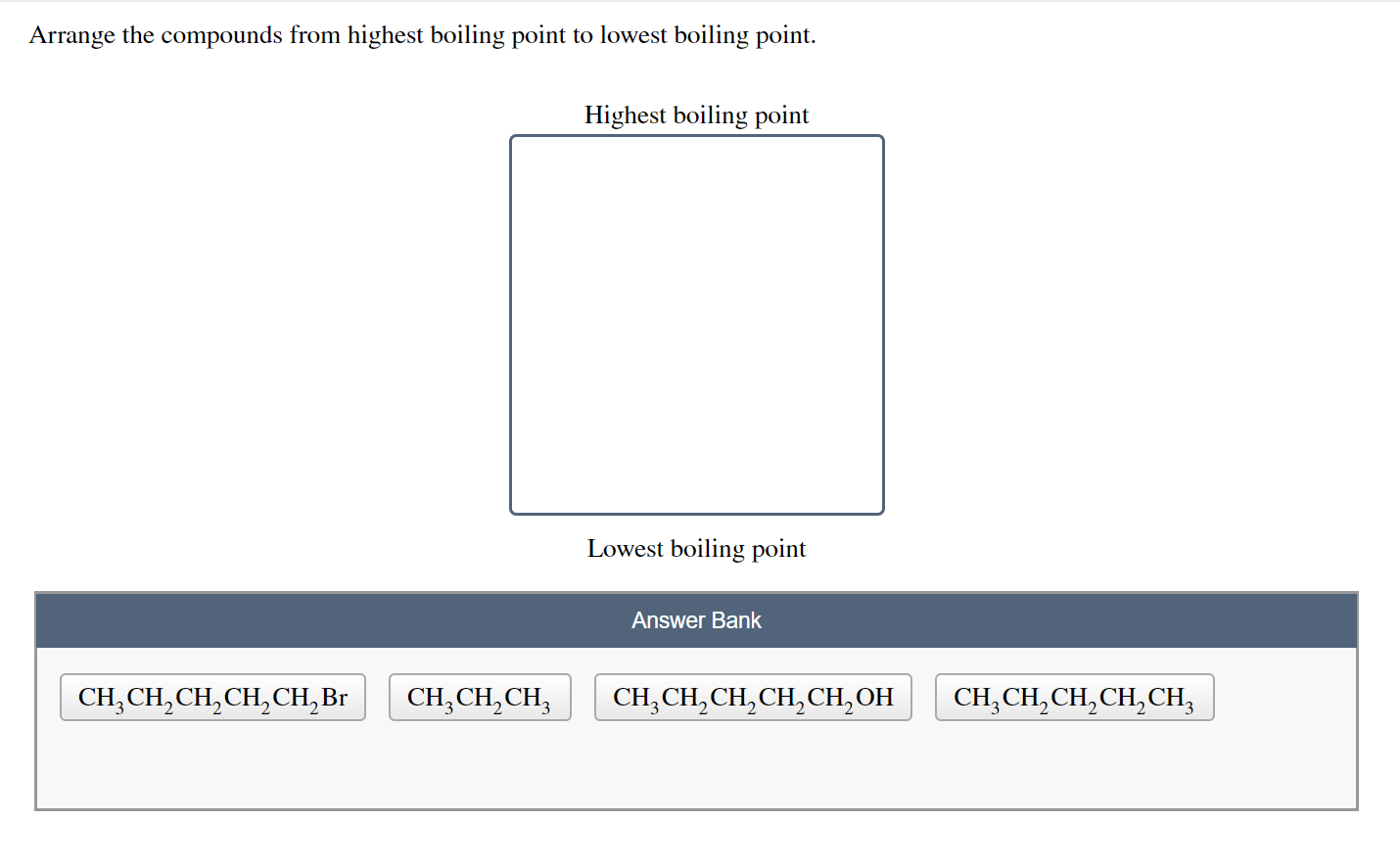
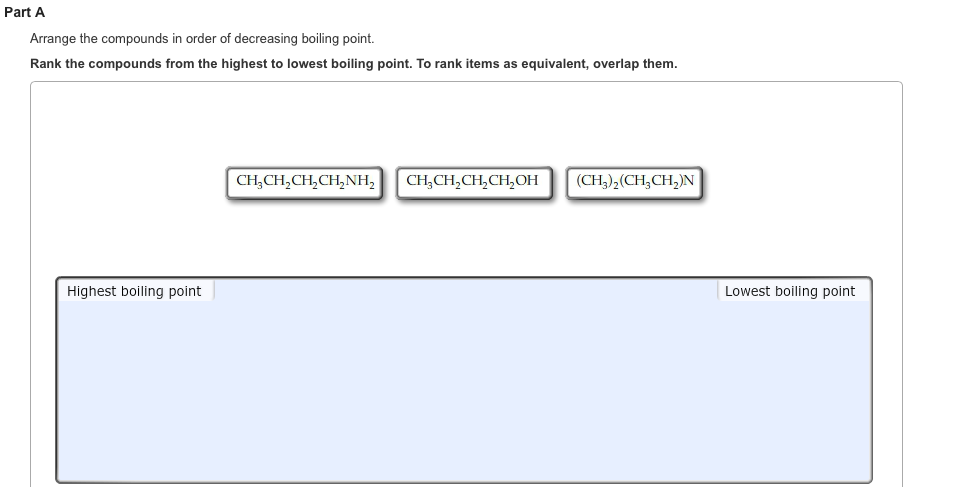
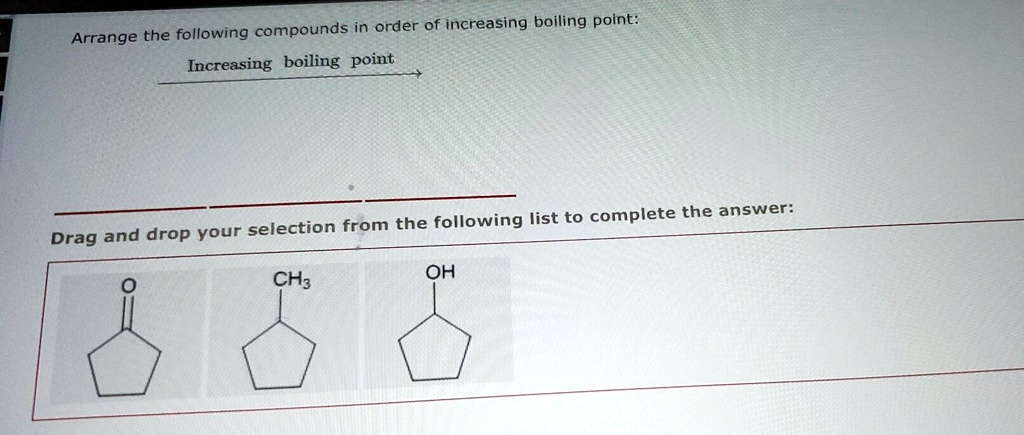
Arrange The Compounds In Order Of Boiling Point
Alright, my fellow kitchen adventurers and curious minds, gather 'round! Today, we're embarking on a super-duper, fun-tastic journey into the wacky world of… boiling points! Now, I know what you might be thinking, "Boiling points? Sounds a bit… science-y." But trust me, it's way more exciting than it sounds, like a secret recipe for how hot things have to get before they start doing their little bubbly dance!
Imagine your favorite drinks. You’ve got that refreshing glass of water, right? It’s the champ of everyday hydration. Now, think about that super-sticky, sweet syrup you drizzle on your pancakes. It’s thick, it’s luscious, and it definitely doesn’t get as excited about heat as plain old water does. And then, let’s throw in something really special, like a dash of pure, unadulterated ethanol, the stuff you might find in, say, a fancy vanilla extract or… well, other grown-up things. These guys all have their own unique personalities when it comes to heat, and it’s our mission today to line them up in a super-organized, totally awesome order based on how much energy they need to get their boil on!
So, here’s the deal. We’ve got our contenders, all lined up like little soldiers ready for a heat wave. We're going to arrange them from the one that’s the most chilled-out, the easiest to get bubbling, all the way up to the real trooper, the one that stubbornly refuses to boil until it’s practically begging for mercy in a hot tub!
First up, the super-star of simplicity, the king of casual boiling: Water! Yep, good old H2O. It’s like that friend who’s always up for anything, needing just a little nudge to get going. At a cozy 100 degrees Celsius (or 212 degrees Fahrenheit for my Fahrenheit-loving pals), it’s ready to start its magnificent transformation into steam. Think about boiling an egg or making a cup of tea – that’s water doing its thing, happy as can be at its boiling point.
Next in line, we have a slightly more sophisticated character, a bit more hesitant to jump into the boiling fray: Ethanol. This is where things start getting a tad more interesting. Ethanol is a little more… clingy. Its molecules like to hold onto each other a bit tighter than water molecules do. It's like they're having a private conversation and don't want to be disturbed by the boiling party! So, it needs a bit more oomph, a little extra heat to break free. Its boiling point is a touch lower than water, around 78.37 degrees Celsius (173.07 degrees Fahrenheit). It’s still relatively easy-going, but it’s definitely got a bit more of an “I’m not quite ready yet” vibe.
Imagine water is your best friend who’s always ready for an impromptu road trip. Ethanol is your other friend who needs a gentle reminder and a quick snack before they’re ready to hit the road.
And now, for the grand finale, the ultimate heat-seeker, the stubbornly serene: Syrup! Or, to be more precise, let's think about something like concentrated sugar solutions, which is what syrup essentially is. These sugary concoctions are like the VIPs of the boiling world. They’ve got a whole bunch of sugar molecules hanging out, getting all cozy and interlocked with water molecules. They’re like a giant, sticky hug that’s incredibly difficult to break apart! To get these sweet, thick beauties to even think about boiling, you need to crank up the heat considerably. Their boiling points are significantly higher than both water and ethanol, often well above 100 degrees Celsius, depending on how much sugar is packed in there. Think of making candy or jam – you're essentially cooking these sugary mixtures to much higher temperatures to get them to achieve that desired consistency!
So, to recap our thrilling boiling point adventure, let’s get them all lined up in order, from the easiest to get bubbling to the most stubbornly resistant:
First, the laid-back legend: Water! It’s the champion of quick boils, the everyday hero.
Then, a little more reserved, but still willing to party with a bit of persuasion: Ethanol! It’s got a slightly more exclusive guest list for its boiling bash.
And finally, the ultimate challenge, the diva of high temperatures: Syrup (or concentrated sugar solutions)! This one needs a serious heat wave to get its groove on.
It’s like a heat race, and we’ve got our winners! From the casual simmer of water to the determined boil of syrup, each compound has its own unique temperature where it decides to get all steamy and exciting. Isn't that just the coolest? You’ve just unlocked a little piece of the universe’s secret language, all thanks to the power of boiling points! Keep that curiosity bubbling, folks!
