Arrange The Bonds In Order Of Increasing Polarity

Let's talk about something that might sound a little bit… intense. We're diving into the wild world of chemical bonds! Now, before you picture lab coats and bubbling beakers, think of it more like a family reunion. Some family members are super chill, others have a bit more… spark. And that's what we're exploring today: how much "spark" different bonds have, or as the fancy folks say, their polarity. It's a bit like a popularity contest, but for atoms!
So, imagine we have a bunch of these tiny little atomic buddies, all wanting to hold hands. They can do this in a few ways. The most basic kind of hold is when they're sharing their hands pretty evenly. Think of two best friends sharing a cookie. It’s equal. It’s friendly. This is like our starting point, the nonpolar covalent bond. Everyone’s happy. No drama. Just good vibes and shared cookies. It’s the ultimate in atomic chill.
Then, things get a tiny bit interesting. Sometimes, one atom is a bit more of a "grabber" than the other. It's not a full-on snatch, mind you. It's more like… one person really likes the last bite of pizza. They don't steal it, but they might nudge the plate a bit closer. This is where we get the polar covalent bond. One atom gets a little more of the "electron cloud" – think of it as getting a slightly bigger slice of the cookie. It’s still sharing, but with a hint of favoritism. This is where the first little bit of "spark" starts. It's like that one cousin who always gets the biggest slice of birthday cake, and everyone else just sighs a little.
Now, if we crank that up a notch, we're talking about a bond where one atom is practically daring the other to try and take its electrons. It's not a fight, but it's definitely a strong pull. This is when we start to see something called an ionic bond. It's like one friend offers you a cookie, but then immediately asks for it back, but in a really insistent way. One atom basically gives an electron to another. It's a bit dramatic, a bit like a cliffhanger in a soap opera. One atom becomes a little bit positive (it lost something), and the other becomes a little bit negative (it gained something). They’re not sharing anymore; they’re essentially stuck together because one is saying "take it!" and the other is saying "fine, I'll take it, but you owe me." It’s the atomic equivalent of a dramatic exchange of borrowed items.
So, let's put these guys in order, from least dramatic to most dramatic. Imagine you're at a party. First, you've got the folks just casually chatting, perfectly balanced. That's our nonpolar covalent bond. No one’s hogging the snacks, no one’s monopolizing the conversation. Everyone’s just vibing.

Then, you've got the people who are really interested in a topic. They're leaning in, their hands are gesturing wildly, and you can tell they're putting a lot of energy into this conversation. That's your polar covalent bond. There's a definite pull, a focus of attention. One side of the conversation is a little more intense than the other, but they're still in the same general area, still participating. It’s like when your friend explains their new obsession with competitive dog grooming. You’re listening, but maybe not as invested.
And finally, you have the people who are having a full-blown, passionate debate. They're not necessarily yelling, but the energy is palpable. You can feel the tension in the air, the strong forces pulling them towards agreement or disagreement. This is your ionic bond. One person is absolutely convinced, the other is completely swayed (or repelled!). It's a full transfer of energy, a definitive win or loss in the exchange of ideas… or in this case, electrons. It’s the ultimate "I'm taking this and you're not getting it back" moment in the atomic world.

It’s fascinating, isn't it? How these tiny, invisible forces dictate so much about how things work. From the water we drink to the air we breathe, these bonds are like the invisible glue holding the universe together. And while the science behind it can get complicated, the basic idea of sharing versus not-so-sharing is something we can all understand. It’s the atomic version of that awkward moment when you realize your friend really wants your fries, and you're not sure whether to offer them or guard them with your life. The nonpolar covalent bond is the "here, have a fry." The polar covalent bond is the "I'll let you have one, but just one, and I'm watching you." And the ionic bond? Well, that’s probably when you’ve already eaten all the fries, and your friend is just staring at your empty wrapper with a look that says, "You owe me big time."
So, next time you hear about these bonds, don't feel intimidated. Think of it as a quirky family gathering of atoms, each with their own personality and their own way of interacting. Some are perfectly content to coexist, others have a little more flair, and some are just all about that dramatic exchange. And honestly, isn't that kind of relatable? We all know people who are like each of these bonds, right? It's just that these atoms do it on a microscopic level, and it's actually quite beautiful in its own way.
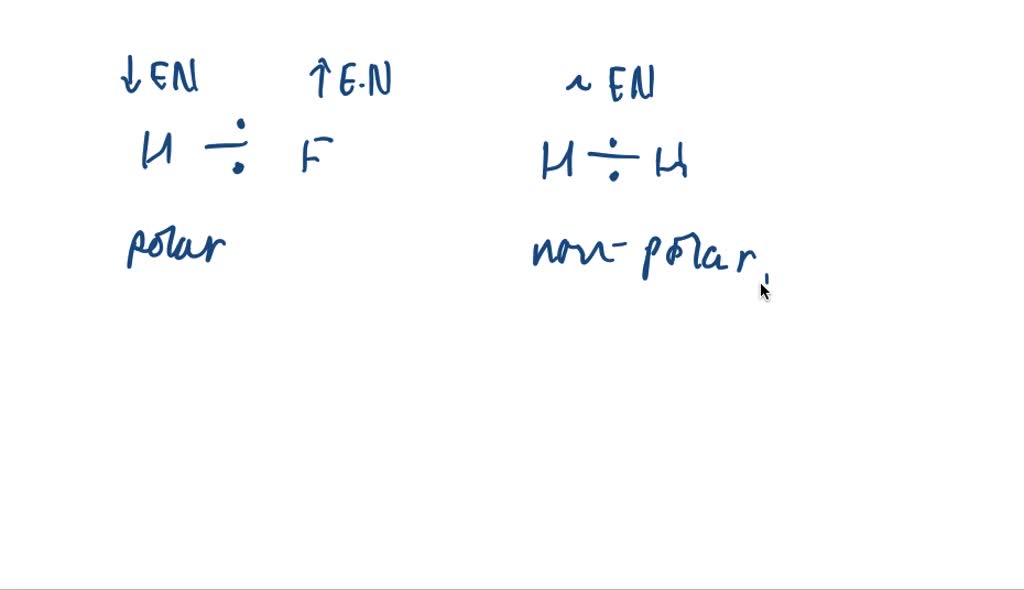
So, in order of increasing polarity, we have the chillest of the chill, the nonpolar covalent bond, followed by the slightly more intense, the polar covalent bond, and finally, the one that really grabs your attention, the ionic bond. It's like a spectrum of atomic enthusiasm!
It’s not about who’s "better," mind you. It’s just about their different styles. The nonpolar folks are the ultimate diplomats. The polar ones are the passionate conversationalists. And the ionic ones? They're the ones who make things happen through their intense interactions. And the universe, in its infinite wisdom, seems to appreciate all of them. Who knew atoms could be so much like us?
