Arrange The Acids According To Their Expected 𝐾a Values.
So, picture this: I'm in my first year of university, buried under a mountain of textbooks, feeling like my brain was about to spontaneously combust. Organic chemistry. The bane of many a student's existence, right? We were learning about acids and bases, and my professor, a brilliant but slightly eccentric woman with a penchant for dramatic pronouncements, tossed this seemingly simple question at us: "Arrange these acids from strongest to weakest." Simple, she said. My internal monologue was screaming, "Simple?! Are you kidding me?!" My classmates and I exchanged panicked glances, a silent SOS passing between us. We were supposed to have memorized all the acids? And their strengths? This felt like a cruel prank.
But then, a few of us started to whisper, to tentatively offer up some ideas, and slowly, a pattern emerged. It wasn't about memorization, not entirely. It was about understanding, about a kind of chemical intuition. And that, my friends, is what we're going to dive into today. We're going to unlock the secrets of predicting acid strength, of arranging those pesky acids according to their expected Ka values. No more panic, just pure, unadulterated chemical curiosity. Well, maybe a little bit of panic is good for the soul, keeps you on your toes!
Let's get one thing straight right off the bat: Ka. What is it? It's the acid dissociation constant. Think of it as a score for how eager an acid is to dissociate, to break apart and donate its proton (that's the H+ you're always hearing about). A bigger Ka means the acid is a stronger acid. It means it’s really good at letting go of its H+. A smaller Ka means it's a weaker acid, holding onto its proton for dear life. Makes sense, right? It’s like a personality quiz for molecules!
So, how do we predict this magical Ka value without actually doing a million experiments? We look at the structure of the acid. And not just the atoms, but how they're arranged, how they're bonded. This is where things get really interesting, and honestly, pretty powerful. We're essentially learning to read the molecule's mind.
The Big Picture: What Makes an Acid Acidic?
At its core, an acid is a proton donor. When an acid (let's call it HA) dissolves in water, it can react like this: HA + H₂O ⇌ H₃O+ + A-. The Ka is essentially the equilibrium constant for this reaction. If the reaction strongly favors the right side (lots of H₃O+ and A-), then HA is a strong acid, and Ka will be large. If it favors the left side (lots of undissociated HA), it's a weak acid, and Ka will be small.
So, we're looking for factors that make that A- ion (the conjugate base) more stable. Why? Because if the conjugate base is happy and stable on its own, it's much more likely to let go of its proton in the first place. It’s like a parent whose kid has a fantastic job and a stable life – they’re much more willing to let them move out and be independent. A parent whose kid is still living in their basement playing video games? Maybe not so much letting go!
Factor 1: Electronegativity – The "Pull" Power
This is one of the most fundamental factors. Electronegativity is basically an atom's greed for electrons. In an acid like HX, the strength of the H-X bond and the stability of the X- anion are key. If X is highly electronegative, it can pull electron density towards itself. This has two main effects:
- Weakens the H-X bond: With X hogging electrons, the bond holding onto the H is stretched thin, making it easier to break. Think of it like pulling on a rubber band; the more it stretches, the easier it is to snap.
- Stabilizes the X- anion: Once the proton leaves, the negative charge on the X- ion is spread out and better accommodated by the electronegative atom. It’s like a big person can handle a heavier load more easily than a small person.
So, if you compare acids like HF, HCl, HBr, and HI, what do you notice? Fluorine (F) is the most electronegative, followed by chlorine (Cl), then bromine (Br), and finally iodine (I). Therefore, HF should be the strongest acid according to this rule, right? Wrong! Wait, what?! This is where it gets tricky, and why we need multiple factors. While electronegativity is important, it's not the whole story, especially down a group in the periodic table.
This is where I always remember those moments in class where you think you've got it, and then... plot twist! Electronegativity plays a HUGE role when comparing atoms in the same period of the periodic table. For example, comparing CH₄ (methane), NH₃ (ammonia), H₂O (water), and HF (hydrogen fluoride). Carbon is less electronegative than nitrogen, which is less electronegative than oxygen, which is less electronegative than fluorine. So, the acidity increases in that order: CH₄ < NH₃ < H₂O < HF. The more electronegative the atom bonded to hydrogen, the more stable the conjugate base, and thus, the stronger the acid. Simple enough for now, eh?
Factor 2: Atomic Size – The "Spread Out" Effect
Now, let's go back to that HF, HCl, HBr, HI series. If electronegativity doesn't fully explain it, what else is going on? Atomic size. As you go down a group in the periodic table, the atoms get bigger. Iodine is much, much larger than fluorine.
How does size help? A larger atom can spread out the negative charge of the conjugate base over a bigger area. Imagine a tiny balloon versus a giant beach ball. If you put the same amount of "charge" (like a drop of paint) on each, it will be much more concentrated on the tiny balloon than spread out on the beach ball. A spread-out charge is a more stable charge. Therefore, the iodide ion (I-) is much more stable than the fluoride ion (F-). This increased stability of the conjugate base means that HI is actually the strongest acid in this series, and HF is the weakest! Mind. Blown. Again.
So, when comparing acids where the acidic proton is attached to atoms in the same group, atomic size is the dominant factor. Bigger atom = more stable conjugate base = stronger acid. This explains why HI > HBr > HCl > HF. It's counter-intuitive at first glance, but it totally makes sense once you visualize that charge spreading out!
Factor 3: Resonance – The "Delocalization Dance"
This one is a bit more advanced but super important. Resonance happens when a negative charge (or a positive charge, or a double bond) isn't localized on a single atom but can be shared among multiple atoms through delocalization. Think of it as musical chairs for electrons. If the electron has more chairs to choose from, it’s a lot less likely to get stuck in one spot. And a delocalized charge is a happy, stable charge.
Let's take carboxylic acids, like acetic acid (CH₃COOH) and formic acid (HCOOH). When acetic acid loses a proton, it forms the acetate ion (CH₃COO-). This negative charge can be delocalized between the two oxygen atoms through resonance. Because the charge is spread out over two oxygens, the acetate ion is quite stable. This makes acetic acid a relatively strong weak acid.
Now, consider something like phenol (C₆H₅OH). When it loses a proton, it forms the phenoxide ion. The negative charge on the oxygen can be delocalized into the benzene ring. This resonance stabilization makes phenol a much stronger acid than a simple alcohol like ethanol, where there's no such resonance to stabilize the conjugate base.
Think of it like this: if you have a lot of friends who can help you move, it's going to be a lot easier than if you have to do it all by yourself. Resonance is like having a whole crew of friends helping to carry that negative charge!
Factor 4: Inductive Effect – The "Electron-Pulling Neighbor"
This is where substituents on the molecule come into play. An inductive effect is the pulling or pushing of electron density through sigma bonds. This is often associated with electronegativity, but it's about the effect of neighboring atoms or groups on the acidity.
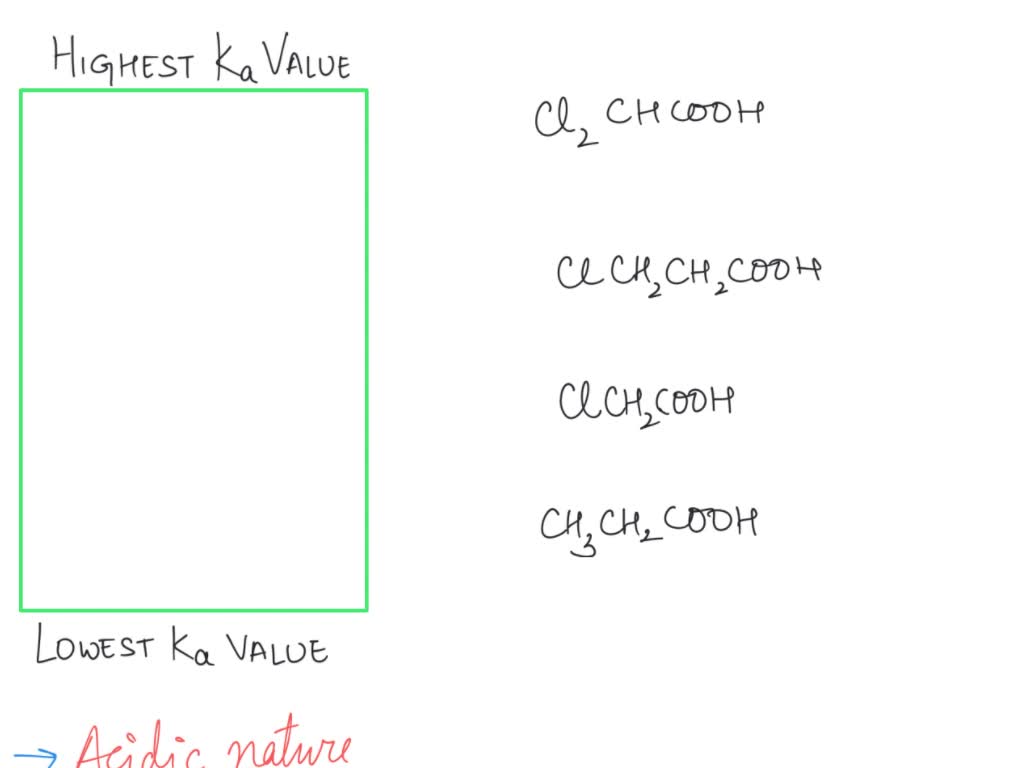
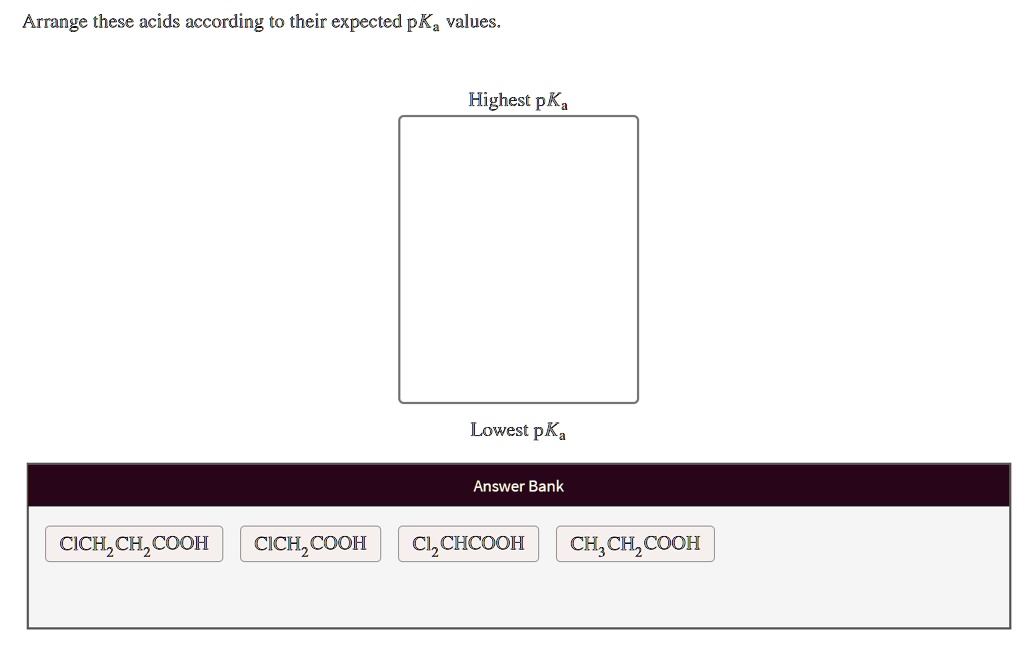
Electronegative atoms or groups tend to pull electron density towards themselves. This is called a negative inductive effect (-I effect). For example, if you have chlorine atoms attached to the carbon next to the carboxylic acid group in chloroacetic acid (ClCH₂COOH) compared to acetic acid (CH₃COOH):
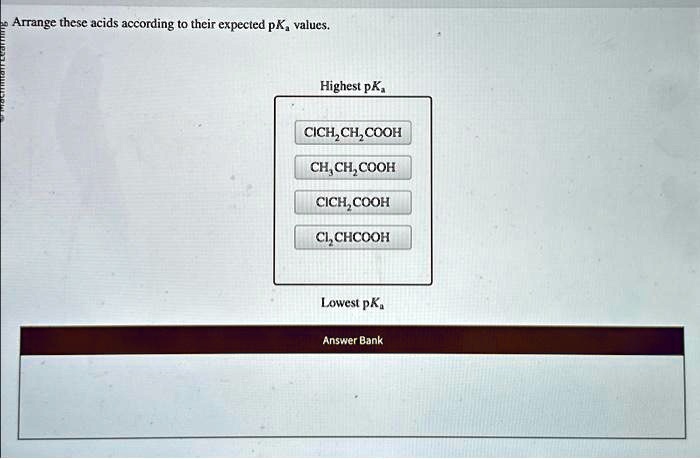
- The chlorine atoms are electronegative and pull electron density away from the carboxylic acid group.
- This withdrawal of electron density makes the O-H bond even more polarized and weaker.
- Crucially, it also helps to stabilize the negative charge on the conjugate base (carboxylate ion) by pulling it away.
So, chloroacetic acid is a stronger acid than acetic acid. The more electronegative atoms you add, and the closer they are to the acidic proton, the stronger the acid becomes. Imagine that chlorine atom as a tiny, incredibly strong vacuum cleaner for electrons, sucking them away from the acidic proton and making it easier for it to leave.
Conversely, electron-donating groups (positive inductive effect, +I effect) would destabilize the conjugate base and make the acid weaker. For example, alkyl groups like methyl (-CH₃) are slightly electron-donating. This is why acetic acid is weaker than formic acid (where there's no methyl group, just a hydrogen).
Putting It All Together: The Grand Arrangement
Now, let's put this knowledge to the test. Imagine you're given a list of acids and asked to arrange them. You need to consider these factors in a sort of hierarchy:
- Same Period: Electronegativity of the atom bonded to H is usually dominant.
- Same Group: Atomic size of the atom bonded to H is usually dominant.
- Resonance: If resonance stabilization is present in the conjugate base, it significantly increases acidity.
- Inductive Effects: Electronegative substituents near the acidic site increase acidity.
It's not always perfectly clear-cut, and sometimes you have to weigh the different factors. For instance, a very strong inductive effect might overcome a slight difference in atomic size. But these rules give you a really solid framework for making educated guesses.
A Practical Example: Let's Play "Acid Detective"
Okay, let's say we have these acids to arrange:

- Ethanol (CH₃CH₂OH)
- Acetic Acid (CH₃COOH)
- Water (H₂O)
- Phenol (C₆H₅OH)
- Formic Acid (HCOOH)
Let's break them down:
- Ethanol: The O-H bond is polarized by oxygen, but the conjugate base (ethoxide ion, CH₃CH₂O-) has a localized negative charge. No resonance, weak inductive effect from the ethyl group. It's a weak acid.
- Water: Similar to ethanol, but the conjugate base (hydroxide ion, OH-) has a localized charge. It's a benchmark for "weak."
- Formic Acid: This is a carboxylic acid (HCOOH). When it deprotonates, it forms the formate ion (HCOO-). The negative charge is delocalized over two oxygen atoms via resonance. This makes it much more acidic than alcohols or water.
- Acetic Acid: This is also a carboxylic acid (CH₃COOH), forming the acetate ion (CH₃COO-). It also has resonance stabilization of the conjugate base, similar to formic acid. However, the methyl group (CH₃) is slightly electron-donating, which will destabilize the negative charge a tiny bit compared to formic acid. So, acetic acid should be a bit weaker than formic acid.
- Phenol: This is where resonance really shines! The phenoxide ion (C₆H₅O-) has its negative charge delocalized into the benzene ring. This is a very effective way to stabilize the charge. This will make phenol significantly more acidic than ethanol and generally more acidic than simple carboxylic acids like acetic and formic acid (though the comparison between phenol and very strong carboxylic acids can be nuanced and depend on specific conditions and substituent effects).
So, our expected order from strongest to weakest Ka would be something like:
Phenol > Formic Acid > Acetic Acid > Water ≈ Ethanol
(Note: the exact Ka values can sometimes be tricky, and the difference between water and ethanol's acidity is often debated in introductory contexts, but this gives you the general idea based on the major stabilizing factors).
The Bottom Line (and a Wink to Your Professor)
Learning to predict acid strength isn't just about memorizing numbers; it's about understanding how molecular structure dictates chemical behavior. It's a powerful skill that will serve you well, whether you're trying to figure out how a drug works or why your chemistry experiment went spectacularly wrong (or right!).
So, next time you're faced with a list of acids and a daunting task to order them, remember the magic trio: electronegativity, atomic size, resonance, and the sneaky inductive effect. They're your secret weapons. And maybe, just maybe, you can even impress your professor with your newfound predictive powers. Just don't tell them I told you it was more about understanding than pure memorization. That's our little secret. Happy acid arranging!
