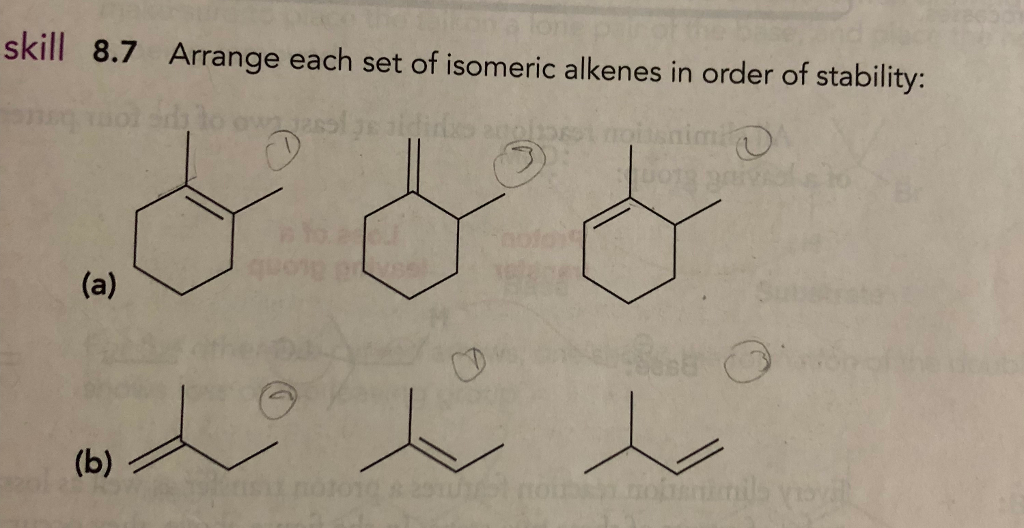
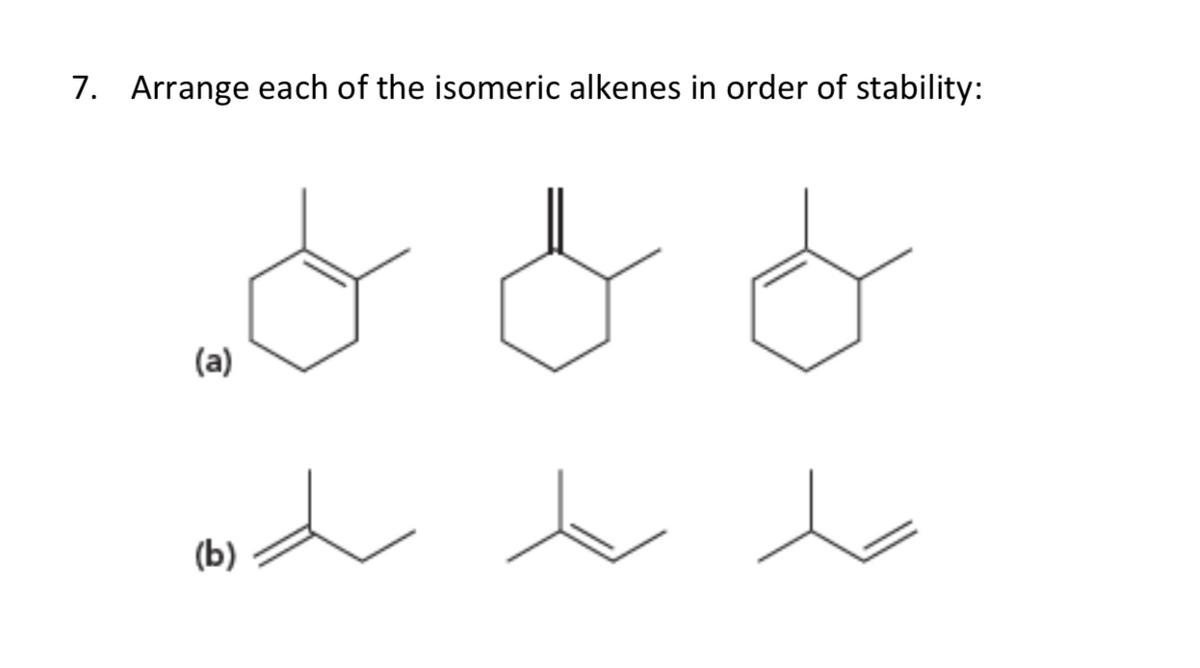
Arrange Each Set Of Isomeric Alkenes In Order Of Stability.
Alright, gather 'round, my fellow caffeine-fueled thinkers and chemistry curious! Pull up a chair, ignore the spilled latte over there (we've all been there), because we're about to dive into something that sounds super serious but is actually, dare I say, kinda fun. We're talking about isomers. Specifically, isomeric alkenes. Sounds like a mouthful, right? Like something you'd find on a particularly gnarly science exam or whispered by a mad scientist in a dimly lit lab. But fear not! Think of me as your friendly neighborhood alkene whisperer, here to demystify these molecular chameleons.
So, what are these "isomeric alkenes" we're so worried about? Imagine you've got a bunch of LEGO bricks, right? You've got the same number and type of bricks, but you can snap them together in different ways. That's basically what isomers are – molecules with the same chemical formula but a different arrangement of atoms. And alkenes, bless their little double-bonded hearts, are the ones with that extra oomph, that jazzy C=C double bond. It's like the sprinkles on the molecular cupcake of life!
Now, the big question, the Everest of alkene knowledge (okay, maybe more like a slightly steep hill), is this: how do we rank their stability? Which alkene is the chillest, the most content, the one that just wants to vibe in its molecular existence? And which one is a hyperactive little molecule, always on the verge of a chemical existential crisis? It's all about how happy and secure those atoms feel, and believe me, in the world of molecules, stability is the ultimate status symbol.
The Great Alkene Stability Showdown!
Picture this: a grand stage, a spotlight, and a lineup of alkenes ready to prove their worth. We're not talking about a beauty pageant here, folks. We're talking about a stability contest. And like any good competition, there are rules, and there are winners. The biggest factor influencing how happy an alkene is? It's all about its substituents. Think of substituents as the entourage of an alkene. The more friends, the more popular, the more… well, stable it tends to be.
So, let's break it down, from the least stable to the most, like a graduating class where the shy kid in the back is surprisingly the one landing the corner office. First up, we have the monosubstituted alkene. This is like the lone wolf of the alkene world. It's got one group hanging off that double bond. It’s okay, it’s not exactly unstable, but it's definitely not the life of the party. Think of it as the sensible sedan of alkenes – gets you from A to B, but doesn't exactly turn heads.
Then we step it up to the disubstituted alkene. Now we're talking! This is where things start to get interesting. We've got two groups attached to the double bond. More friends, more stability! It's like upgrading from a sensible sedan to a sporty hatchback. But here's a little secret, a molecular plot twist: not all disubstituted alkenes are created equal. There's a whole cis and trans thing going on, remember that from chemistry class? Cis means the bulky groups are on the same side of the double bond, giving each other a grumpy side-eye. Trans means they're on opposite sides, giving each other a polite nod and plenty of personal space. And guess what? The trans isomer is generally more stable. Why? Because those bulky groups are like two grumpy sumo wrestlers trying to share a tiny apartment. They just don't get along! The trans arrangement is like giving them separate rooms – much more peaceful, much more stable. It’s a whole molecular détente.
Next in line, strutting its stuff, is the trisubstituted alkene. This is the popular kid, the one with the big group of friends all around. Three substituents means it's feeling pretty darn secure. It's like a seasoned veteran, seen it all, done it all, and is just calmly exuding confidence. It's got more electron density pushing into that double bond, making it a bit more resistant to, you know, breaking apart. Think of it as the luxury SUV of alkenes – spacious, comfortable, and unbothered.
And finally, the undisputed champion, the king of the alkene castle, the one that practically oozes contentment: the tetrasubstituted alkene. This, my friends, is the alpha and the omega of alkene stability. We're talking four substituents, a molecular mosh pit of stability! It's got so many electron-donating groups surrounding that double bond, it’s practically a fortress. It's so stable, it might just start lecturing other molecules on the benefits of mindfulness. It’s the Rolls-Royce of alkenes – pure, unadulterated stability.
Putting It All Together: The Grand Ranking!
So, if we were to line them all up, from the most nervous nellie to the most zen master, the order of stability would look something like this:
Tetrasubstituted Alkenes > Trisubstituted Alkenes > Disubstituted Alkenes > Monosubstituted Alkenes

Think of it like this: imagine you're building a molecule out of sticky notes. The more sticky notes you attach to the central bond, the harder it is to rip it apart. The double bond in an alkene is like a super-strong bond, but those substituents act like extra layers of superglue. The more substituents, the more superglue, the happier and more stable our alkene is. It’s a beautiful, sticky, molecular symphony!
Now, a quick note on that disubstituted squabble. Remember our cis and trans friends? When comparing alkenes with the same number of substituents, the trans isomer will almost always be the more stable one. It's like humans trying to live in close quarters – we generally prefer a bit of personal space, and so do molecules with bulky bits. It’s an unspoken molecular etiquette. So, if you have a choice between a cis-disubstituted and a trans-disubstituted alkene, bet your bottom dollar on the trans one being the chiller molecule.
And there you have it! The not-so-scary, surprisingly logical world of alkene stability. It’s all about those substituents, those little helpers that make our double bonds feel safe and sound. So next time you’re staring at a chemical structure, remember this little story. You can now impress your friends (or at least yourself) by confidently declaring which alkene is the most chill. Now, who’s up for another coffee? I think I just blew my molecular mind.
