Are The Primary Water Insoluble Components Of Cell Membranes

Hey there! So, we’re gonna chat about cell membranes, right? Yeah, I know, sounds super science-y. But honestly, it’s kind of like peeking behind the curtain of life itself. And the star of our show today? The stuff that makes these membranes, well, membranes. Specifically, the bits that don’t like water. Pretty wild when you think about it, huh?
Because, you know, our bodies are like, 70% water. Everything’s watery, watery, watery. And then you’ve got these little walls, these cell membranes, that are totally water-repellent. It’s like a tiny, biological bouncer saying, “Whoa there, water! You can’t just waltz in everywhere!”
So, what are these mysterious, anti-water dudes? The main culprits, the heavy hitters, are these things called lipids. Yep, lipids. Sounds a bit fancy, but think of it this way: they’re basically fats and oils. You know, the stuff that makes your fries crispy or your salad dressing slick. Turns out, they’re also the building blocks of pretty much every cell in your body. Who knew, right?
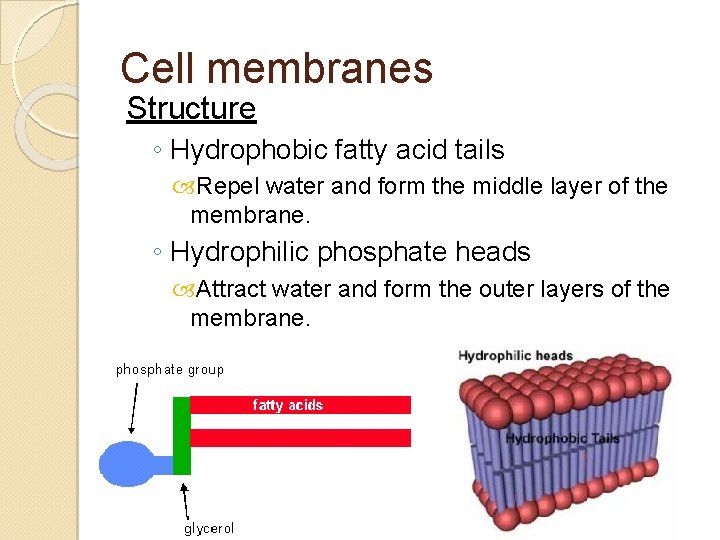
And not just any old lipids, oh no. The rockstars of the cell membrane world are a special kind called phospholipids. They’re like the superheroes of the lipid family, with a very specific superpower: they’re amphipathic. Now, that's a mouthful, I know. But it just means they have two very different personalities. One side of them is like a social butterfly, totally loving water. This is the "hydrophilic" head. Think of it as the part that’s always waving hello to H2O.
But then, the other side? Oh, that’s a whole different story. This is the "hydrophobic" tail. It’s like the introverted sibling, really shy around water. It’d rather hide in a dark corner, away from all that splashy business. These tails are made of long fatty acid chains. Think of them as little water-hating tentacles.
Now, here’s where the magic happens. When you’ve got a whole bunch of these phospholipids hanging out together in an watery environment, like inside or outside a cell, they don't just float around randomly. Oh no. They're far too organized for that. They start to arrange themselves in a very specific way to, you know, deal with their water problem.
They form this thing called a lipid bilayer. Imagine a sandwich. A really, really tiny, microscopic sandwich. The filling of this sandwich is made of the water-hating tails. They all huddle together in the middle, creating a barrier. They're like, "Nope, water, you are NOT getting through this middle section." They’re basically building a hydrophobic fortress.
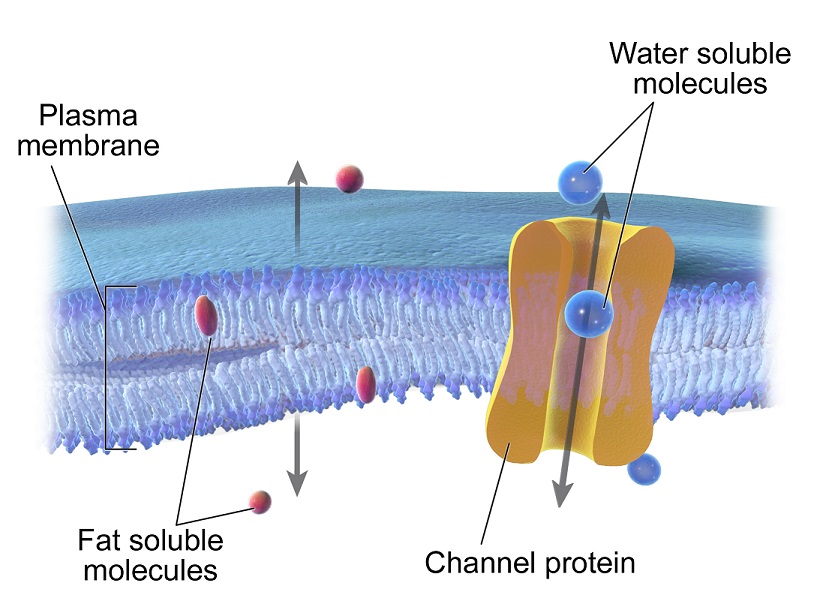
And what about the bread slices on either side of this hydrophobic filling? Those are the water-loving heads! They’re facing outwards, towards the watery environments on both the inside and outside of the cell. They’re like, “Come on in, water! We’re friendly over here!” But they can’t get to the tails, because, well, the tails are busy being anti-water.
So, this lipid bilayer acts like a selective barrier. It lets some things through, but not others. It’s like the bouncer at a really exclusive club, checking IDs at the door. Water, for the most part, can't just zip across this oily middle. It needs a little help, or it just kind of… bounces off. Pretty cool, right? This whole arrangement is what gives the cell membrane its fundamental structure. It’s the basic architecture, the skeleton, if you will.
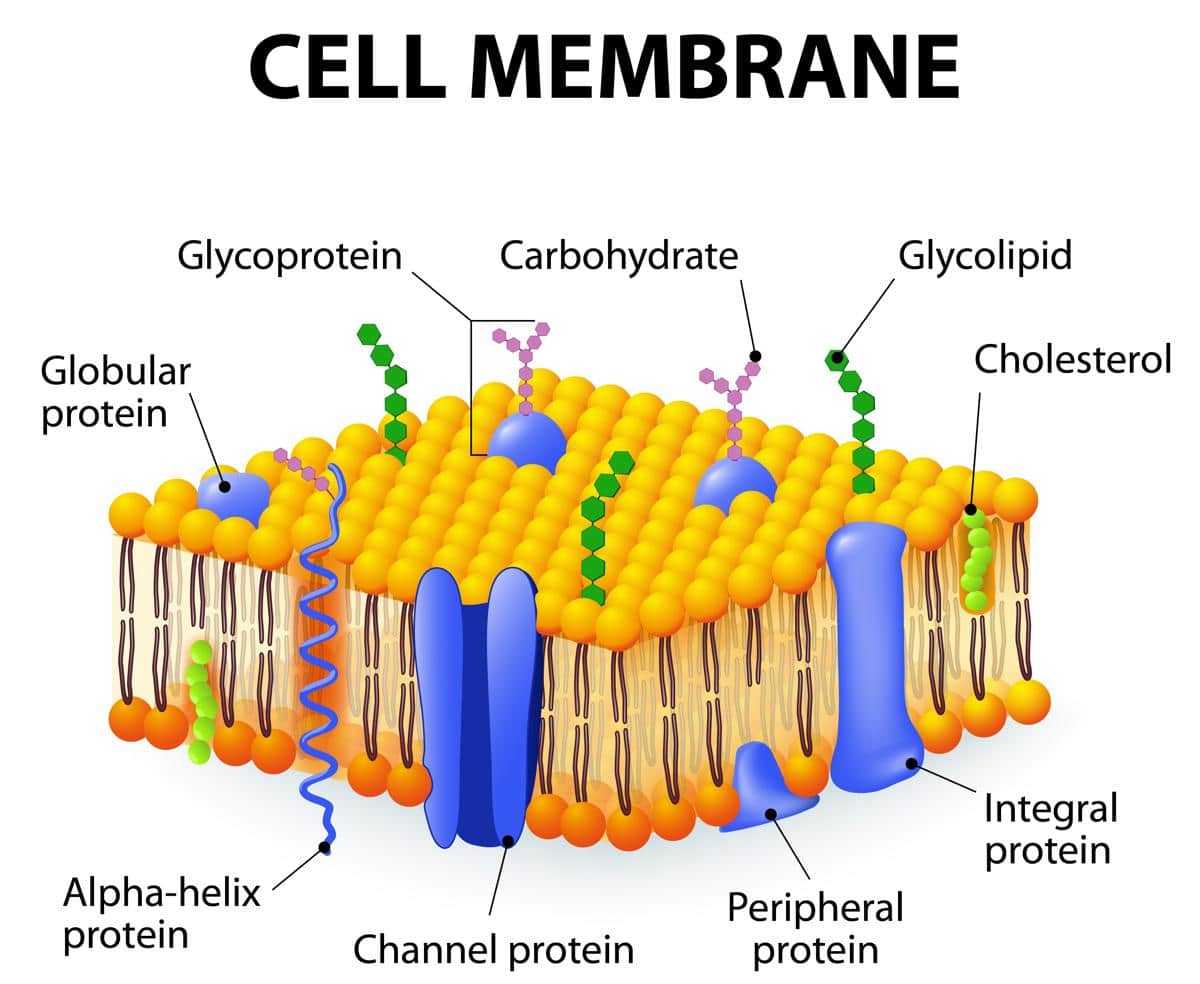
But wait, there’s more! While phospholipids are the absolute superstars, there are other lipids in the mix that are also water-insoluble and play important roles. One of these is cholesterol. Yep, that’s right, cholesterol! You might have heard about it in the context of your arteries, but it’s also chilling in your cell membranes, doing important work.
Cholesterol molecules are smaller than phospholipids, and they kind of slide in between them in the bilayer. Think of them as little spacers, or maybe even tiny, well-placed bricks. Their job is to help regulate the fluidity of the membrane. So, when it’s hot and things are getting a bit too wiggly, cholesterol can help to stiffen things up. It's like putting on a jacket on a chilly day.

And when it’s cold and the membrane is starting to get a bit brittle, cholesterol can help to loosen things up. It's like taking off that jacket when you're getting too warm. It keeps the membrane from becoming too rigid or too fluid, maintaining that Goldilocks zone of just right. Without cholesterol, the membrane would be way too floppy or way too stiff, and that’s not good for the cell. It’s like trying to build a house with jelly – it just wouldn’t stand up!
So, while phospholipids are busy forming the basic bilayer structure, cholesterol is like the unsung hero, fine-tuning the flexibility and stability. It's the quiet one in the back, making sure everything runs smoothly. It's a team effort, you see!
Now, you might be thinking, "Are there any other water-insoluble things in there?" And the answer is, well, yeah! There are other types of lipids, like sphingolipids. They’re a bit more complex than phospholipids, with a different backbone. But guess what? They also have those hydrophobic tails and tend to hang out in the water-repellent parts of the membrane. They often cluster together, forming little specialized platforms within the membrane that have specific jobs.
Think of these platforms as little VIP lounges within the cell membrane. Things that need to communicate with each other, or process certain signals, might gather in these sphingolipid-rich areas. It's like having designated spots for different conversations to happen without all the general hubbub. Very efficient, if you ask me!
And you know what’s really cool? These water-insoluble components, these lipids, they’re not just passive builders. They’re actually quite dynamic. The membrane isn’t some rigid, static wall. It’s more like a fluid mosaic. Things can move around within it. Lipids can swap places, cholesterol can shift around, and even these specialized sphingolipid platforms can form and break apart.
It's like a constantly shifting, oily sea, with bits and pieces embedded in it. And the water-insoluble nature of these lipids is absolutely key to this. If the whole membrane was water-soluble, it would just dissolve into the watery environment. Poof! Gone. The cell would just fall apart, which would be a real bummer, to say the least.
So, the primary water-insoluble components of cell membranes are these amazing lipids. Primarily the phospholipids, which form that essential lipid bilayer, acting as the fundamental barrier. Then you have cholesterol, the unsung hero of membrane fluidity, keeping things just right. And let’s not forget the specialized role of sphingolipids, creating little functional hubs.
It's a testament to nature’s cleverness, isn’t it? How these simple (well, maybe not so simple when you get down to the chemistry) molecules come together to create something so vital for life. They create a boundary, control what goes in and out, and allow for all the complex processes that happen within a cell to occur. It’s like the ultimate waterproof, yet permeable, shield.
And the fact that they are water-insoluble is the whole reason they can do this! Imagine trying to hold water back with… well, more water. It just wouldn’t work, would it? It would be like trying to build a dam out of sponges. Utter chaos!
So, next time you think about your cells, give a little nod to the lipids. They’re the silent guardians, the oily architects, the water-repelling wizards that keep everything inside safe and sound. They’re the unsung heroes of every single cell in your body. Pretty amazing stuff, when you think about it over a cup of coffee, right?
And it’s not just about keeping water out, either. This hydrophobic core of the membrane is also crucial for keeping other things in. Think of all the enzymes, proteins, and other important molecules that are necessary for the cell’s function. If they were just floating around willy-nilly in the watery cytoplasm, they’d get lost, or interact in ways they shouldn’t. The lipid bilayer acts like a container, holding all these vital components together, allowing them to work efficiently.
It’s like a well-organized workshop. You have your tools (proteins, enzymes) neatly arranged on your workbench (the membrane), ready to be used. If everything was just dumped in a big pile, it would be a disaster! The water-insoluble nature of the lipids provides that essential structural integrity to keep the workshop from falling apart.
And even the proteins that are embedded within or traverse this membrane often have regions that are also water-insoluble. These parts interact with the hydrophobic lipid tails, anchoring the proteins in place. So, it’s a beautiful interplay of water-loving and water-hating molecules, all working together in this incredible biological dance. It’s a complex, yet elegant, system.
So, to sum it all up in a friendly, coffee-break kind of way: the main water-repelling dudes in your cell membranes are these awesome things called lipids. We’re talking about the phospholipids, forming that crucial two-layered sandwich structure – the lipid bilayer. Then there’s cholesterol, the membrane’s personal trainer, keeping it flexi. And a little shout-out to sphingolipids for creating those special little clubhouses. All of them, with their water-hating tails, are the backbone, the essential structure that makes cell membranes work. Pretty neat, huh?
