Are All The Carbons In This Structure Coplanar
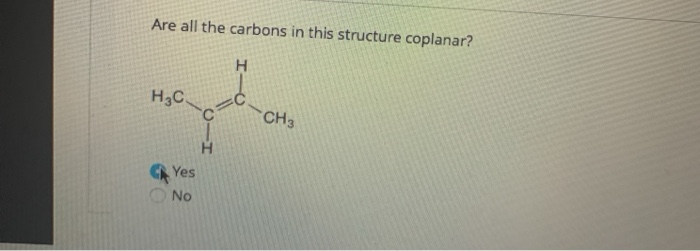
Okay, let's talk about chemistry. Specifically, about those tiny, hardworking things called carbon atoms. You know, the backbone of pretty much everything we see, touch, and even are. We're going to have a little chat about their living arrangements, and whether they all like to hang out on the same flat surface.
Imagine a molecule as a little party. Sometimes, everyone at the party likes to stand in a perfectly straight line, or maybe a neat little circle. It’s very orderly, very tidy. You can imagine them all being on the same dance floor.
Then there are other parties, where things get a bit more… 3D. People are milling about, some are leaning in to chat, others are doing a little jig in the background. It’s a bit more chaotic, a bit more exciting. They're definitely not all on the same dance floor anymore.
And that, my friends, is kind of what we're getting at with this whole coplanar idea. Are all the carbon atoms in a particular molecular structure all sitting pretty on the same flat plane? Like a perfectly laid out picnic blanket? Or are they reaching out, bending, twisting, and generally making a bit of a mess in three dimensions?
Now, I have a slightly unpopular opinion. I think sometimes we get a bit too hung up on the idea of perfect flatness. Like, the universe itself isn't perfectly flat, right? There are mountains, valleys, and the occasional rogue asteroid. So why should our little carbon buddies always be so… two-dimensional?
Think about something simple like benzene. Oh, benzene! That lovely, hexagonal ring of carbons. It’s the poster child for coplanarity. All six carbons and the hydrogens attached to them are practically holding hands in a perfect flat circle. It’s elegant. It’s predictable. It’s like the perfectly organized bookshelf of the molecular world.
And then you have things like, well, pretty much everything else. Take a long chain of carbons, like in a fat molecule. Those carbons aren't all in a line. Oh no. They're doing the zigzag. They're bending. They're doing the molecular equivalent of a shrug and a sigh. It's not flat. It's not even close.
The Case of the Wandering Carbons
We're talking about structures like alkanes. You know, the boring, saturated ones? They’re supposed to be simple. But those carbons, they just won't behave! Each carbon in an alkane chain is attached to four other things. To keep all of those things happy, the angles have to be just right. And that rightness, my friends, pushes things out of flatness.
So, the carbons in a long alkane are like a string of pearls that’s been slightly tangled. They're still connected, but they're not lying flat on the table. They're making little bumps and curves. You can try to push them flat, but they'll just spring back to their preferred, slightly jiggled shape.
It’s like trying to get a toddler to sit still for a photograph. You can try. You can coax. You can bribe. But sooner or later, they’re going to wiggle. And those carbons? They’re the wiggling toddlers of the molecular world.
Even in things that seem somewhat flat, there can be little sneaky deviations. We're not talking about a full-on disco dance party here, but a slight tilt, a tiny bend. It’s enough to ruin the perfect coplanar picnic blanket, if you ask me.
And what about when you start adding other atoms? Like oxygen, or nitrogen? They don't always play by the same rules as carbon. They have their own preferences for where they like to point their arms, and that can push the carbons around too.
The Allure of the Flat and Famous
But there’s a certain beauty to coplanarity, I’ll admit. When molecules are flat, they often have special properties. Take those aromatic compounds, like benzene again. That flatness allows for something called delocalization of electrons. It’s like all the electrons are sharing one giant, communal piggy bank, and it makes the molecule very stable and reactive in interesting ways.
Think of a guitar string. When it’s plucked, it vibrates in a plane. That’s a bit like coplanar molecules. They have a certain harmony, a certain resonance. They’re predictable. They’re the mathematicians of the molecule world.
And then there are molecules with double or triple bonds. These bonds are rigid. They don't allow for much wiggling. So, if you have a series of double bonds all linked up, like in polyenes, you often end up with a pretty flat structure. It's like a molecular tightrope walker, holding perfectly still.

These flat, conjugated systems are often brightly colored. Think of dyes and pigments. That coplanarity is what allows light to interact with them in just the right way to produce those vibrant hues. It’s the molecular equivalent of a perfectly polished mirror, reflecting everything.
So, while my heart might yearn for the chaotic, three-dimensional dance of most carbon structures, I can appreciate the elegant simplicity and the functional power of coplanarity. It’s a specific aesthetic, a specific way of being in the world, for certain molecules.
The Sneaky Exceptions
But let’s not forget the rebels. The troublemakers. The ones who insist on being not coplanar, even when you’d expect them to be. Take something like cyclopropane. It's a three-membered ring of carbons. You'd think, "Three points make a plane, easy peasy!"
But those carbons in cyclopropane are under a lot of strain. The angles are all squished. To relieve some of that tension, the molecule distorts. It’s not perfectly flat. It’s like trying to fit three people into a car designed for two – someone’s got to be a bit contorted.

And then you have larger rings. Cyclohexane, for instance. It’s a six-membered ring, and it’s famously not flat. It adopts a comfy, puckered shape called the 'chair' conformation. It’s much happier that way. It’s the molecular equivalent of finding your favorite comfy armchair.
So, when someone asks, "Are all the carbons in this structure coplanar?" My immediate, slightly cheeky, internal response is often: "Probably not, but let's see how much they're trying to be!" Because, in the grand, wiggly, wonderfully complex universe of molecules, true, perfect flatness is often a rare and special thing.
It's the exceptions that make things interesting, right? It's the unexpected bends and twists that give molecules their unique personalities and abilities. So, while we might admire the elegance of a perfectly planar molecule, let's also give a nod to the ones who are bravely exploring the third dimension.
They’re out there, bending and flexing, creating the complex tapestry of life and matter. And honestly, I find that a little more entertaining than just a flat, boring line. It’s the subtle curves, the gentle undulations, the very three-dimensionality of it all that makes chemistry so fascinating to me.
So, next time you see a chemical structure, don't just ask if it's flat. Ask yourself: how much fun are those carbons having? Are they chilling on the same plane, or are they out there doing their own thing, in glorious 3D? I’m betting on the latter, and I wouldn’t have it any other way.
