Aqueous Acetic Acid Is Neutralized By Aqueous Barium Hydroxide
Ever feel like your daily life is a bit… unbalanced? Like you’re juggling a million things, and some of them just don't quite mix? Well, sometimes in chemistry, things that seem like they’d be at odds actually find their perfect harmony. Today, we're diving into a fascinating little interaction that’s as elegant as a perfectly brewed cup of tea, and surprisingly relevant to how we navigate our own lives: the neutralization of aqueous acetic acid by aqueous barium hydroxide.
Now, before you start picturing bubbling beakers and intimidating equations, let's chill. Think of acetic acid as the zing in your life. It’s the tangy bite of vinegar on your salad, the essential ingredient in that delicious homemade pickle, or even the subtle note that makes your favorite barbecue sauce sing. It’s a common household chemical, readily available and, in diluted forms, totally safe to have around. We're talking about the stuff that makes food taste great!
On the other side of the ring, we have barium hydroxide. This one might sound a bit more exotic, a little more… science-y. But even this has its place, often found in more specialized industrial applications, like in certain types of greases or as a flux in metallurgy. It’s a bit of a quieter character, but just as crucial to our story.
When Worlds Collide (In a Good Way)
So, what happens when these two meet? When you mix a solution of acetic acid (the tangy one) with a solution of barium hydroxide (the quieter one), a beautiful chemical dance begins. It’s a process called neutralization. Imagine it like this: the acetic acid, with its sharp, acidic personality, encounters the barium hydroxide, which has a distinctly basic, or alkaline, character. They’re like two people with different energies who, when they connect, create a sense of calm and balance.
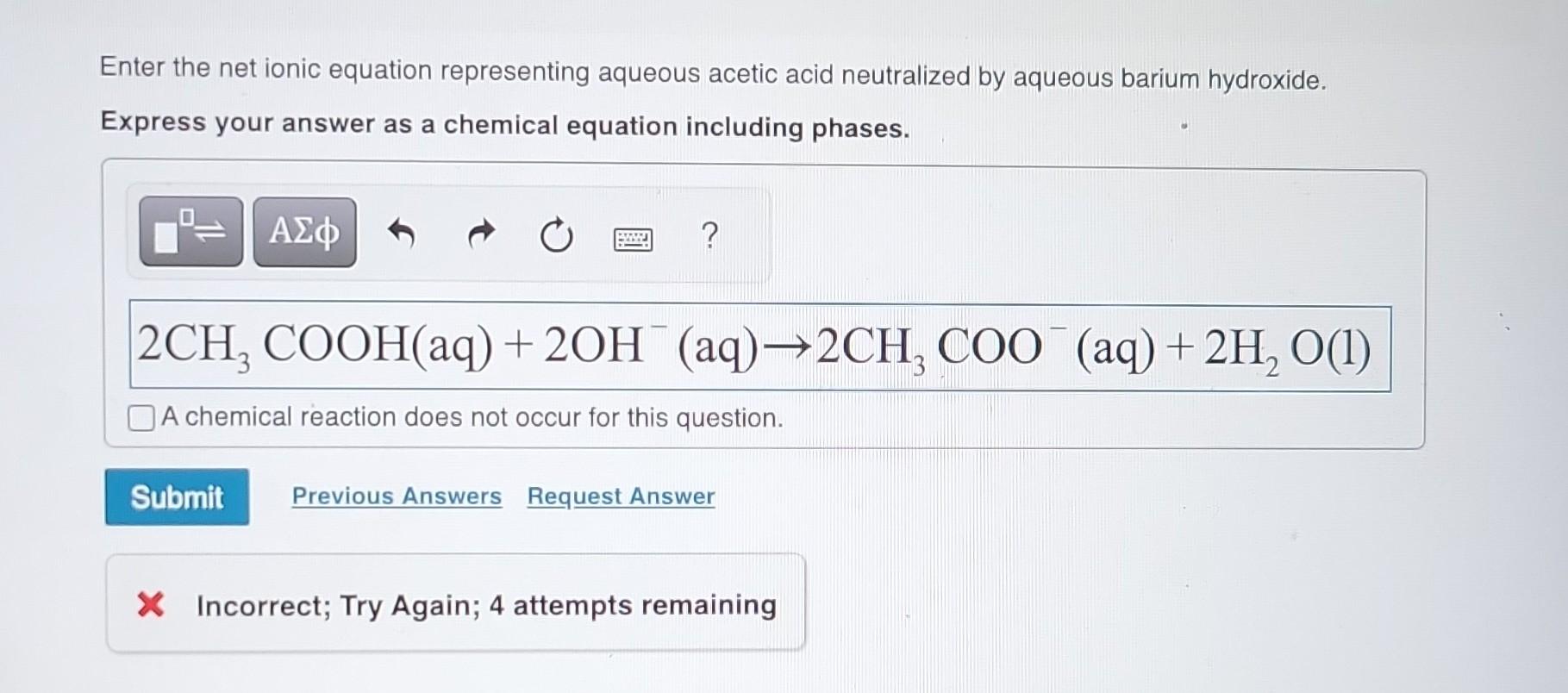
The acid (acetic acid) is all about those positively charged hydrogen ions, H+. They’re the ones giving it that acidic punch. The barium hydroxide, on the other hand, is brimming with hydroxide ions, OH-. These two are the ultimate opposites, and when they meet in the aqueous (watery) environment, they can't resist pairing up. They combine to form water, H₂O. This is the ultimate symbol of harmony – a stable, neutral molecule.

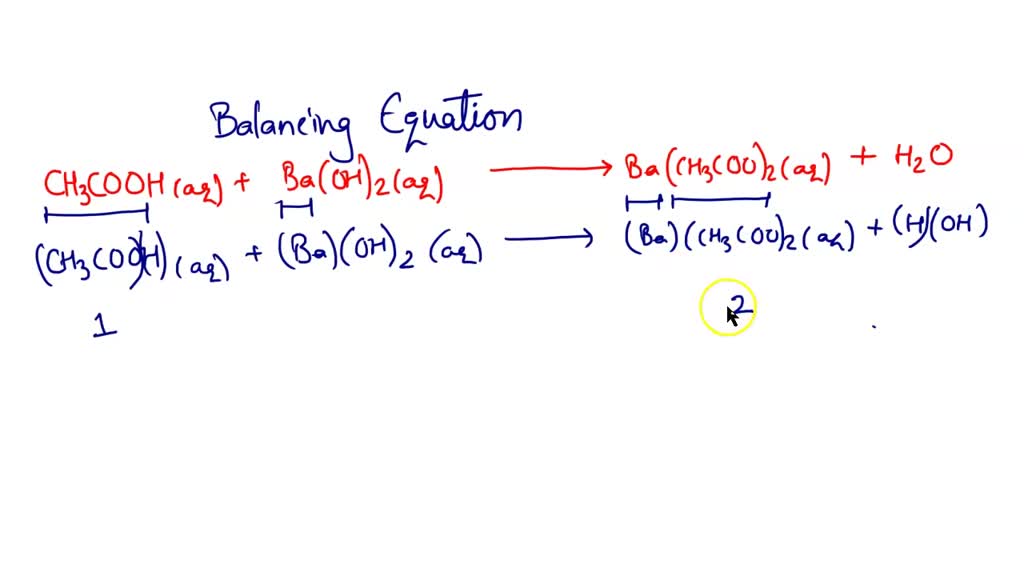
But it’s not just water that’s formed. Since barium is involved, and acetic acid is a salt of acetic acid, we also get a new compound: barium acetate. This is a salt, and in this case, it’s soluble in water, meaning it just hangs out, peacefully dissolved, contributing to the overall balanced solution. The reaction looks something like this, if you’re feeling a bit adventurous: 2CH₃COOH (aq) + Ba(OH)₂ (aq) → Ba(CH₃COO)₂ (aq) + 2H₂O (l).
Think of it like a lively party (the acid) meeting a calm, collected presence (the base). The initial energy is high, maybe a bit chaotic. But as they interact, the excess energy is absorbed, creating a more serene and balanced atmosphere. The final result? Not just peace, but also something new and useful – the balanced solution and the newly formed barium acetate.

Practical Pointers: Bringing Chemistry to Your Kitchen (and Beyond)
Now, you’re probably not going to be mixing barium hydroxide in your kitchen anytime soon – it’s a bit more hands-on than that. But the principle of neutralization is everywhere! We see it in action when we…
- Baking Soda and Vinegar Volcanoes: Remember those classic science fair projects? The fizzy eruption is a mini-neutralization reaction! The acetic acid in vinegar reacts with the sodium bicarbonate (baking soda), producing carbon dioxide gas, water, and sodium acetate. It's a fun way to see chemistry in action. The "volcano" is just the gas escaping!
- Antacids: Got a little heartburn after that spicy meal? Over-the-counter antacids often contain bases like magnesium hydroxide or calcium carbonate. They work by neutralizing the excess stomach acid (hydrochloric acid), bringing relief. It’s your digestive system getting back to its happy, balanced state.
- Gardening: If your soil is too acidic for your prize-winning tomatoes, a gardener might add lime (calcium carbonate) to raise the pH. This is another form of neutralization, making the soil more hospitable for plants. Your garden, like your stomach, needs a good pH balance!
The beauty of neutralization is its ability to restore equilibrium. It’s about taking something with an extreme characteristic and bringing it back to a more moderate, stable state. This is a concept that resonates deeply, isn't it?
Fun Facts and Cultural Quips
Did you know that the term "acetic" comes from the Latin word "acetum," meaning vinegar? It’s been a part of human civilization for millennia, used for everything from preserving food to medicinal purposes. The ancient Romans even used vinegar to clean wounds and as a preservative.
Barium, on the other hand, gets its name from the Greek word "baros," meaning heavy. It’s a pretty dense element, and its compounds, like barium sulfate, are famously used in medical imaging – think of those chalky drinks you have before an X-ray! It’s pretty amazing how the same elements can have such different applications, from industrial uses to helping us see inside our bodies.
Culturally, the idea of balance and harmony is celebrated across the globe. In Japan, the concept of wa (和) emphasizes harmony and social cohesion. In yoga, we strive for asanas that bring our bodies and minds into balance. Even in music, dissonant chords eventually resolve into a pleasing harmony. This chemical reaction, in its own small way, is a perfect illustration of this universal pursuit of balance.

The Ripple Effect: Beyond the Beaker
So, what’s the takeaway from this little chat about aqueous acetic acid and barium hydroxide? It's a reminder that even seemingly disparate elements can come together to create something stable and beneficial. It’s about the power of interaction, the beauty of compromise, and the essential nature of balance.
Think about your own life. Are there situations where you feel too acidic – maybe stressed, overly reactive, or sharp-edged? Perhaps you need a dose of your own “barium hydroxide” – a calming influence, a moment of reflection, a chance to find your equilibrium. Or maybe you feel too much like a bland base, lacking that necessary spark. Introducing a little bit of your own “acetic acid” – a new perspective, a burst of creativity, a willingness to be a little more… zingy – could be just what you need.
The chemical world is full of these elegant partnerships. They remind us that sometimes, the most profound changes happen when different energies meet, interact, and find a way to coexist, creating a new, more stable, and often more interesting whole. It’s a gentle nudge from the universe, perhaps, to seek balance in our own interactions and to appreciate the beautiful chemistry that unfolds when we allow ourselves to connect, neutralize, and grow.
