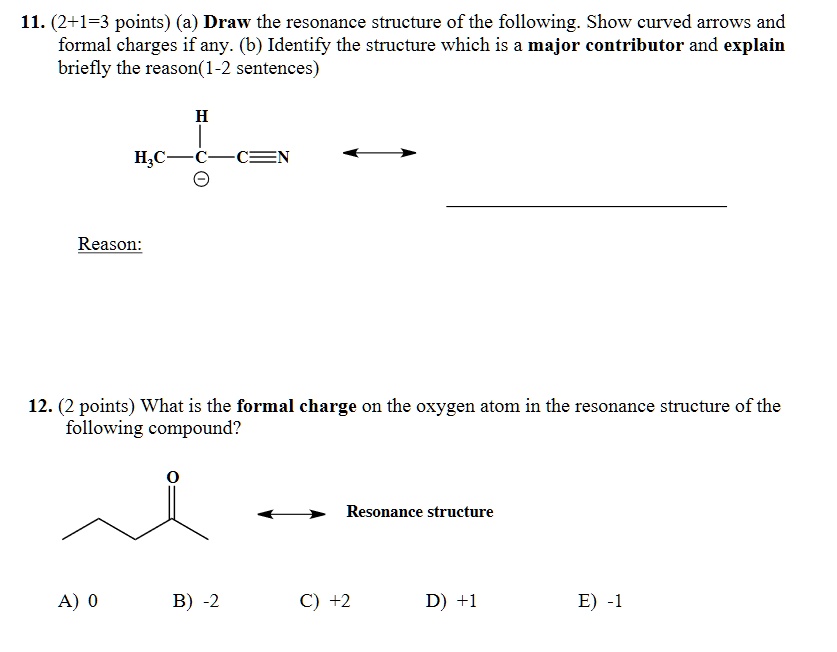
Ap Chemistry Formal Charge And Resonance Worksheet Answers

Okay, confession time. The other day, I was trying to make my famous (in my own head, at least) lemon meringue pie. I’d gotten the meringue just right – stiff peaks, glorious and cloud-like. But then, disaster struck. I somehow misread the recipe and ended up with a pie that tasted… well, let's just say it tasted less like sunshine and more like a slightly confused science experiment. Oops.
It got me thinking. You know, sometimes in chemistry, we have these moments where things just don't seem to add up perfectly. You've got your atoms, your electrons, all supposed to be behaving themselves according to the rules, and then… poof. Things get a little fuzzy. That's kind of where formal charge and resonance come in, and why tackling those AP Chemistry worksheets can feel a bit like trying to bake that perfect pie – sometimes you need a secret ingredient or a little extra finesse.
Unpacking the "Oops" Moments: Formal Charge
So, what is this "formal charge" thing anyway? Think of it like assigning a "score" to each atom in a molecule. It's not the actual charge, mind you. That’s a whole other ballgame involving electronegativity and bond polarity. Formal charge is more like a bookkeeping tool. It helps us figure out which arrangement of electrons in a Lewis structure is the most likely or the most stable. You know, the one that makes the most sense, the one that minimizes those awkward "oops" moments.
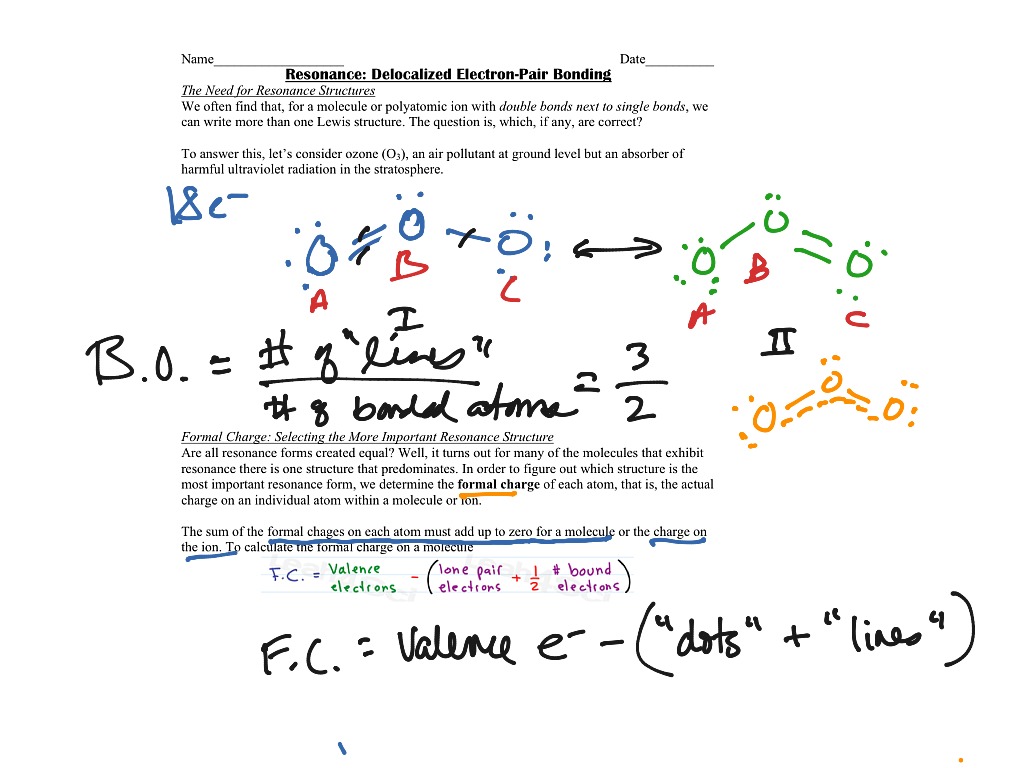
The formula itself is pretty straightforward, which is a relief. It’s basically: (Number of valence electrons in a free atom) – (Number of non-bonding electrons) – (1/2 * Number of bonding electrons). Easy peasy, right? So, if you're looking at your AP Chemistry formal charge and resonance worksheet answers, and you're scratching your head, this is the first place to start.
Let's break it down. "Number of valence electrons in a free atom" – that's just looking at your periodic table. For carbon, it's 4. For oxygen, it's 6. Simple enough. "Number of non-bonding electrons" – these are the lone pairs, the little guys hanging out by themselves on an atom. "Number of bonding electrons" – that's all the electrons involved in the bonds between atoms. Remember, each bond has two electrons, so we divide that by two.
Why do we even bother with this formal charge thing? Well, it’s like having a cheat sheet for Lewis structures. When you can draw multiple valid Lewis structures for a molecule (and trust me, you will!), the one with the lowest sum of formal charges, and where any negative formal charges are on the most electronegative atoms, is usually the one that’s closest to reality. It's the scientifically "least awkward" structure, if you will. It’s the molecular equivalent of a perfectly balanced soufflé, not a deflated pancake.

So, when you're working through those problems, and you have a choice between two structures, calculate the formal charges for each atom in both structures. The structure where the atoms have formal charges closer to zero is generally the winner. If you have to have non-zero charges, try to put the negatives on the atoms that are really good at holding onto electrons (hello, oxygen and fluorine!). This is where those worksheet answers will start to click.
The "But What If?" of Resonance
Now, for the real mind-bender: resonance. Imagine you've drawn a Lewis structure, and you've calculated your formal charges. Everything looks pretty good. But then, you realize… wait a minute. That double bond could totally be over here instead of there. Or that negative charge could be on this oxygen atom, or that other oxygen atom.
This is where resonance structures come into play. Resonance occurs when you can draw more than one valid Lewis structure for a molecule or ion, but the actual molecule isn't flipping between these different structures. Instead, the electrons are delocalized. They’re spread out over multiple atoms.
Think of it like this. Let's say you're trying to describe a really cool band. You could describe the lead singer (one Lewis structure), or the guitarist (another Lewis structure). But the real band is the whole group working together, right? The sound isn't just the singer or just the guitarist; it’s the synergy of everyone. That’s kind of what resonance is like for molecules.
The actual molecule is a resonance hybrid. It's an average of all the contributing resonance structures. It's like a blended smoothie of all the possible Lewis structures. The bonds in a resonance hybrid often have characteristics of both single and double (or even triple) bonds. They're stronger and shorter than typical single bonds, but weaker and longer than typical double bonds.
When you're looking at your AP Chemistry formal charge and resonance worksheet answers, and you see those double-headed arrows (↔) between structures, that's a big clue that you're dealing with resonance. Those arrows don't mean the molecule is switching back and forth. They mean "these are contributors to the overall structure."
How do you identify resonance? Usually, it's when you have a multiple bond adjacent to an atom that has a lone pair or a negative charge. Or when you have a positive charge next to a multiple bond. These are the situations where electrons can shift around to create those different, but equally valid, Lewis structures.
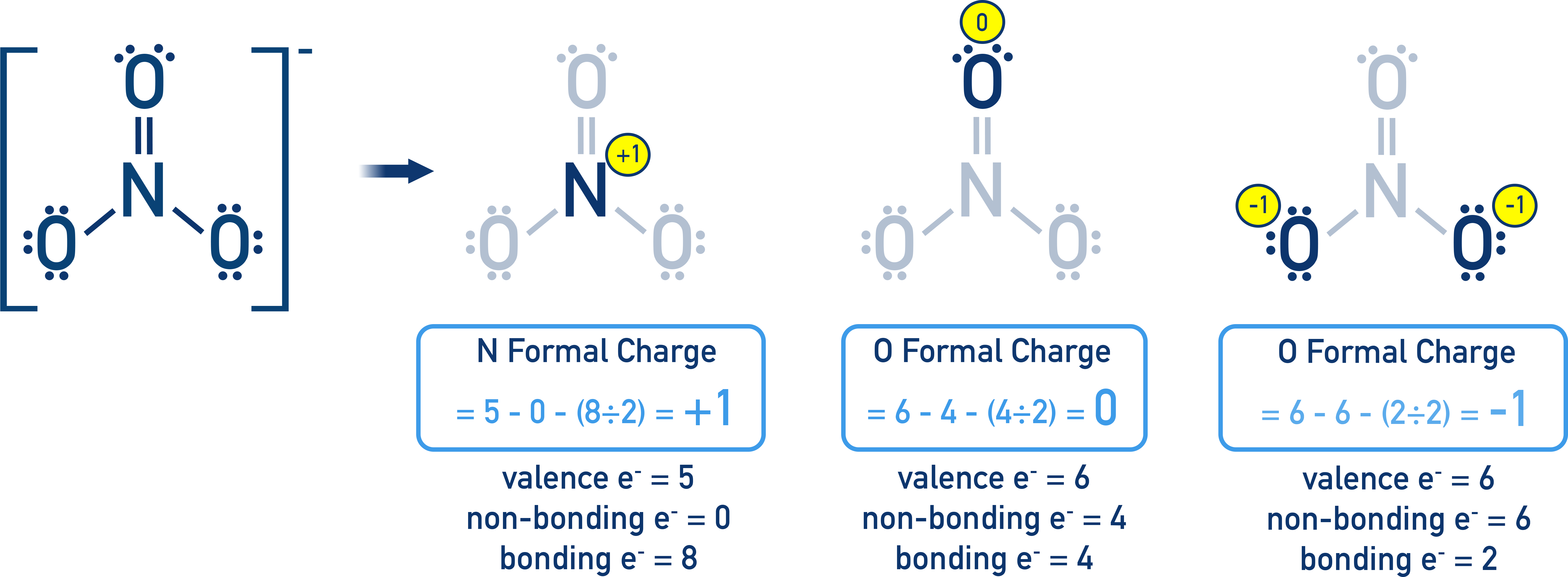
For example, consider the carbonate ion, CO₃²⁻. You can draw it with a double bond between carbon and one oxygen, and single bonds to the other two. But that double bond could be with any of the three oxygens! So, you have three resonance structures. In reality, all three C-O bonds are identical, somewhere between a single and a double bond. The negative charge is also spread out over the oxygen atoms.
This is a crucial concept for understanding molecular stability and reactivity. Molecules with resonance are generally more stable because those delocalized electrons mean the energy is distributed more evenly. It’s like having your energy spread out over a whole day of studying instead of trying to cram it all into one hour – much more sustainable!
Putting It All Together: The Worksheet Journey
So, let’s say you’re staring at that AP Chemistry formal charge and resonance worksheet. You’ve drawn your Lewis structure, you’ve counted your electrons, and now you’re wondering if you’ve got it right. Here's a little game plan:
- Draw the skeletal structure: Figure out which atom is the central atom. Usually, it's the least electronegative atom (but not hydrogen!). Connect the outer atoms to the central atom with single bonds.
- Distribute valence electrons: Count all the valence electrons from all the atoms (and add or subtract for ions). Start by giving octets to the outer atoms, then fill the octet of the central atom.
- Check octets: Does every atom (except hydrogen, which is happy with two) have a full octet? If not, you might need to form multiple bonds. Move lone pairs from outer atoms to create double or triple bonds.
- Calculate formal charges: This is where the magic happens! Use the formula: (Valence electrons) – (Non-bonding electrons) – (1/2 * Bonding electrons) for each atom.
- Evaluate stability: The best Lewis structure will have:
- A sum of formal charges equal to the overall charge of the molecule or ion.
- Formal charges as close to zero as possible.
- Any negative formal charges on the most electronegative atoms.
- Any positive formal charges on the least electronegative atoms.
- Identify resonance: If you can draw multiple valid Lewis structures by simply moving electrons (lone pairs or pi electrons), and the atoms themselves stay in the same positions, then you have resonance! Use the double-headed arrow (↔) to show these contributing structures. Remember, the real molecule is the hybrid.
Those worksheet answers are your guides, your maps through this sometimes-tricky terrain. When you get one wrong, don't beat yourself up. It’s part of the learning process. Think about why it was wrong. Did you miscalculate formal charges? Did you miss a possibility for resonance? Understanding the mistake is way more valuable than just seeing the correct answer.
And hey, sometimes those worksheets can feel like a mountain to climb. You might be looking at a problem and thinking, "Is this it? Am I ever going to get this?" Take a breath. Step away for a minute. Come back with fresh eyes. Often, the solution is hiding in plain sight, just waiting for you to notice it.
Remember that lemon meringue pie I mentioned? Turns out, I’d accidentally grabbed a recipe for a lemon custard pie. Totally different vibe! But once I realized my mistake and adjusted, it was still delicious. Similarly, once you understand formal charge and resonance, you'll see how these concepts help explain the real behavior of molecules, not just some idealized version. They’re the tools that help us move from just drawing structures to understanding them. So, keep practicing, keep questioning, and soon those worksheets will feel less like a chore and more like a fascinating puzzle to solve!
