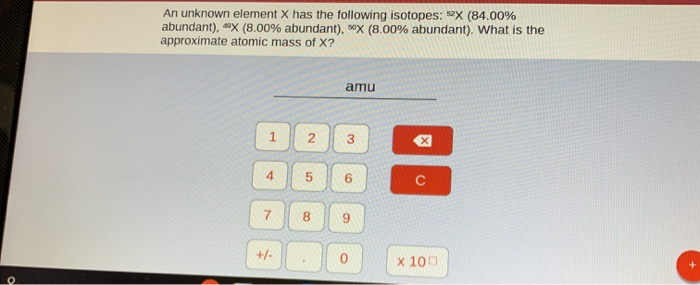
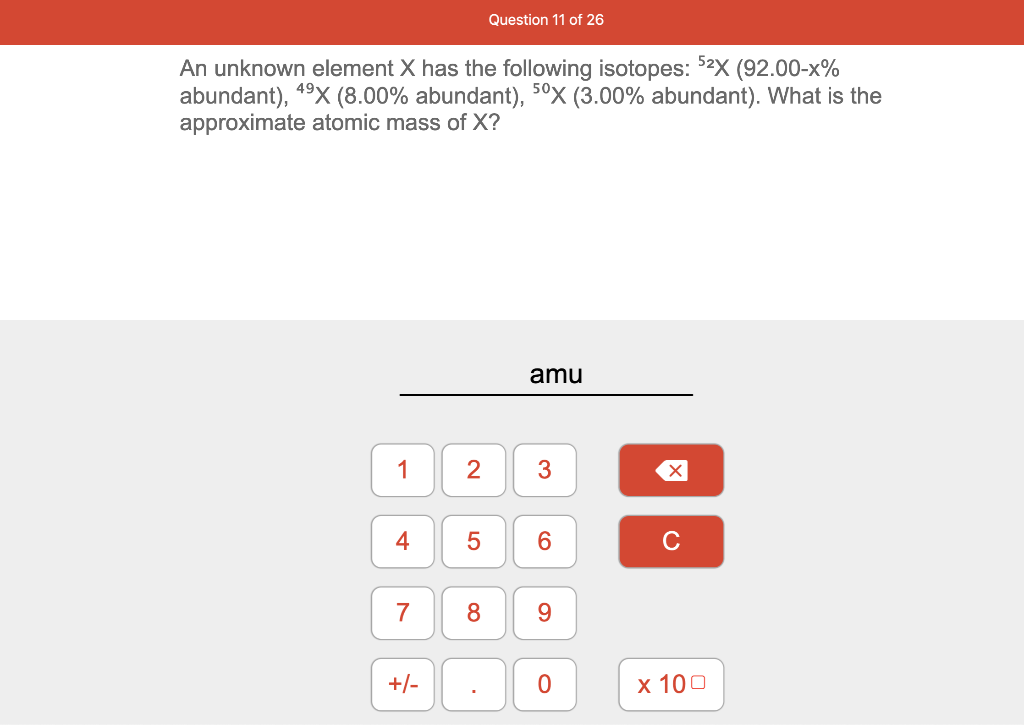
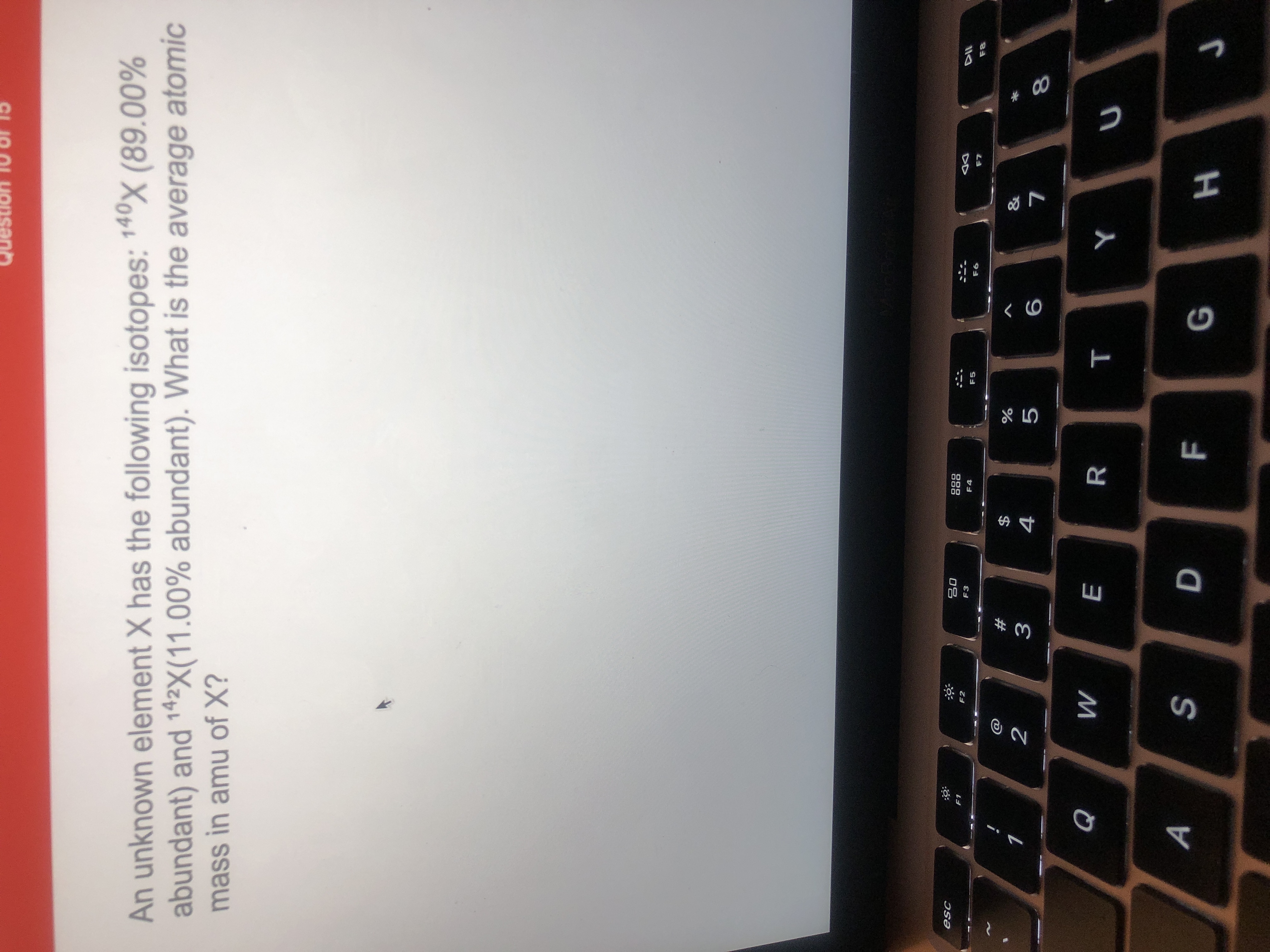
An Unknown Element X Has The Following Isotopes
Okay, so, picture this. We're all hanging out, maybe with a big ol' mug of coffee, right? And suddenly, BAM! Someone drops a bombshell. There's this brand new element on the scene. We're talking about Element X, because, you know, it’s so mysterious, it needs a mysterious name. It's like the shy kid in class, doesn't want anyone to know its real name. Heh.
And get this – this Element X isn't just one thing. Oh no. It's got isotopes. Yeah, you remember those? Those sneaky variations of an element that have the same number of protons, but different numbers of neutrons. It's like siblings, but one's got a bit more… baggage. Or maybe just a bit more weight. Literally. More neutrons mean more mass, obviously. Elementary, my dear Watson, but also kind of mind-bending when you think about it too hard.
So, we've got these isotopes of Element X, and they're not exactly lining up nicely, are they? They’re like a bunch of personalities all crammed into one element. You’ve got your ‘X-150’, your ‘X-152’, and then, because why not, your ‘X-154’. Seriously, who came up with these numbers? Did they just grab them out of a hat? It’s like naming your pets. “Let’s call this one… Fluffy!” or “Hmm, I feel like a ‘152’ today!” It's a whole vibe.
Now, the big question, right? Why should we even care about these specific isotopes? I mean, they sound like they’re from some sci-fi movie, don’t they? Element X-150. Sounds like a robot name. Or a really bad band name. But here’s the kicker: each one of these isotopes has its own little quirks. And those quirks are what make them important. It’s like with people, you know? We all have our own little quirks, and they’re what make us… us. Or at least, what make us interesting enough to write an article about, apparently.
Let's dive into the nitty-gritty, shall we? Or, you know, the slightly less gritty but still important bits. Because understanding these isotopes is key to unlocking the secrets of Element X. It's like a treasure hunt, but instead of gold, we're looking for… scientific knowledge. And maybe a free sample if we’re lucky. A girl can dream, right?
So, we have our first contender: Element X-150. Now, this one, from what I understand – and trust me, I’m no Nobel laureate yet – is pretty darn stable. Stable, you say? What a concept! It’s like the grandparent who’s seen it all, done it all, and is just chilling on the porch, watching the world go by. No drama, no fuss. Just… existing. And that, my friends, is a superpower in its own right, especially in the chaotic world of atoms.

This stability means X-150 hangs around for a good long while. It's not going to, you know, spontaneously combust or turn into something else in a blink of an eye. It's reliable. It's the friend you can always count on to bring the snacks to the party. The one who remembers your birthday. It’s the backbone of the X family, really. Without X-150, who knows what kind of mess the other isotopes would get into?
But wait, there's more! Because, as we know, life is rarely that simple. Enter Element X-152. This guy? It’s a bit of a… middle child. It’s not as chill as X-150, but it’s not exactly a wild card either. It’s got a half-life that's… let’s just say ‘moderate’. Moderate is a polite word for ‘it decays, but not super fast’. Think of it as the teenager of the X family. Still figuring things out, prone to occasional bursts of energy, but generally manageable. Probably listening to a lot of questionable music.
The half-life of X-152 is what makes it interesting for certain applications. Imagine you need something that lasts for a while, but not forever. Like, you want to track something, but you don’t want the tracker to be there until the end of time. X-152 could be your guy! It’s like the perfect amount of commitment. Not too much, not too little. Just right. Goldilocks would approve, I bet.
And then, because the universe clearly likes to keep us on our toes, we have Element X-154. This one? This is the… energetic one. The one who’s always buzzing, ready to go, ready to do something. It’s the Labrador puppy of the X isotopes. Bouncing off the walls, wagging its tail at everything. And guess what? It’s also the least stable. Yep. It decays the fastest. It’s got a short fuse, this one. Like a firecracker on the Fourth of July. Lots of flash, maybe a bit of bang, and then… gone. Poof!
This rapid decay is actually super important, though. Think about it. If something decays quickly, it means it’s giving off energy. And energy, my friends, is a powerful thing. X-154 could be the key to some really cool technologies. Maybe it’s used in medical imaging, where you need a quick burst of something to see what’s going on inside you. Or maybe it's used to power some futuristic gadget that we haven't even invented yet. The possibilities are, dare I say it, endless.
So, we’ve got our trio: the stable grandparent (X-150), the moderate teenager (X-152), and the energetic puppy (X-154). Each with their own unique characteristics, their own little personalities. It’s like a reality TV show, but with atoms. Who would watch? Probably me. And maybe you. If there were snacks involved, definitely me. And possibly a dramatic soundtrack.
Now, how do we know all this? Are scientists just, like, poking these isotopes with sticks and seeing what happens? Well, not exactly. They use this fancy thing called a mass spectrometer. Sounds intimidating, right? But basically, it's like a really, really precise scale for atoms. It can tell you the exact mass of each isotope. And that, my friends, is the first clue. Because the mass is directly related to the number of neutrons. More neutrons, more mass. Simple as that. Or, you know, as simple as atomic physics gets.
And once you know the mass, you can start figuring out other things. Like, how common is each isotope? Is X-150 the most popular kid in school? Or is X-154 the rare, exotic one that everyone’s talking about? This is determined by something called natural abundance. It’s basically the percentage of each isotope you’d find if you just scooped up a big ol’ chunk of Element X from wherever it lives. Is it hiding deep in the Earth’s crust? Is it floating around in space? We don’t know yet, but that’s part of the mystery!
The natural abundance tells us a lot. If X-150 is, say, 90% of the mix, then that tells us it's the dominant player. It's the one that's most likely to be found. If X-154 is only 1%, then it’s a rare gem. We have to work a bit harder to find it. It’s like finding a four-leaf clover. You don’t stumble upon them every day. You gotta put in the effort!
But it’s not just about mass and abundance. Oh no. We also have to consider their properties. What does Element X-150 do when you put it in different situations? Does it react with oxygen? Does it conduct electricity? Does it have a particular color? These are the things that make it useful, or dangerous, or just plain interesting. It's like getting to know a new friend. You don't just know their name and their birthday; you want to know their hobbies, their dreams, what makes them laugh.
And this is where things get really exciting, because different isotopes of the same element can behave slightly differently. It's subtle, mind you. Not like, "Oh, X-150 is a solid at room temperature, but X-154 is a gas!" It’s more like tiny, almost imperceptible differences in how they form bonds, or how quickly they undergo certain chemical reactions. Think of it as a different flavour of the same ice cream. Vanilla, vanilla bean, French vanilla. All vanilla, but with a little something extra.
The half-lives, though, are probably the most dramatic difference. The speed at which they decay. As we said, X-150 is a marathon runner. X-152 is a decent sprinter. And X-154? It's a flash. Gone in a heartbeat. This has huge implications. For example, if you need to do some kind of radioactive dating – like figuring out how old that ancient artifact is – you need an isotope with a predictable, long half-life. So, maybe X-150 is your guy there. Or, if you need to generate a burst of energy for a specific purpose, and you want it to dissipate quickly, then X-154 becomes your best friend. It’s all about matching the right isotope to the right job. It's like a perfectly tailored suit. Or, you know, a perfectly tailored atom.

So, why all this fuss about Element X? Well, every new element, and every new isotope, is a piece of the puzzle of the universe. It’s like finding a new word in a language you’re trying to decipher. Suddenly, you can say a little bit more, understand a little bit more about how everything works. It expands our knowledge, our capabilities, and our sense of wonder. It’s a testament to human curiosity and our relentless drive to understand the world around us.
Imagine the possibilities! What if Element X is crucial for a new type of battery? Or a revolutionary medical treatment? Or even the key to faster-than-light travel? (Okay, maybe that last one’s a bit of a stretch, but you never know! Science is full of surprises.) Each isotope offers a unique set of characteristics that could unlock these doors. It’s like having a whole set of keys, and each key opens a different, exciting lock.
And the best part? This is just the beginning. We've identified these three isotopes, but who knows what else is out there? Maybe there’s an X-148 lurking in some undiscovered corner of the periodic table. Or an X-160 that we haven't even dreamt of yet. The universe is vast, and full of scientific surprises waiting to be uncovered. It’s like a never-ending buffet of discovery. And I, for one, am ready for seconds. And thirds. And maybe even dessert.
So, next time you’re pondering the mysteries of the universe over your morning brew, give a little thought to Element X and its intriguing isotopes. They might just be the key to unlocking the next big scientific breakthrough. Or, at the very least, they make for a pretty interesting conversation starter. Cheers to the unknown, and to the atoms that make it all possible!
