An Ionic Compound Of Calcium And Chlorine Would Be Named
Ever found yourself staring at a mysterious label, wondering what on earth is in that thing you just bought? Or maybe you've tinkered with a science kit, fascinated by how different elements combine? Well, buckle up, because we're about to dive into the delightful world of naming chemical compounds, a surprisingly fun and incredibly useful skill! Think of it like decoding a secret language that unlocks the secrets of everything around us, from the food we eat to the medicines that heal us. It's a little bit like solving a puzzle, and the satisfaction of getting it right is absolutely chef's kiss!
But why bother with this seemingly complex task? The benefits are far more widespread than you might imagine. Understanding how compounds are named allows us to identify the substances we're interacting with, which is crucial for safety. Knowing that a compound is, say, an ionic compound of calcium and chlorine tells you a lot about its potential properties. It helps us predict things like solubility, reactivity, and even its role in biological processes. For instance, calcium and chlorine are fundamental to our bodies! You've probably heard of calcium for strong bones, right? Well, it also plays a role in muscle function and nerve signaling. And chloride, the form chlorine takes in these compounds, is essential for maintaining fluid balance and for producing stomach acid.
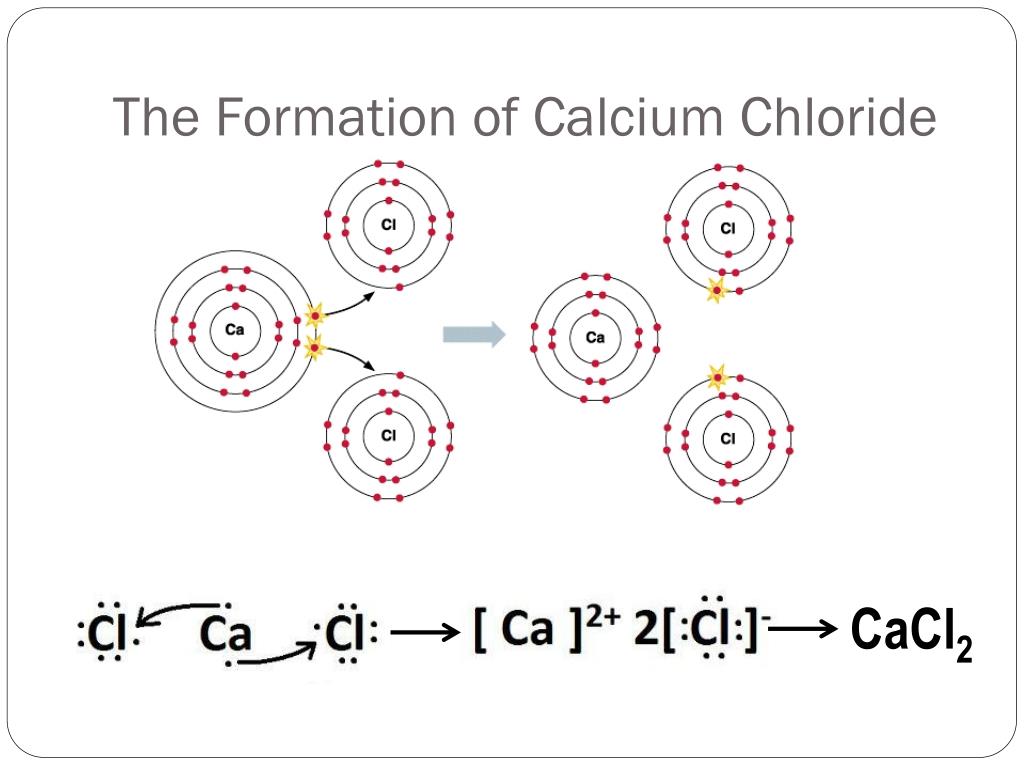
The most common example of an ionic compound formed from calcium and chlorine is, you guessed it, calcium chloride! You might have encountered it in its various forms: as a de-icing agent for roads in winter (it lowers the freezing point of water, making it super effective at melting snow and ice), as a food additive (E number E509) to help firm up canned vegetables or preserve cheese, or even as a dietary supplement. It’s also used in concrete to speed up setting time and in some industrial processes. Pretty versatile, wouldn't you say?
So, how can you get better at this naming game and enjoy it even more? Firstly, start simple. Focus on the basic rules of naming ionic compounds. Remember that for a binary ionic compound (one made of two elements), you take the name of the metal cation (the positive ion) and add the name of the nonmetal anion (the negative ion) with its ending changed to "-ide". In our case, calcium is a metal that typically forms a Ca²⁺ ion, and chlorine is a nonmetal that typically forms a Cl⁻ ion. To balance the charges, you need two chloride ions for every one calcium ion. When you combine these, you get CaCl₂, and the naming is straightforward: calcium chloride. It’s that easy!
Secondly, practice makes perfect! Grab some common household items and look at their ingredient lists. See if you can identify any ionic compounds and try to name them. You'll be amazed at how many you can find! Finally, don't be afraid to look things up. There are tons of great online resources and apps that can help you learn and verify your answers. The more you engage with it, the more intuitive it becomes. It's a journey of discovery, and with a little curiosity and practice, you'll be a chemical naming pro in no time, truly appreciating the chemistry that surrounds us every single day!
