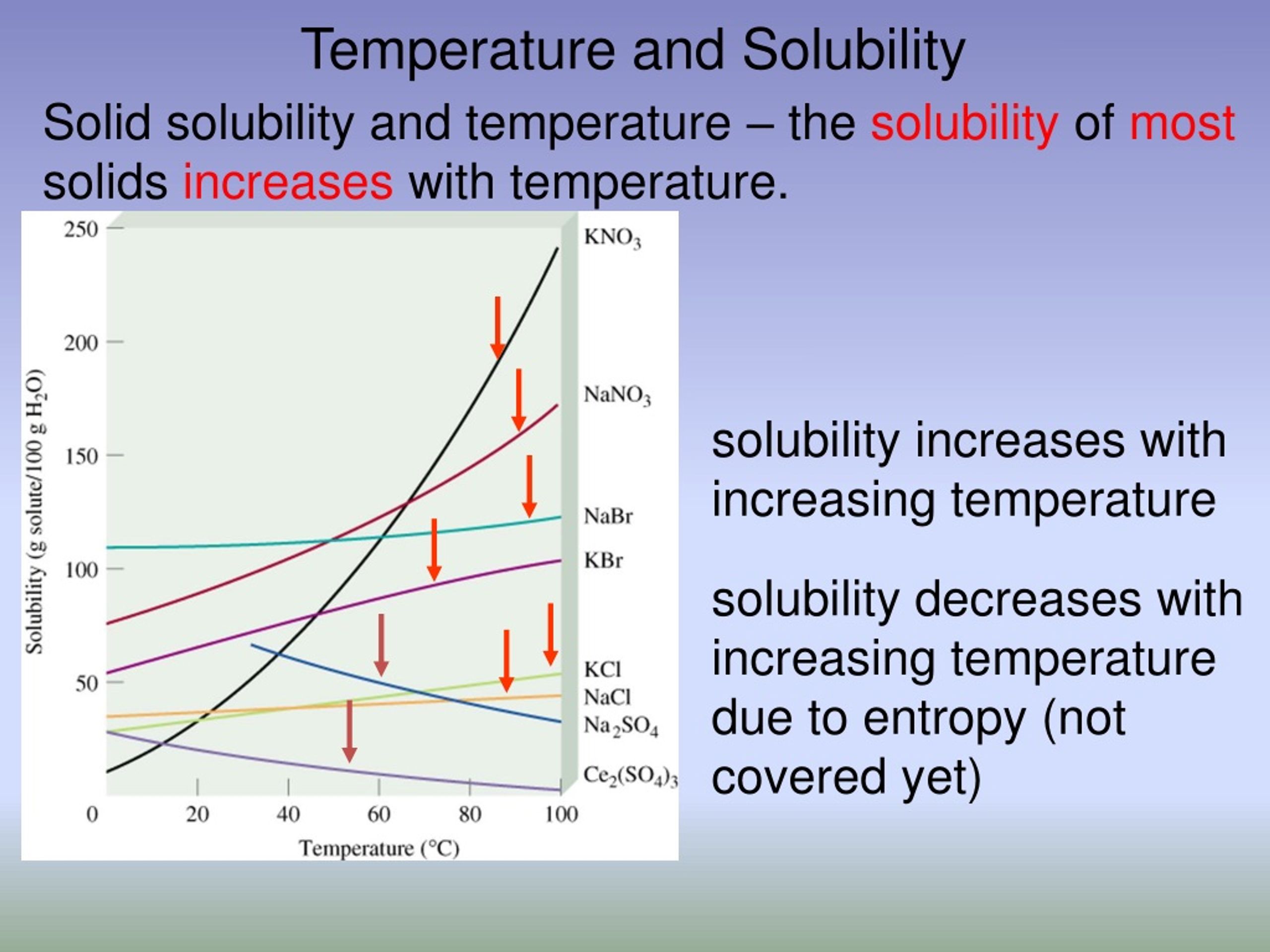
An Increase In The Temperature Of A Solution Usually ________.
Hey there, science pals! Ever wonder what happens when you crank up the heat on a solution? Like, when you're making that perfect cup of tea or boiling pasta? It's not just about getting things hot. Something super cool is actually going down at a molecular level. Get ready, because we're about to spill the beans on what happens when a solution gets a temperature upgrade!
So, the big question is: An increase in the temperature of a solution usually ________. What's the missing word? Drumroll, please... It usually speeds things up! Yep, that's the magic word. Think of it like a party for your molecules. When you add heat, you're basically giving them a shot of espresso. They get all jazzed up and start moving faster. It's a molecular mosh pit, and everyone's invited!
The Molecular Marathon
Imagine your solution is a busy city. Molecules are like tiny little cars zipping around. When it's cold, they're kinda cruising, maybe stuck in a bit of traffic. But when you turn up the thermostat? WHOOSH! The traffic lights turn green for everyone. The cars speed up, change lanes like crazy, and bump into each other way more often. This is the core of what's happening.
This increased speed translates to a few key things. For starters, things tend to dissolve better. Think about stirring sugar into iced tea versus hot tea. Which one dissolves faster? The hot one, obviously! That's because the hotter water molecules have more energy to break apart the sugar crystals and mingle with them. It’s like the sugar is more welcome at the hot party than the cold one.
And it's not just dissolving. Chemical reactions? They get a serious boost! Reactions are basically molecules having a little chat or a big argument. More heat means they're having those chats and arguments at warp speed. They bump into each other more, with more force, making it easier for them to change into something new.
![[ANSWERED] An increase in the temperature of a solution usually - Kunduz](https://media.kunduz.com/media/sug-question/raw/61891909-1657296823.5497665.jpeg?h=512)
Why So Speedy? The Energy Boost!
It all comes down to kinetic energy. This is the energy of motion. When you heat a substance, you're pumping in energy. This energy makes the molecules vibrate more, move faster, and basically have a grand ol' time. The higher the temperature, the higher the kinetic energy, and the more energetic the molecular dance!
It’s kinda like when you're feeling sluggish and someone blasts your favorite upbeat song. Suddenly, you've got the energy to clean the whole house, right? Solutions are the same. A little bit of heat is their favorite playlist, and it makes them ready to rock and roll.

This is why chemists love playing with temperature. It’s a super easy way to control how fast reactions happen. Need something done in a hurry? Crank up the heat! Want to slow things down, maybe to study a reaction more closely? Cool it off. It’s like having a remote control for the pace of chemistry.
Quirky Facts and Fun Tidbits
Did you know that some reactions can be dangerously fast if you heat them up too much? It’s like giving those molecular cars a rocket booster! Sometimes, you have to be careful not to make things too exciting. This is why some experiments are done in fume hoods – just in case things get a little too wild and produce some unexpected gases or even a small pop!
And here’s a funny thought: Imagine if humans behaved like solutions. When we're cold, we're all huddled together, moving slowly, maybe even grumpy. But when we're warm and happy, we're chatting, laughing, and mingling! We’re basically warm-blooded solutions, and a good sunny day can make us all a lot more energetic.

Think about cooking. When you fry an egg, the heat causes chemical reactions that change the egg white from clear and gooey to white and firm. That’s a temperature-driven transformation! Or when you bake a cake, all sorts of complex reactions are happening because of the heat, creating that delicious fluffy texture and yummy smell. Heat is the ultimate chef's secret ingredient!
Beyond Dissolving: More Than Just Speed
While speed is the main event, there are other cool things that can happen. Sometimes, an increase in temperature can change the way things interact. For instance, some substances might become more soluble at higher temperatures, meaning they can dissolve in greater amounts. Others might become less soluble. It's not always a straightforward "more heat, more dissolving" situation for everything. It depends on the specific molecules involved and their delicate dance.
And then there's the concept of equilibrium. In reversible reactions, there’s a point where the forward reaction (reactants turning into products) and the reverse reaction (products turning back into reactants) happen at the same rate. Heating things up can shift this equilibrium. Sometimes it pushes the reaction more towards making products, and sometimes it pushes it back towards the reactants. It’s like a molecular tug-of-war, and temperature can be the referee!
It’s pretty mind-boggling when you think about it. The tiny, invisible world of molecules is constantly reacting and transforming, and temperature is one of the biggest levers we have to influence it. It’s a fundamental principle, but it has so many fascinating implications in everything from brewing coffee to developing new medicines.
So, next time you're warming up your soup or boiling water, take a moment to appreciate the molecular party you're creating. The increase in temperature is usually making everything speed up, from dissolving to chemical reactions. It’s a fundamental, yet incredibly fun, aspect of the world around us. Keep exploring, keep questioning, and keep that curiosity simmering!
