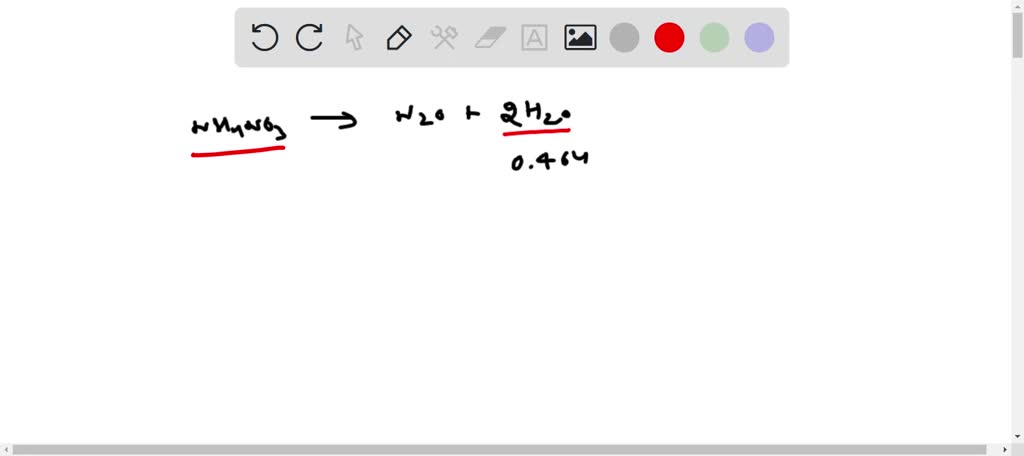
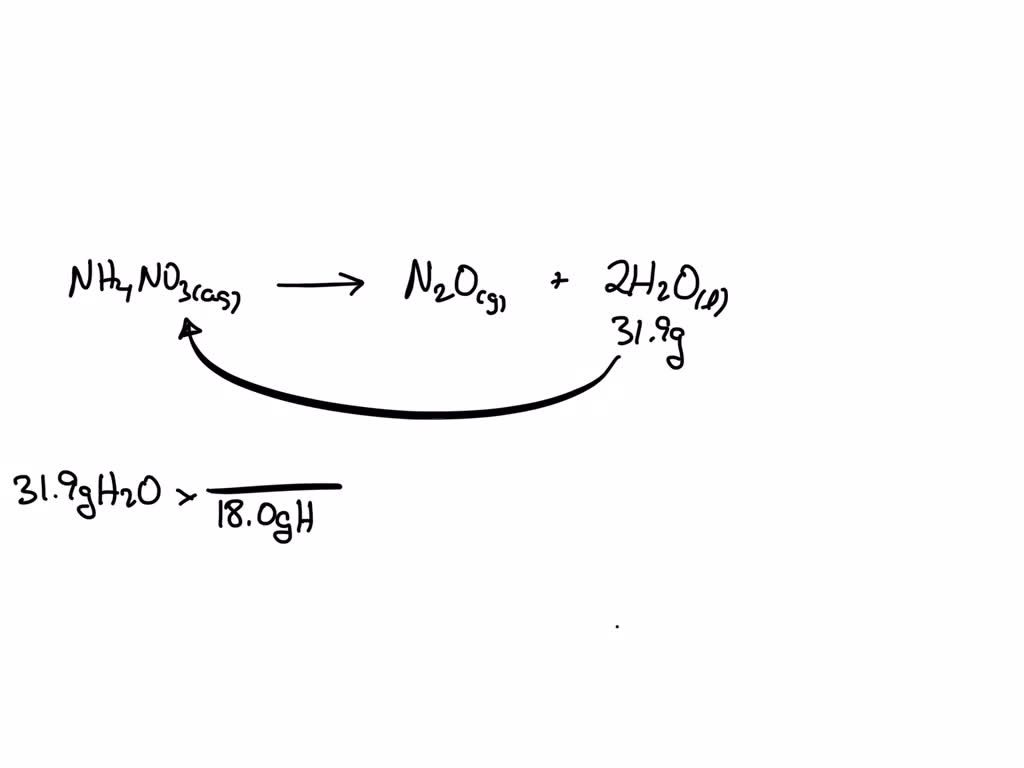
Ammonium Nitrate Aq Dinitrogen Monoxide G Water L
Have you ever wondered what happens when you mix a few everyday things and get something truly spectacular? Well, get ready to be amazed! We're diving into a world where simple ingredients create a showstopper, a real crowd-pleaser.
Imagine a little bit of science magic happening right before your eyes. It’s not about complicated formulas or stuffy labs. It's about a surprising combination that leads to some seriously cool results. Think of it as a science experiment that’s actually… fun!
So, what’s the secret ingredient? Or rather, what are the secret ingredients? We’re talking about a trio that, when brought together, create a fascinating reaction. It's like a recipe for wonder!
The Stars of the Show!
Let's meet our main characters in this exciting little drama. First up, we have Ammonium Nitrate. Now, this might sound like something out of a chemistry textbook, but it's actually a common compound. It's used in a lot of different things, from fertilizers to even some fireworks.
Don't let the fancy name fool you. Ammonium Nitrate is the foundation for our adventure. It's the dependable friend in this dynamic duo (or should we say trio?). It sets the stage for all the exciting things to come.
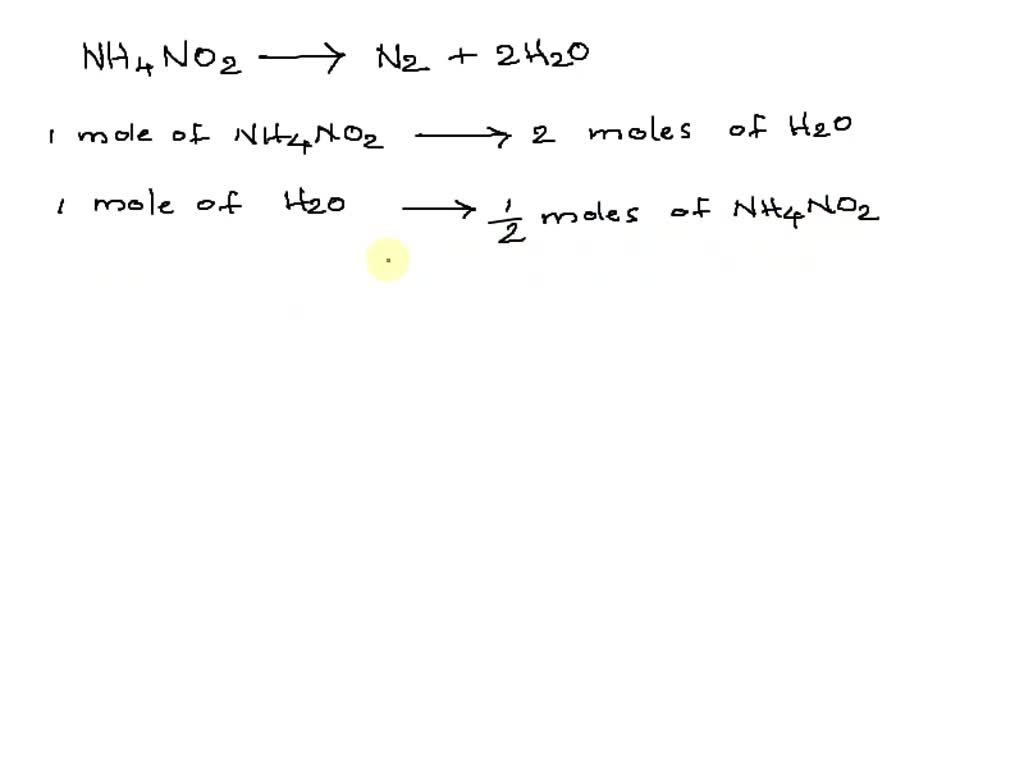
Next on our list is Dinitrogen Monoxide. This one has an even more intriguing name, doesn't it? But you might actually know it by a more common, and much more fun, nickname: Nitrous Oxide. Yes, the same stuff that makes whipped cream so fluffy!
Dinitrogen Monoxide, or Nitrous Oxide as some prefer, is known for its playful properties. It’s often associated with laughter and a bit of lightheartedness. It brings the "wow" factor to our equation.
And finally, the essential element that ties it all together: Water. Yep, plain old H2O. It might seem ordinary, but Water plays a crucial role in how everything unfolds. It's the unsung hero, the smooth facilitator.
The Grand Unveiling
Now, what happens when these three get together? It’s not about an explosion, so don't worry! Instead, we’re talking about a very specific and rather delightful reaction. It's a controlled release of something quite special.
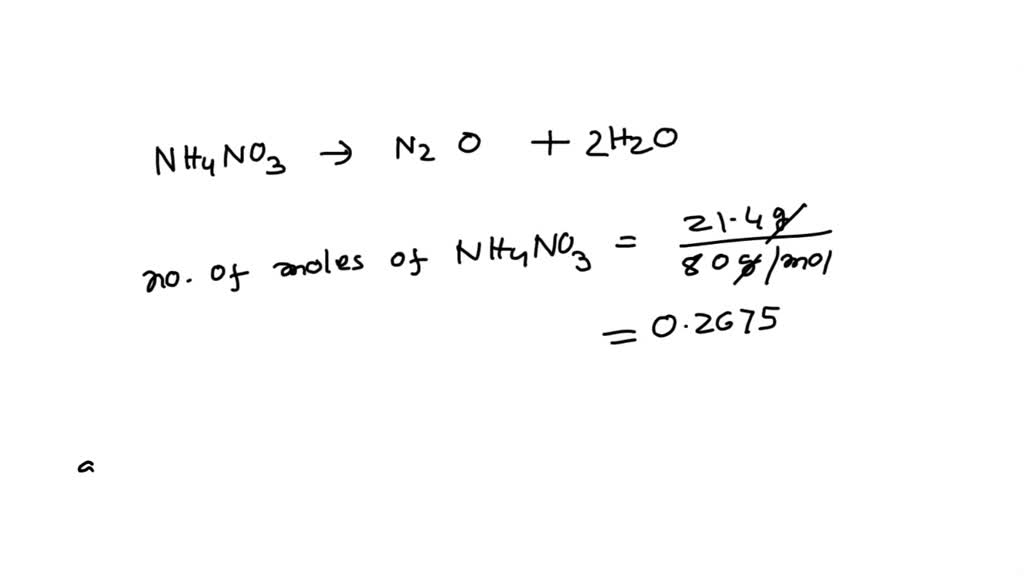
When Ammonium Nitrate meets Dinitrogen Monoxide in the presence of Water, something truly remarkable occurs. It's a chemical dance that produces a beautiful outcome. Imagine a gentle, yet impressive, transformation.
The key here is the word "Aq." In the world of chemistry, "Aq" is a shorthand. It means that the substance is dissolved in Water. So, when we talk about Ammonium Nitrate Aq, it means Ammonium Nitrate is happily mixed with Water.
This aqueous solution of Ammonium Nitrate is then combined with Dinitrogen Monoxide. The Water acts as a medium, allowing the Ammonium Nitrate to interact freely with the gas. It's like setting up a perfect environment for something special to happen.
It's a bit like preparing a delicious soup. You have your main ingredients, and then you add the broth to bring it all together. In our case, the "broth" is the Water!
The magic truly happens when the Dinitrogen Monoxide gas is introduced to this watery mix of Ammonium Nitrate. What do we get? A fascinating display of gas production!
The Hilarious Hatching
So, what’s the entertaining part? It’s the sheer volume and the nature of the gas that's produced. We’re talking about Dinitrogen Monoxide itself, in its purest, gaseous form. And it’s generated in a rather impressive amount.
This process is often used to create a concentrated source of Nitrous Oxide. Think of it as producing a lot of laughing gas, but in a controlled and scientific way. The reaction is efficient and yields a pure product.
The visual can be quite striking. Imagine a container where this reaction is taking place. You'll see bubbles forming, indicating the release of the gas. It's a dynamic and engaging process to witness.
And why is it entertaining? Because Dinitrogen Monoxide is known for its unusual properties. While we're not advocating for inhaling it, its association with merriment is undeniable. The very idea of generating this gas in significant quantities is inherently intriguing.
It’s the unexpectedness of it all. You take these common substances, and with a little bit of chemical know-how, you create something that has a reputation for being… well, fun! It’s a testament to the hidden wonders within everyday chemistry.
The "Aq" part is essential. Without the Water, the Ammonium Nitrate might not react as effectively or as cleanly. Water makes the whole thing work smoothly, like a well-oiled machine.
What Makes it So Special?
What makes this particular combination special is its relative simplicity and its impressive outcome. It’s a classic example of how chemistry can be both practical and visually captivating.
The purity of the Dinitrogen Monoxide produced is a key factor. This isn't just any old gas; it's a specific compound with unique applications. And seeing it generated from seemingly ordinary materials is a real thrill.
Think of it as a science demonstration that’s both educational and entertaining. It sparks curiosity and makes people think about the chemical world around them. It's a conversation starter, that's for sure!
It’s the transformation that’s so engaging. You start with solids and liquids, and you end up with a gas. It’s a change of state that’s captivating to observe. It’s like watching something quite ordinary become extraordinary.
And the fact that it involves Ammonium Nitrate, a compound with a bit of a reputation itself, adds another layer of interest. While this reaction is safe and controlled, the association with energetic applications makes it more exciting.
It’s a little bit of science alchemy. Turning common elements into something that’s recognized for its unique, and often amusing, properties.
The simplicity of the setup, when done correctly, is also appealing. It doesn't require highly specialized equipment. This makes the concept more accessible and, therefore, more intriguing to a wider audience.
It's the perfect blend of ingredients. Ammonium Nitrate providing the nitrogen and oxygen, Water acting as the perfect solvent, and Dinitrogen Monoxide being the star gaseous product. Each component plays its part beautifully.
The Curious Case of Gas Production
The production of Dinitrogen Monoxide from this reaction is a significant aspect. This gas has various uses, from medical anesthesia to food production. So, it's not just about the fun; it's about creating a useful substance.

Seeing this process in action can demystify a bit of chemistry. It shows that these reactions aren't just theoretical concepts; they are tangible and observable events. It’s real-world science in action!
The visual cues are important too. The bubbling, the steady stream of gas – these are all indicators of a successful and ongoing reaction. It’s a dynamic process that keeps you watching.
And let’s not forget the "Aq" again. It’s a subtle but vital detail. It signifies that the Ammonium Nitrate is in a solution, ready to interact. This makes the entire process more controlled and predictable.
It's a great way to understand how different substances behave when combined. The way Ammonium Nitrate dissolves and then facilitates the release of Dinitrogen Monoxide is a beautiful illustration of chemical principles.
So, the next time you hear about Ammonium Nitrate Aq and Dinitrogen Monoxide, remember this fascinating combination. It’s a simple recipe for a scientifically engaging and, dare we say, amusing outcome!
It's a reminder that even the most common substances can hold surprising secrets, waiting to be discovered through the wonders of chemistry.
