Ammonium Chloride And Sodium Hydroxide Balanced Equation
Ever wondered what happens when you mix a couple of common household-ish chemicals? You know, the stuff you might see in a science lab demonstration or even find lurking in some everyday products? Well, let's dive into one of those neat little chemical reactions: the one between ammonium chloride and sodium hydroxide. It sounds a bit science-y, I know, but stick with me, it's actually pretty cool!
So, what are these two players, anyway? Ammonium chloride, if you’ve ever had a weirdly salty cough drop or used certain types of fertilizer, you might have encountered its vibes. It's basically a salt, and in the world of chemistry, salts are formed when an acid and a base have a little chat and neutralize each other. Sodium hydroxide, on the other hand, is what many people know as lye. Yep, that stuff used in soap making and drain cleaners. It’s a strong base, meaning it's pretty eager to react and give up some of its hydroxide ions.
Now, when you bring these two together, things get interesting. It's not like they just sit there and wave hello. Oh no. They get down to business and rearrange themselves. Think of it like a chemical square dance, where partners switch up and create something entirely new. The original partners are ammonium chloride (NH₄Cl) and sodium hydroxide (NaOH).
What’s so exciting about this particular dance? Well, the star of the show is the gas that gets produced: ammonia. You might have smelled ammonia before – it has a pretty pungent, sharp odor, kind of like some cleaning supplies. That distinctive smell is a tell-tale sign that this reaction is happening!
The Chemical Shuffle: What's Actually Happening?
Let's break down the "who's dancing with whom." In the world of chemistry, we often write down these interactions as a balanced equation. Don't let the word "balanced" scare you! It just means that on both sides of the reaction (before and after the shuffle), we have the same number of each type of atom. It’s like making sure you have the same number of red socks and blue socks before and after you put them all in the laundry basket.
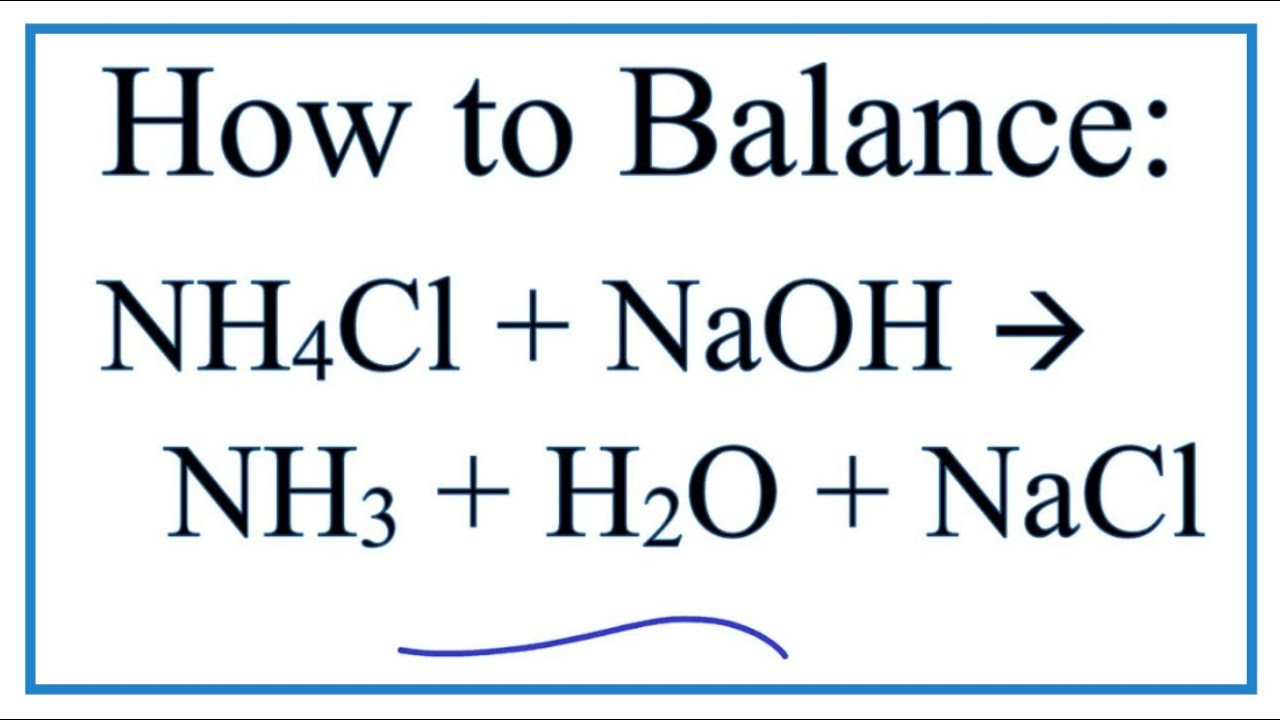
The equation for our ammonium chloride and sodium hydroxide reaction looks like this:

NH₄Cl + NaOH → NH₃ + H₂O + NaCl
Whoa, what does all that mean? Let’s decode the hieroglyphics, shall we?
- NH₄Cl is our ammonium chloride. Think of it as a pair of ammonium ions (NH₄⁺) and chloride ions (Cl⁻) holding hands.
- NaOH is our sodium hydroxide. Here, we have sodium ions (Na⁺) and hydroxide ions (OH⁻) doing their thing.
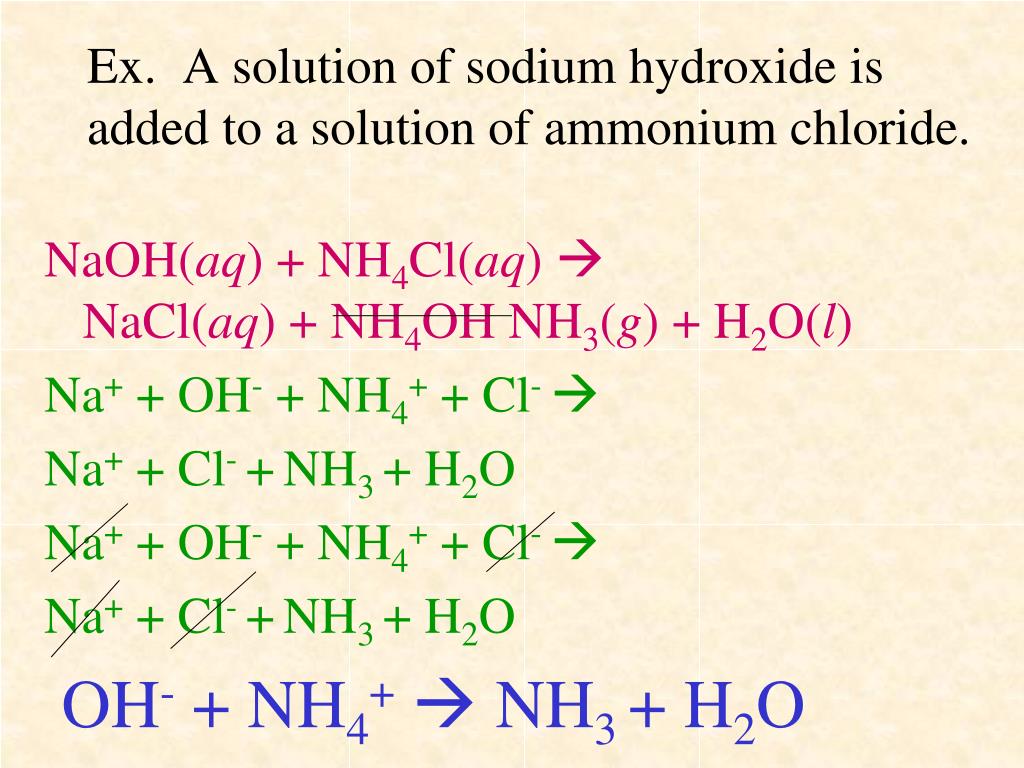
When they meet, it’s like they realize they’d be happier with different partners. The sodium ion (Na⁺) from the sodium hydroxide decides it quite likes the chloride ion (Cl⁻) from the ammonium chloride. They form a little bond and become sodium chloride (NaCl). Yes, the same stuff as table salt! How cool is that? We’re making salt out of a chemical reaction involving a strong base and another salt.

But wait, there's more! What about the other bits? The ammonium ion (NH₄⁺) and the hydroxide ion (OH⁻) are left. They get together, but they're a bit unstable in this form. Instead, they break apart. The hydroxide ion (OH⁻) is happy to lend a hydrogen atom (H⁺) to the ammonium ion (NH₄⁺). This transforms the ammonium ion into ammonia (NH₃) – our pungent gas! And what’s left from the hydroxide ion? Just a lone oxygen and hydrogen, which team up to form water (H₂O).
Why Is This Reaction So Interesting?
There are a few reasons why this particular chemical dance is worth a second glance. First off, it’s a classic example of a double displacement reaction, but with a twist. In a typical double displacement, you’d just swap partners and end up with two new salts. Here, one of the products – ammonia – is a gas. This makes the reaction really observable and, frankly, a bit dramatic!
Think of it like this: imagine you have two couples at a party. Couple A is Mr. and Mrs. Red, and Couple B is Mr. and Mrs. Blue. In a regular double displacement, Mr. Red might dance with Mrs. Blue, and Mr. Blue might dance with Mrs. Red, forming two new couples. But in our chemical reaction, it's like Mr. Red and Mrs. Blue decide to do something completely different. They turn into a puff of smoke (ammonia gas) and a puddle of water, leaving Mr. Blue and Mrs. Red to become a new, stable couple (sodium chloride).
The fact that we get a gas out of it is a big deal. It means the reaction is happening quickly and noticeably. You can often see bubbles forming as the ammonia gas escapes. It's a visual cue that the chemical transformation is underway, which is always a bit more exciting than a reaction that just quietly sits there.

Secondly, it highlights the concept of acid-base reactions, even though ammonium chloride isn't what we’d typically call a strong acid. Ammonium chloride is the salt of a strong acid (hydrochloric acid) and a weak base (ammonia). When it meets a strong base like sodium hydroxide, it can actually behave like an acid by donating a proton (H⁺) to the hydroxide ion. This ability of certain salts to act as acids or bases depending on the situation is a fascinating part of chemistry.
Practical Uses and Cool Connections
So, is this reaction just for lab geeks to play with? Not exactly! While you probably shouldn't be mixing drain cleaner and weirdly salty cough drops in your bathroom, the products and principles involved are super relevant.
As we saw, we produce sodium chloride, which is, well, table salt. While we don't usually make it this way, it’s a reminder that common substances can be formed from less common reactions.
The ammonia gas that's released is a vital industrial chemical. It’s used in everything from fertilizers that help grow our food to refrigerants that keep things cool, and even in some cleaning products (though usually in a diluted form!). Understanding how to produce ammonia, even through a simple reaction like this, opens doors to understanding larger-scale industrial processes.
And what about the water? Well, water is, you know, water! It's the universal solvent and essential for life. It’s always a nice bonus when a chemical reaction produces something so fundamental and harmless.
The balanced equation itself is a powerful tool. It allows chemists to predict exactly how much of each substance they'll need and how much product they'll get. It’s like having a recipe that guarantees you won’t end up with too much of one ingredient or not enough of another when you’re baking a cake. For large-scale industrial processes, getting these ratios right is crucial for efficiency and safety.
The Takeaway?
So, the next time you hear about ammonium chloride and sodium hydroxide reacting, you can think of it not just as a jumble of letters and numbers, but as a dynamic chemical dance. It’s a process where familiar partners rearrange themselves, releasing a pungent gas, creating the salt we eat, and a bit of water. It’s a great little peek into how the world around us works, one chemical reaction at a time. Pretty neat, huh?
