Amine That Results From The Base Hydrolysis Of Acetaminophen
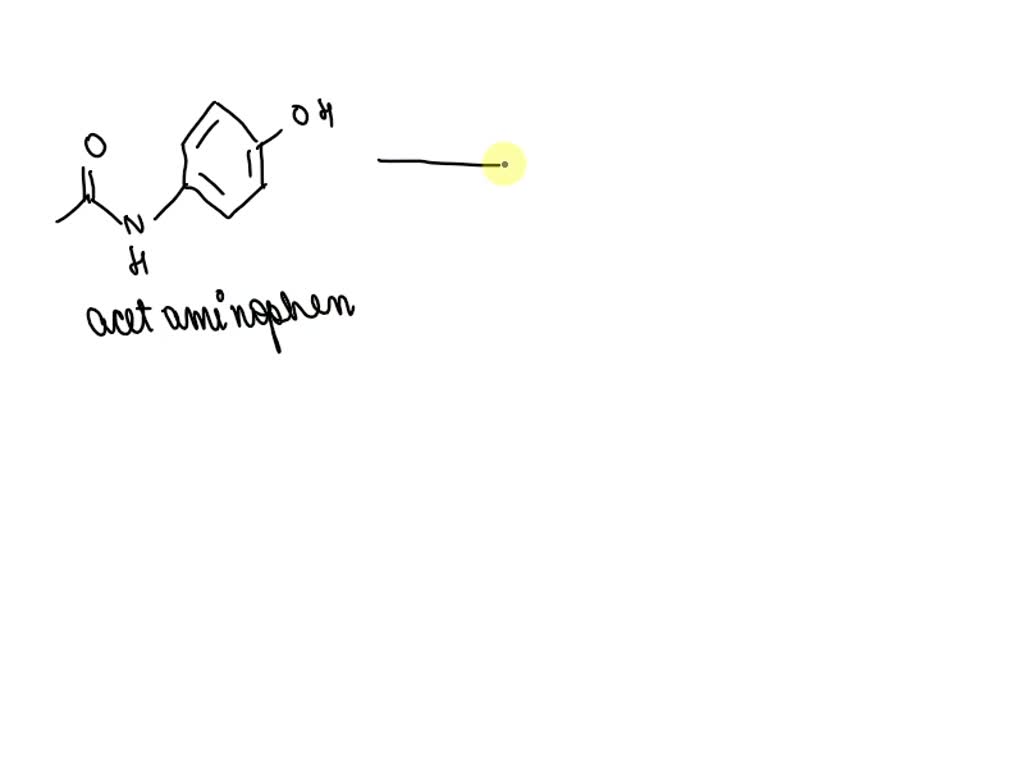
Ever wondered what happens to your trusty pain reliever, Acetaminophen, when it meets a little bit of mischief, like a strong base? Well, prepare for a tiny chemical adventure! It turns out that when Acetaminophen takes a dip in a bubbly, basic bath, something pretty cool happens. A brand new molecule pops into existence, and it’s called... let's just say it's a bit of a rockstar in the chemistry world.
This new kid on the block is an amine. Now, don't let the fancy name scare you! Think of it as a special ingredient that changes how our original molecule behaves. It’s like giving Acetaminophen a little makeover, and the result is a whole new personality. This transformation is all thanks to a process called base hydrolysis.
Imagine Acetaminophen is a building block. When the base comes along, it’s like a friendly demolition crew. They don’t smash everything to bits, though. Oh no, they’re quite precise! They gently break one of the bonds in Acetaminophen, like carefully unsnapping a Lego piece.
And what do we get after that little un-snapping? We get our star, the amine! It’s a fascinating transformation, a little bit of chemical magic happening right before our eyes. This isn't just some obscure reaction for scientists in lab coats. It's a glimpse into the hidden world of how everyday things can change.
So, what makes this particular amine so special? Why should you care about what happens when Acetaminophen gets a little wet and wild with a base? Well, it's all about the surprises. Chemistry often throws us delightful curveballs, and this is one of them.
This amine, the one that emerges from the smoky (or rather, watery) aftermath of Acetaminophen’s base bath, is actually quite an important character in its own right. It's not just a byproduct; it’s a molecule with its own story to tell and its own set of talents.
Think of it like this: you eat an apple, and it gives you energy. But what if, as your body processes the apple, it also creates a tiny, sparkly dust that can make flowers bloom brighter? That's kind of the vibe we're going for here. This amine isn't just hanging around; it's got potential!
The base hydrolysis of Acetaminophen is a classic example in organic chemistry. It’s one of those reactions that, once you see it, you can’t unsee it. It opens up a whole new perspective on how molecules interact.

Let’s talk about the name again. Acetaminophen. It’s a name you probably recognize. It’s in those over-the-counter remedies that help you when you’re feeling under the weather. It’s familiar, comforting even.
And then, amine. This is where the plot thickens! Amines are a big family of chemical compounds. They’re found all over the place, from the building blocks of life itself to the ingredients in some of your favorite snacks (yes, really!).
So, when Acetaminophen meets a base and poof – an amine is born, it’s like a familiar character in a story suddenly revealing a hidden superpower. It’s exciting! It’s unexpected! It makes you want to know more.
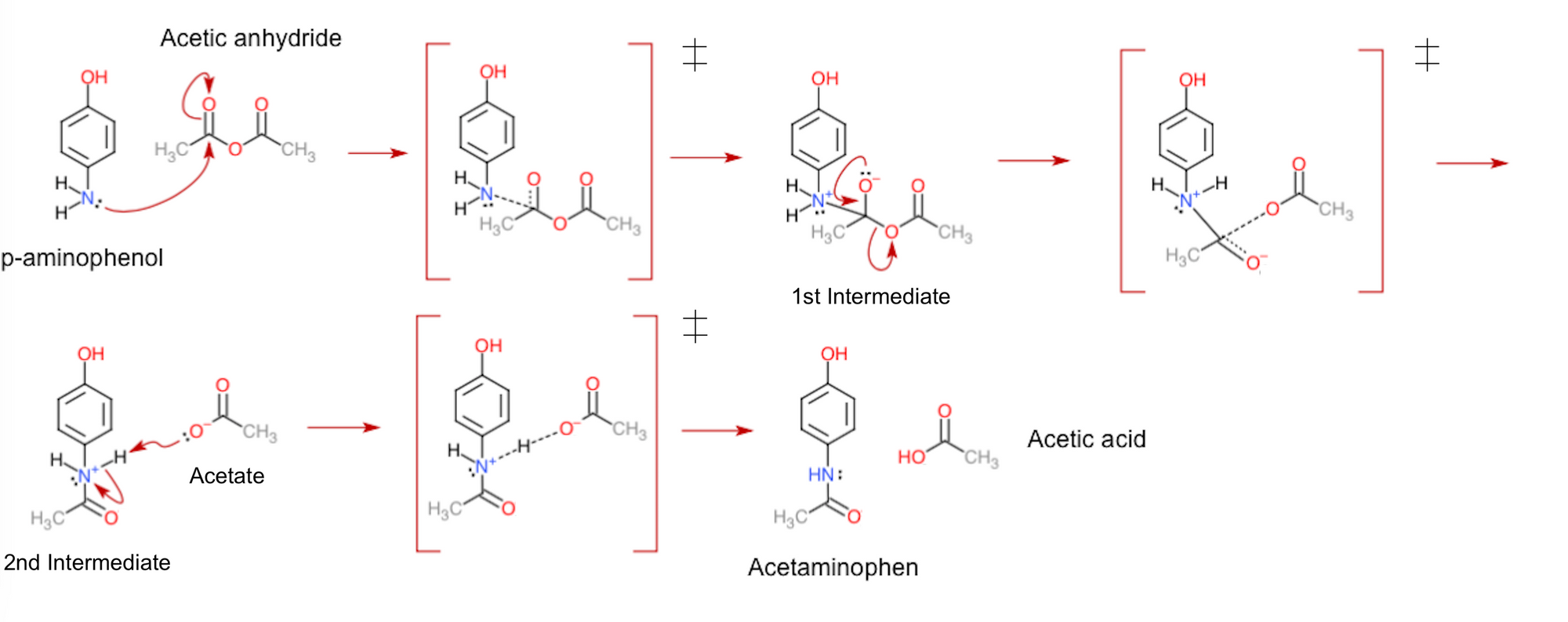
The actual process of base hydrolysis is pretty neat. The base, like sodium hydroxide (a strong alkali), is a bit of a molecular matchmaker. It sees a specific bond in Acetaminophen that's ripe for breaking.
With a gentle nudge (or a strong chemical push!), the base helps to break that bond. This usually involves water molecules playing a supporting role. They get in there, insert themselves, and help to split things up. It's a coordinated effort!
And what’s left behind? Not just fragments, but our lovely new amine! It’s a clean break, a neat transformation. This isn't messy chaos; it's elegant chemistry.

The amine that results from this reaction is actually quite a simple structure. It has a nitrogen atom attached to some carbon atoms. But don't let its simplicity fool you. This fundamental structure is the key to a whole world of chemical possibilities.
Why is it so entertaining? Because it’s relatable! We all know Acetaminophen. We all understand the idea of something changing into something else. This reaction bridges the gap between the everyday and the scientific.
It's like watching a caterpillar transform into a butterfly. You know the caterpillar, you recognize it. But the butterfly? That's a whole new level of wonder. This amine is the butterfly of the Acetaminophen world.
And the base hydrolysis part? That’s the magical chrysalis! It’s the process that allows for this beautiful metamorphosis. It’s the secret ingredient that makes the transformation possible.
What makes it special? It’s the unexpected nature of it. You pop a pill to feel better, and behind the scenes, chemistry is doing its thing, creating these fascinating new molecules. It’s a reminder that even the most familiar things have hidden depths.
This specific amine is often referred to by a more scientific name, like para-aminophenol. But honestly, for our purposes, thinking of it as the "miracle child" of Acetaminophen and a base is much more fun, right?
The para-aminophenol is actually an intermediate in the industrial production of other important chemicals. So, it's not just a pretty face in a test tube; it has practical applications! It’s a building block for even more interesting things.
Imagine a domino effect. One reaction leads to another, and then another. The amine created from Acetaminophen’s base hydrolysis is like the first domino that sets off a whole chain of exciting chemical events.
This is where the true entertainment lies – in the cascading possibilities. It's like unlocking a secret level in a video game. Suddenly, the world of chemistry opens up in new and surprising ways.
So, next time you reach for that familiar box of pain relief, take a moment to appreciate the unseen chemistry. The base hydrolysis of Acetaminophen isn't just a laboratory procedure; it's a miniature marvel.
It’s a testament to the dynamic nature of molecules. They’re not static; they’re constantly interacting, transforming, and creating. And this particular amine is a shining example of that playful molecular dance.
The simplicity of the reactants – a common drug and a common base – leading to the formation of a significant amine is what makes this reaction so captivating. It’s accessible. It’s understandable.
It’s the kind of chemistry that sparks curiosity. It makes you wonder, "What else can happen?" And that, my friends, is the most exciting part of all. This little chemical story is just the beginning.
Think about the journey of these molecules. From raw materials to a useful drug, and then, under specific conditions, transforming into something else with its own unique properties. It's a narrative of change and adaptation.
And our star, the amine, is a key character in this ongoing story. It’s a testament to the fact that chemistry is not just about formulas and equations. It’s about transformation, potential, and the endless possibilities that arise when molecules get together.
So, while you're enjoying the relief Acetaminophen provides, remember the hidden drama. The base hydrolysis, the breakout amine – it’s a little slice of chemical wonder, just waiting to be discovered. It's a reminder that even the mundane can be extraordinary.
Perhaps this tiny tale will inspire you to look at the world around you a little differently. To see the chemistry in action, even in the simplest of things. The amine from Acetaminophen is a friendly invitation to explore more. Dive in and see what other molecular marvels await!
