Alpha D Glucose And Beta D Glucose Are Enantiomers
Hey there, sugar lovers! Ever wonder what makes sugar… well, sugar? It's a wild world out there, filled with molecules that look almost identical but are, like, total opposites. Today, we're diving into the sugary universe of glucose. Specifically, we're gonna chat about two versions: Alpha D Glucose and Beta D Glucose. Sounds fancy, right? But trust me, it’s way more fun than it sounds.
Think of it like this. You've got your best friend. You look a lot alike, right? Maybe you have the same hair color. Same smile. But if you shake hands, your right hand and their left hand, that's a little different. Or maybe you're mirror images. That's kinda what Alpha and Beta D Glucose are doing. They're like molecular twins, but with a twist.
The Tiny Difference That's a Big Deal
So, what's the actual difference? It all boils down to one tiny part. Imagine a glucose molecule is like a little house. It has lots of windows and doors. In this house, there's a special spot. It's called the anomeric carbon. On this carbon, there’s a little thingy – an -OH group. You know, that part that makes things… well, watery. Chemists love their -OH groups.
In Alpha D Glucose, this -OH group sticks out in a certain direction. Let's say it points "down." Now, in Beta D Glucose, this exact same -OH group, on the exact same carbon, points "up." That’s it! Seriously. That’s the whole deal. A subtle flip, and bam! You’ve got a different molecule.
This is what we call enantiomers. It’s a fancy word for molecules that are non-superimposable mirror images of each other. Like your left hand and your right hand. You can put them next to each other, and they look the same, but you can’t stack them perfectly on top of each other. They’re reflections. Pretty neat, huh?
Why Does This Even Matter?
You might be thinking, "So what? It's just a tiny flip. Why should I care?" Well, my friend, that tiny flip makes a huge difference. It affects how these sugars behave. It's like the difference between a key that opens a lock and a key that looks identical but is just slightly wrong – it won't work.
Think about it in terms of food. Our bodies are picky eaters when it comes to sugars. They have special little "docking stations" or enzymes that are designed to recognize specific shapes. The shape of Alpha D Glucose is different enough from Beta D Glucose that our bodies might process them differently.
For example, when we eat starch, which is a long chain of glucose molecules, the way those glucose units are linked together matters. If they're linked in the "Alpha" way, our digestive system can break them down easily. That’s why we get energy from bread and potatoes. Yay carbs!

But what about cellulose? That’s the stuff that makes plants rigid. It’s also made of glucose! But this time, the glucose units are linked in the "Beta" way. And guess what? Most of us? We can’t digest it. Yup. Cows can do it. Rabbits can do it. We just… can’t. Our digestive enzymes aren't built for those beta links. Sad trombone sound.
The Sweet, Sweet World of Sugars
Glucose itself is the powerhouse of our bodies. It's the primary source of energy for our cells. When you're running a marathon, or even just thinking really hard (guilty!), your brain is chugging down glucose. So, understanding its forms is like understanding the fuel that keeps you going.
And these Alpha and Beta forms? They're everywhere! They're not just in boring lab experiments. They're in the foods you eat. They're in the plants around you. They're even in the building blocks of life itself!
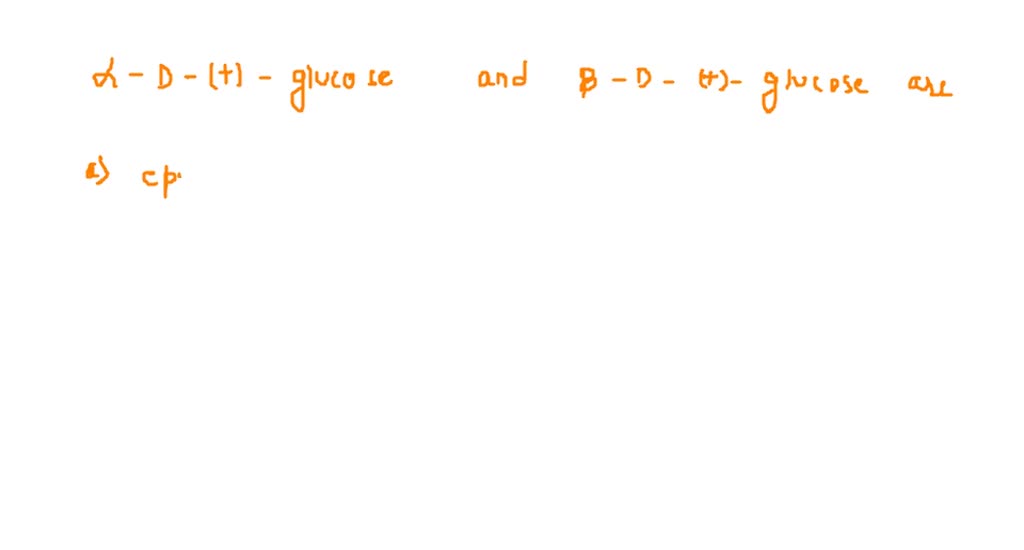
Did you know that when glucose molecules decide to link up and form rings, they can do it in either the Alpha or Beta form? It's like choosing a handshake for your sugar molecules. And get this, in a solution, these two forms are constantly interconverting. It's a sugary dance!
Imagine a bunch of glucose molecules in a glass of water. They're all chilling, bumping into each other. Some are Alpha, some are Beta. And they're flipping back and forth, like a crowd at a concert trying to decide which way to jump. It's a dynamic equilibrium, as the science folks say. But I like to think of it as sugar's existential crisis.
It's All About the Shape!
The shape of a molecule is everything in chemistry. It dictates how it interacts with other molecules. Think of a lock and key. Same idea. The precise 3D arrangement of atoms matters.

Alpha D Glucose and Beta D Glucose are so close, yet so different. They’re like fraternal twins who decided to wear slightly different outfits. One might be a bit more formal, the other a bit more casual, and they might get invited to different parties.
This concept of stereoisomers, like enantiomers, is super important. It helps explain why drugs work the way they do. Why one version of a medicine might be helpful and another could be useless, or even harmful! It's all about how well that molecule fits into its target in your body.
So, next time you're enjoying a sweet treat, spare a thought for the humble glucose molecule. It's not just a simple sugar. It’s a complex character with different personalities, all thanks to a tiny, but significant, twist in its structure. Alpha and Beta D Glucose – proof that even the smallest differences can create a whole new world of possibilities. Isn't science just the coolest?
