Allosteric Enzyme Regulation Is Usually Associated With
Imagine your body is a super-duper, bustling city, and enzymes are the tiny, hardworking citizens responsible for getting everything done. They're like the chefs in the city's kitchens, whipping up all sorts of essential molecules, or the delivery drivers, zipping around to bring vital supplies where they're needed. Without them, the city would grind to a halt faster than you can say "slow metabolism!"
Now, how do these enzyme citizens know when to speed up their work, or when to take a much-needed break? That's where a really cool concept called allosteric regulation swoops in, like a helpful city planner with a walkie-talkie.
Think of it this way: our enzyme friends aren't just blindly working away. They've got special spots on them, like little docking stations or secret hatches, that can receive messages. These messages don't come from the usual "ingredients" they're working with. Nope! They come from entirely different molecules, acting like VIP guests arriving at the enzyme's party.
When these VIP molecules show up and park themselves at these special docking stations, something magical happens. It's like they're whispering secrets to the enzyme, telling it what to do. And here's the super neat part: these VIPs aren't part of the main ingredient list for the enzyme's current job. They're like someone arriving at a bakery and saying, "Hey bakers, you're doing a fantastic job with the croissants today, but maybe put on some extra jazz music for a little pep!"
The Famous Friends of Allosteric Regulation
So, who are these important VIPs that hang out with our allosteric enzymes? They often show up in the context of feedback inhibition. It sounds a bit dramatic, doesn't it? Like a detective movie plot! But it's actually super clever and incredibly common in biology.
Imagine you're making cookies, and you've got a whole chain of steps. The first enzyme starts by mixing flour and sugar. The second enzyme adds eggs. The third one bakes them. Now, let's say you've baked way too many cookies and your cookie jar is overflowing! You don't need any more cookies, right?
This is where feedback inhibition shines! The cookies themselves, or rather, the product of the entire cookie-making process, can send a message back to the very first enzyme in the chain. This message is like a polite but firm "Hold up there, cookie chef! We've got enough deliciousness for now!"
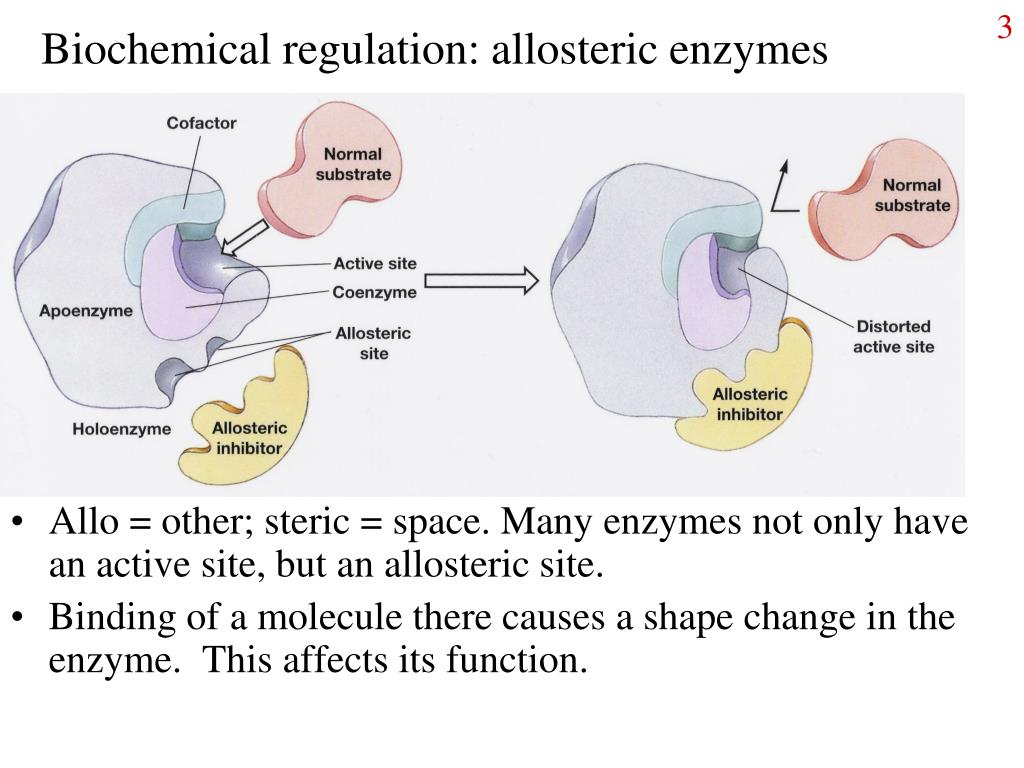
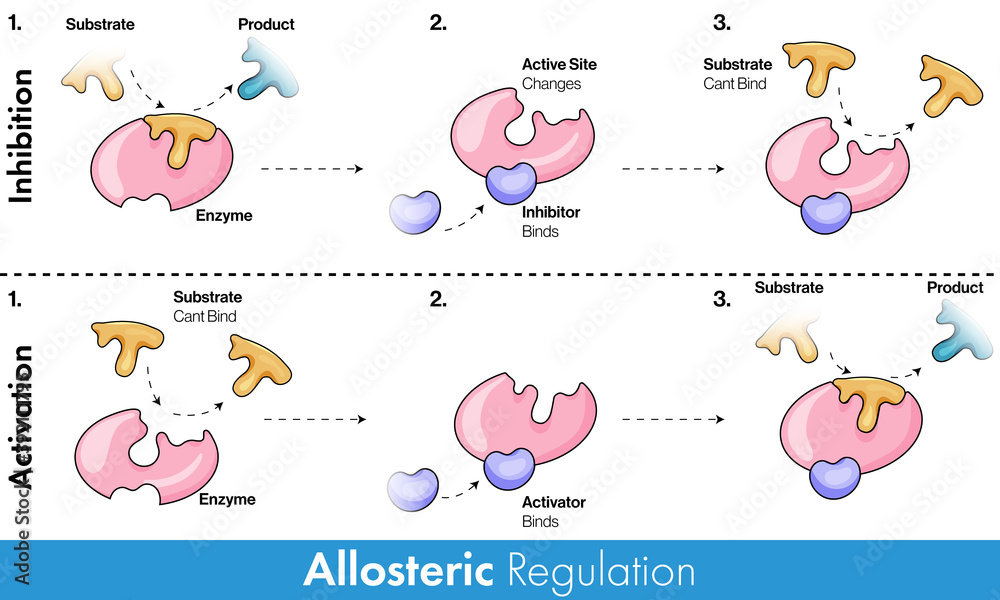
This molecule, the finished cookie in our analogy, then acts as an allosteric effector. It attaches to a special spot on the first cookie-making enzyme, not where the flour and sugar usually go, but somewhere else. And guess what? By attaching there, it changes the shape of the enzyme just enough that it can't do its job anymore. It’s like putting a little "closed for business" sign on the enzyme's main work area.
It's All About the "Other" Spot!
This is the core idea of allosteric regulation: a molecule binding to an enzyme at a site other than the active site (where the real action happens) causes a change in the enzyme's shape and, consequently, its activity. It's like a remote control for the enzyme's performance!
Think of a really fancy faucet with a temperature dial and a flow knob. The active site is where the water comes out. But imagine there's a little sensor on the side that can detect the air temperature. If the air gets too hot, this sensor might signal to the faucet to reduce the water flow, even though you haven't touched the main knobs. That's kind of how allosteric regulation works!
This "other spot" is called the allosteric site. And the molecule that binds there? That's your allosteric effector. They're like the remote control's buttons, and the enzyme is the TV, with its activity being the channel it’s showing.

In our cookie example, the cookies (the product) are the allosteric effectors. They bind to the allosteric site on the first enzyme (the cookie-making enzyme) and cause it to slow down or stop. This prevents you from making a ridiculous mountain of cookies that nobody can eat!
Keeping Things in Harmony
So, allosteric enzyme regulation is usually associated with keeping biological processes running smoothly. It's about preventing a buildup of unnecessary products and making sure that just the right amount of things are being made, at just the right time.
It’s like having a team of musicians in an orchestra. They all have their instruments (their active sites), but they also have a conductor (the allosteric effectors). The conductor doesn't play an instrument, but by signaling, they can make the orchestra play louder, softer, faster, or slower, ensuring a beautiful and coordinated performance.
This feedback mechanism is crucial for maintaining a stable internal environment in your body. It's called homeostasis, and it's a fancy word for keeping everything balanced and just right, like a perfectly tuned instrument.
Without allosteric regulation, your body could be like a car with a stuck accelerator – things would just keep going and going, potentially leading to all sorts of problems. Imagine your body churning out way too much of a certain molecule, filling up all the available "parking spots" and causing chaos!

This is why allosteric regulation is so incredibly important. It's a sophisticated, built-in quality control system, ensuring that your cellular city functions efficiently and harmoniously. It’s a testament to the incredible ingenuity of nature!
More Than Just Stopping Things
But it's not just about stopping things! Allosteric regulation can also be used to speed things up. Sometimes, a molecule will bind to an allosteric site and actually activate an enzyme, making it work even faster. It's like giving the cookie-making enzyme a shot of espresso!
This is often seen when a signal molecule indicates that a certain pathway needs to be turned on quickly. For example, if your body senses a sudden need for energy, specific molecules can bind to enzymes and ramp up their production of energy-generating compounds. It’s like the conductor shouting, "Let's hear that crescendo!"
So, while feedback inhibition is a very common partner for allosteric regulation, it's not the only act in town. Activation is just as vital for keeping your biological city humming along.

The beauty of it lies in its sensitivity. A tiny change in the concentration of these allosteric effectors can lead to a big change in enzyme activity. It’s like a light switch – a small flick can turn on or off a whole room!
This fine-tuning allows your body to respond very precisely to its needs. It’s a constant dance of activation and inhibition, all orchestrated by these clever allosteric interactions.
So, the next time you hear about allosteric enzyme regulation, remember the busy city, the diligent enzyme citizens, and the wise allosteric effectors acting as messengers. They are the unsung heroes ensuring everything runs like a well-oiled, happy machine, keeping you healthy and energized for all your adventures!
It's a beautiful system, isn't it? A perfect example of how complex processes can be managed with elegant simplicity. It’s truly a marvel of biological engineering, and a key player in the grand symphony of life!
