All The Following Are Acid Ionization Reactions Except

Imagine you’re at a party, and someone asks you to name all the people who brought a dessert, except for, well, Bob. Bob brought a weirdly shaped rock, remember? This is kind of like what we’re talking about today, but instead of desserts and rocks, we’re chatting about acids and how they behave. Acids are these cool chemical critters that love to share. Specifically, they share tiny little things called protons. Think of protons like little invisible bouncy balls that acids happily toss around in water. When an acid tosses one of these protons into water, it’s like a mini-explosion of awesomeness, creating something new and exciting. We call this tossing a proton-sharing party or, in fancy science talk, an acid ionization reaction.
So, what does this proton-sharing party look like? Let’s take a common friend, like vinegar. You know, the stuff that makes your salad taste zesty? When vinegar, also known as acetic acid, gets a bit cozy with water, it gets excited. It’s like the vinegar is saying, "Hey water, want a proton? I’ve got loads!" So, it hands one over. The vinegar molecule then becomes a bit of a different character, and the water molecule, now happily holding an extra proton, also changes. This is a classic acid ionization reaction. It’s the acid doing its thing, making things a little more… charged up!
Think about lemon juice. That sharp, tangy taste? That’s thanks to citric acid, another proton-sharer extraordinaire. When you squeeze a lemon into your water, those citric acid molecules are just itching to share their protons with the water. It’s like a friendly game of tag, with protons being the "it." The water molecules are always ready to catch a proton, and when they do, it’s a whole new chemical dance happening. These are all examples of acids being, well, acidic in the way we expect them to be. They are actively donating these tiny, energetic protons.
Now, here’s where things get interesting. Just like at our dessert party, there are some things that might look like they’re participating, but they’re actually doing something else entirely. They might be present, they might even be involved in a chemical reaction, but they aren't doing the specific proton-sharing thing that defines an acid ionization reaction. These are the Bob’s of the chemical world in our analogy – they brought the rock, not the dessert.
So, what kind of things might seem like they’re part of the proton-sharing party but aren’t? Let’s consider something like table salt, or sodium chloride. When you dissolve salt in water, it breaks apart into tiny pieces called ions – a positively charged sodium bit and a negatively charged chloride bit. These bits are already stable and happy. They don't have any protons they're looking to donate to the water. The water might swirl around them, but the salt itself isn't initiating a proton donation. It’s more like they’re just chilling, letting the water molecule do its own thing. It’s a dissolution, not a proton-sharing spree.

Another example could be a reaction where a molecule accepts a proton, but it’s not an acid doing the donating. Imagine a molecule that’s really hungry for protons. It’s like a little proton magnet. While this molecule is definitely involved in a reaction with protons, the source of the proton isn't this molecule itself acting as an acid. The acid ionization reaction is specifically about the acid giving away the proton. If something else is taking the proton, and the thing giving it isn't an acid, then that’s not an acid ionization reaction. It’s like someone bringing a really empty plate to the dessert party – they’re there, but they’re not contributing a dessert.
Sometimes, chemical changes happen that involve rearranging atoms or breaking apart molecules, but without that specific act of an acid releasing a proton. Think about the process of rust forming on your bike. That’s a chemical reaction, for sure! Iron is reacting with oxygen. It’s a transformation, but it’s not the same kind of proton-sharing dance that acids do. It's like the rock Bob brought to the party. It's there, it's interesting, but it's not a dessert. The rock is not a proton donor in this scenario. It’s just a rock.
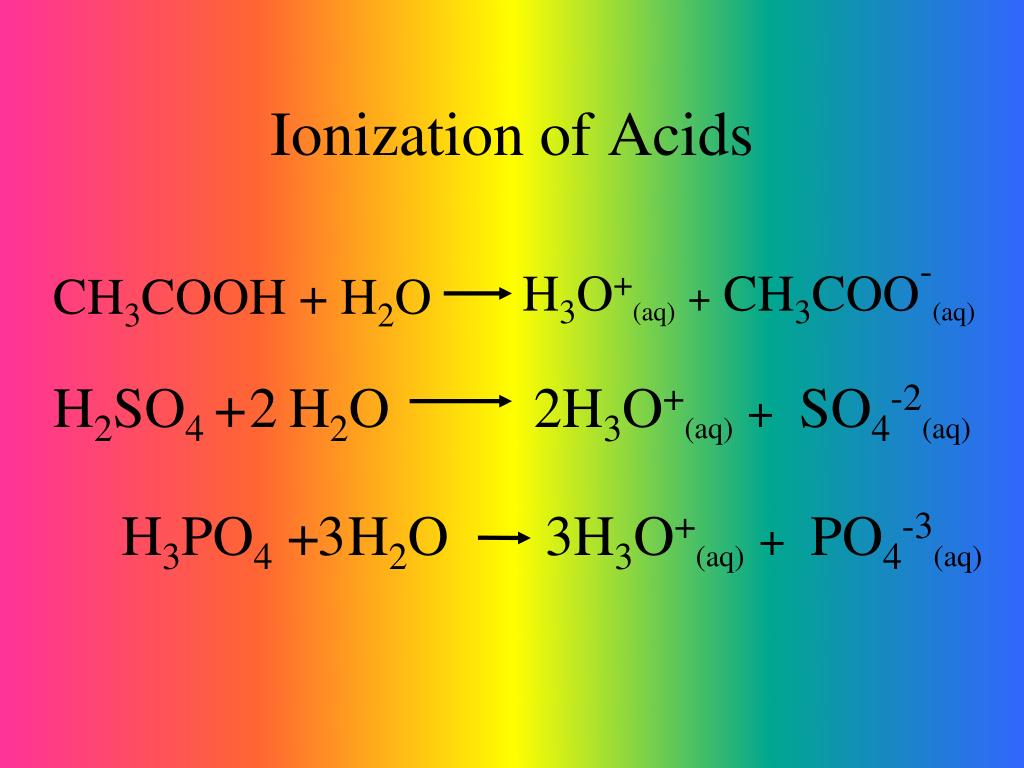
The beauty of chemistry is in these subtle differences. An acid ionization reaction is a very specific event: an acid saying, "Here, have a proton!" and the water (or other solvent) accepting it. It’s the fundamental way acids contribute to the character of a solution, making it, well, acidic. Everything else might be happening in the watery world, but if it's not that proton-sharing action initiated by an acid, it’s not an acid ionization reaction. It's a different chemical story, a different kind of party guest, or maybe just a quiet observer.
So, next time you’re enjoying a sour candy, or marveling at the fizz of baking soda in your kitchen, remember the tiny proton-sharing parties happening. And when you encounter something that’s chemically active but isn’t performing that specific proton-giving dance, you’ll know it’s like Bob at the dessert party – present, but not part of the main event we’re talking about. It's all about that proton exchange, the heart and soul of acid behavior!
