All Resonance Structures Must Be Considered When Assigning Hybridization
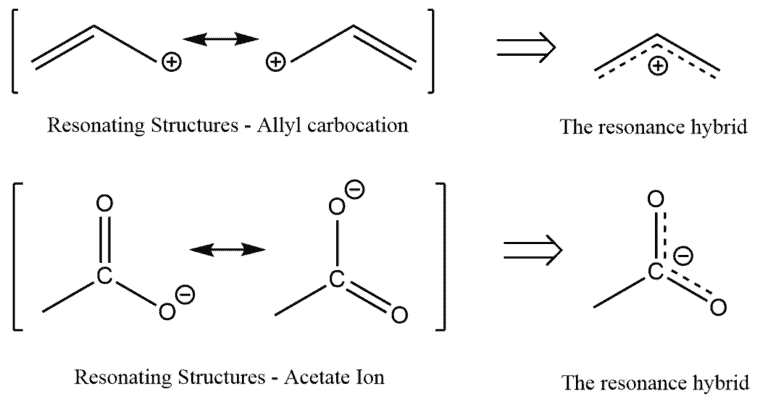
Okay, my fellow science enthusiasts, gather 'round! We're about to dive into a little secret that chemists use to understand how molecules behave, and trust me, it's way cooler than it sounds. Imagine you're trying to describe a really awesome, shape-shifting superhero. You can't just look at them in one pose, right? You have to consider all their different stances, their dynamic movements, their whole awesome repertoire to truly get what they're about. That's exactly what we're doing when we talk about how atoms in a molecule hug each other – it’s called hybridization, and it’s a super important concept.
Now, here's the kicker, the absolutely crucial, can't-miss, gotta-have-it-in-your-life rule: When you're figuring out the hybridization of an atom in a molecule, you absolutely, positively, without a shadow of a doubt, MUST consider ALL the resonance structures! No exceptions! Think of resonance structures like different "costumes" a molecule can wear. One moment it might look like this, the next, with a little electron wiggle, it looks like that! And to truly understand the molecule's "true self," its underlying character, you've got to peek at it in every single one of its costumes.
The Superhero Analogy, Amplified!
Let's say we're talking about our hypothetical superhero, "Captain Charge." Captain Charge has a main form, let's call it the "Classic Cape." This is like one of your basic Lewis structures. It shows the atoms and bonds. But Captain Charge also has a "Stealth Suit" form and a "Power Surge" form. These are like our resonance structures. If you only looked at Captain Charge in his Classic Cape, you might think, "Okay, he's got two arms, two legs, standard superhero stuff." But then you see him in his Stealth Suit, and suddenly, one of his arms has morphed into a grappling hook! Or in his Power Surge form, both his hands are glowing with energy!
If you were trying to explain Captain Charge's fighting style and only looked at his Classic Cape, you'd be missing out on a huge part of his awesome abilities. You wouldn't understand how he can adapt, how he can change his attack patterns. That's the same pitfall you run into if you ignore resonance when assigning hybridization!
So, imagine trying to describe Captain Charge's "hand posture" (which is kind of like our atomic orbitals in hybridization). If you only look at the Classic Cape, you might say his hands are always in a "fist" position. But if you consider the Stealth Suit where one hand is a grappling hook, or the Power Surge where both hands are open and glowing, you realize his hand posture is much more dynamic, much more adaptable! It’s a blend of possibilities! That’s the magic of resonance, and that’s why we can’t ignore it!

Why This Matters (Without Getting Too Technical!)
Think about it like this: When an atom is deciding how to form bonds, it's not just looking at a single static picture. It's looking at the average of all its possible appearances. It's like you trying to decide what to wear. You don't just pick an outfit based on what you look like sitting on the couch. You consider all the places you might go, all the things you might do! You're thinking about being ready for anything!
So, when we assign hybridization, we're essentially saying, "Okay, this atom is preparing to interact with its neighbors." And if that atom can shift its electron distribution – if it can move electrons around to form those different resonance structures – then its hybridization needs to reflect that flexibility, that ability to be in multiple configurations.

It's like trying to guess a person's shoe size by only looking at them while they're wearing ballet slippers. You're going to get a completely wrong idea if they usually wear enormous clown shoes or tiny sparkly sneakers! You need to see their whole footwear collection!
If you only look at one resonance structure, you might be assigning the wrong hybridization. This can lead to all sorts of misunderstandings about the molecule's shape, how it reacts, and even its physical properties (like whether it’s sticky, or slippery, or smells like lavender). And nobody wants to get the molecular smell wrong, right? That’s just embarrassing!
So, the next time you're playing the fun game of assigning hybridization, remember Captain Charge and his many costumes. Take a good, long look at all the possible resonance structures. Consider every wiggle of those electrons. It’s the secret ingredient to truly understanding the molecule, to seeing its full, magnificent, shape-shifting glory. It's about embracing the entire story, not just a single frame. Embrace the resonance, and your hybridization assignments will be as accurate and awesome as a superhero in peak performance! Happy bonding, everyone!
